An Ethical Future for Brain Organoids Takes Shape
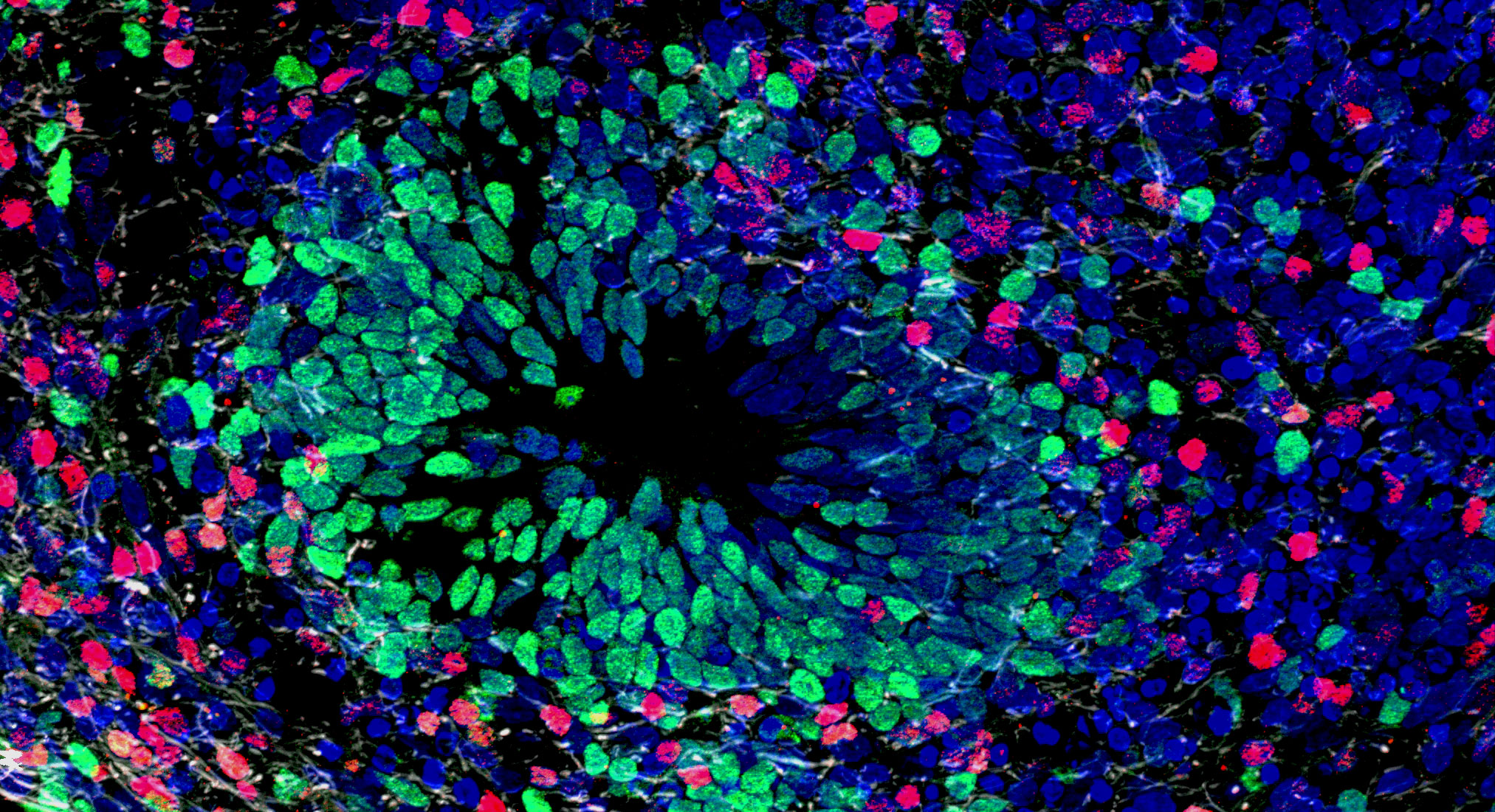
Cells in a cerebral organoid differentiate and form structures related to those seen in embryonic tissues, as seen in this micrograph. The green cells are the progenitors of neurons; the red cells are immature neurons migrating to form a cortical layer. As the similarities between organoids and brains increase, researchers need to pay close attention to the potential for ethical issues.
Alysson Muotri
Introduction
Part of the brain’s allure for scientists is that it is so deeply personal — arguably the core of who we are and what makes us human. But that fact also renders a large share of imaginable experiments on it monstrous, no matter how well intended. Neuroscientists have often had to swallow their frustration and settle for studying the brains of experimental animals or isolated human neurons kept alive in flat dishes — substitutes that come with their own ethical, practical and conceptual limitations.
A new world of possibilities opened in 2008, however, when researchers learned how to create cerebral organoids — tiny blobs grown from human stem cells that self-organize into brainlike structures with electrically active neurons. Though no bigger than a pea, organoids hold enormous promise for improving our understanding of the brain: They can replicate aspects of human development and disease once thought impossible to observe in the laboratory. Scientists have already used organoids to make discoveries about schizophrenia, autism spectrum disorders and the microcephaly caused by the Zika virus.
Yet the study of brain organoids can also be fraught with ethical dilemmas. “In order for it to be a good model, you want it to be as human as possible,” said Hank Greely, a law professor at Stanford University who specializes in ethical and legal issues in the biosciences. “But the more human it gets, the more you’re backing into the same sorts of ethics questions that are the reasons why you can’t just use living humans.”
In the popular imagination, fueled by over-the-top descriptions of organoids as “mini-brains,” these questions often center on whether the tissue might become conscious and experience its unnatural existence as torture. The more immediate, realistic concerns that trouble experts are less sensational but still significant. It also doesn’t help that the study of organoids falls into an odd gap between other areas of research, complicating formal ethical oversight. Still, no one wants to see brain organoids’ potential discarded lightly.
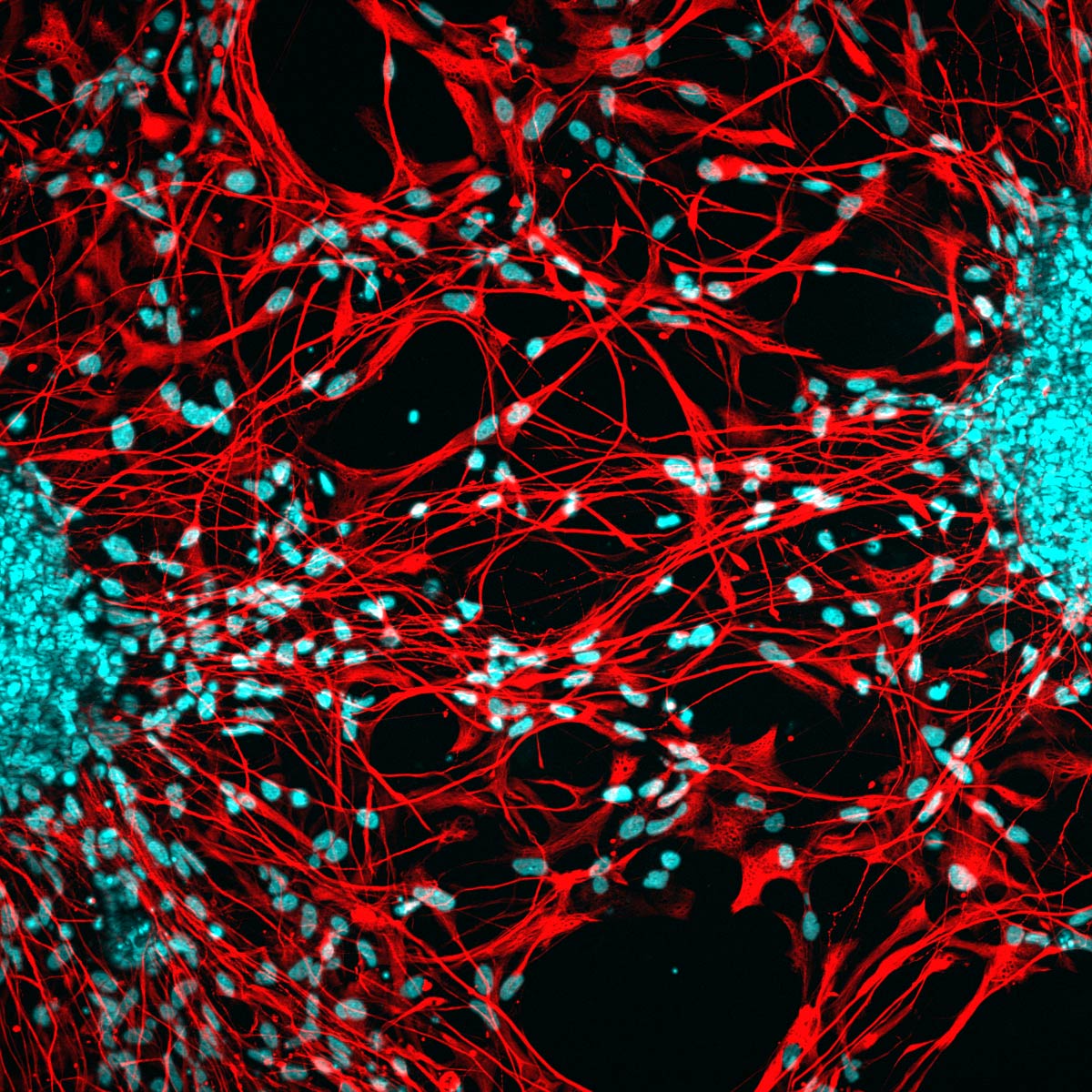
The projecting axons of neurons (red) from a human brain organoid grow out across a culture dish. Their nuclei are stained blue.
Alysson Muotri
Donald O’Rourke, for example, is a neurosurgeon at the University of Pennsylvania’s Perelman School of Medicine who is using organoids, including ones transplanted into rodents, to test treatments for an aggressive type of brain cancer. The ethics of organoid research don’t trouble him at all. “I’m dealing with a deadly disease that kills people in 15 months,” he said. “Here we’ve developed an advanced diagnostic tool to evaluate in real time what therapies might be beneficial. In my mind, that solves ethical problems.”
So organoid science is moving forward. And even if organoids are a long way from feeling pain or becoming conscious, Greely and other ethicists and biologists stress that it’s important to start talking about those possibilities now to steer away from them later. To that end, they’re employing a collaborative approach to bring the science and ethics together early. As a first step, they’re embarking on studies to illuminate the differences between organoids and real brains, and developing benchmarks for comparing them.
Unconscious but Increasingly Complex
There is nearly universal agreement among those familiar with the field that today’s brain organoids, and any likely to be grown in the near future, will not be conscious. “They’re still very, very primitive and rudimentary compared to even the brain of a mouse,” said Han-Chiao Isaac Chen, a professor of neurosurgery at the Perelman School of Medicine, “let alone the brain of a human.”
Even state-of-the-art brain organoids lack the size, structure and interconnectivity of actual human brains. They’re missing important types of cells and lack the blood vessels needed to keep their tissues nourished and healthy. They’re immature: One technique for estimating their developmental age has consistently likened them to the brain of a second-trimester fetus. And perhaps most important, they lack sensory input. “We are who we are because of our experience,” said Hongjun Song, a neuroscientist at the Perelman School of Medicine. “The organoid in a dish doesn’t really have the stimulation to shape the brain circuitry in a way where it’ll develop more like a brain.”
In short, organoids are nowhere near consciousness. “That gives us a safety margin now, for some number of years,” said John Aach, a senior scientist at Harvard University and a colleague of George Church, a prominent geneticist and synthetic biologist who uses organoids in his research.
But everyone in the field recognizes that organoids are getting more complex and realistic. Insoo Hyun, a bioethicist at Case Western Reserve University, recently noted in an online essay that over the next five years or so, researchers will likely try to create organoids with networks of functional blood vessels and a full complement of brain cell types; the organoids may also be designed to emulate particular brain regions, to process neural data inputs, and to produce electrical outputs. Moreover, many of the experiments that researchers are doing or envision trying with organoids involve transplanting them into rats or other laboratory animals, which do have well-developed, functioning nervous systems that the organoids might integrate with. So even if organoids don’t cross the threshold to consciousness or start experiencing pain in the next few years, it’s reasonable to be concerned that they could someday.
An Incomplete Rule Book
Organoid research isn’t developing in a regulatory vacuum. Existing regulatory frameworks for other kinds of biological research offer many ethical protections for organoids, too.
Since brain organoids are grown from induced pluripotent stem cells, they are at least somewhat covered under the existing ethical infrastructure that governs experiments using them. Specific rules and guidelines dictate where scientists can get those cells, and what kinds of lab animals they can transplant them into. (Primates are off the table.) Committees made up of scientists, as well as nonscientists with legal, ethical and social expertise, oversee how the cells are used. Similarly, when organoids get transplanted into animals, they fall under the purview of animal welfare committees.
But the peculiar nature of organoids makes it uncertain how well some of the older systems of rules apply — or even whether they should.

In his laboratory at the University of California, San Diego, the researcher Alysson Muotri, holds a culture dish that contains cerebral organoids.

The organoids — tiny self-organized lumps of tissue that can stand in for human brain tissue in experiments — are currently quite simple, but scientists are working to make them resemble real brain tissue more closely in form and function.
In his laboratory at the University of California, San Diego, the researcher Alysson Muotri holds a culture dish that contains cerebral organoids (left). The organoids (right) — tiny self-organized lumps of cells that can stand in for human brain tissue in experiments — are currently quite simple, but scientists are working to make them resemble parts of real brains more closely in form and function.
Courtesy of Alysson Muotri
Take the “14-day rule,” a guideline that limits how long human embryos can be grown in vitro. Certain structures appear in an embryo 14 days after fertilization, signaling that the nervous system has started to assemble. But the 14-day rule doesn’t work for organoids because there’s no actual embryo involved. Brain organoids follow an entirely different pathway toward achieving neural architecture. Moreover, the whole point of having the organoids is to create brain structures, so that criterion for rejecting them is useless. And so the organoids “have this characteristic flavor, where you know you’re coming close to something that in the past created ethical quandaries,” Aach said.
Organoids don’t fall into a neat category for committee oversight: They’re not exclusively human-subject research, or animal research, or ex vivo stem cell research. “There’s no regulatory structure necessarily for organoids,” Greely said. “That is not a terrible thing right now. But it’s a gap that probably should be filled, in case the organoids do develop in such a way that the concerns are more realistic.”
Ethicists and scientists are now working together to figure out whether organoids warrant their own rules. The ethical discussions are progressing at a fast clip, well ahead of the science. The National Institutes of Health has sponsored meetings and workshops, and there’s been talk of creating another kind of oversight committee. Scientists have reached out privately to ethicists to discuss the particulars of their research: Greely remembers getting emails as early as 2013 from experimentalists seeking to open up a dialogue. And Alysson Muotri, a biologist at the University of California, San Diego, has organized conferences to connect ethicists with both stem cell biologists and consciousness researchers.
Brainstorming Collaborative Ethics
Of particular interest is the Brainstorm Project, a two-year, NIH-funded program co-chaired by Hyun and Jeantine Lunshof, an ethicist at Harvard. It provides institutional support for bringing ethicists and scientists together in small working groups — rather than waiting for them to volunteer — so they can determine which ethical issues to focus on, and which areas of brain organoid research they’d recommend for future NIH funding.
The Brainstorm Project is modeled in part on Lunshof’s day-to-day practices: Since 2006, she has been working with research groups at Harvard’s Wyss Institute for Biologically Inspired Engineering, including the Church laboratory. (She has an office on the lab floor.) She collaborates with the biologists on their research full time, from beginning to end — attending weekly investigator meetings, poring over experimental designs, scanning new papers, and engaging in lively conversation about the methods and goals of their ongoing studies.
It’s a two-way street, she says: She asks questions and learns about the work they’re doing, while they become more familiar with how ethicists think. The regular contact encourages them to consult with her when they think they’ve entered, or are about to enter, an ethical gray zone. “It’s a continuous learning process,” Lunshof said. “And it’s mutual.” This, she added, is where the field of organoid research is headed — toward what she dubs “collaborative ethics.”
She’s careful to differentiate her role from one of judgment. “I am not the ethics police,” she said. “I do not have any formal oversight role. That’s also not my aim.” Instead, collaborative ethics has “the scientists working with us, and us working with them, to find out what the future could be, and what they feel is needed.”
Similarly, the Brainstorm Project is decidedly not an immediate effort to create a set of organoid research guidelines. “It’s too early for that,” Hyun said. Right now, “it’s not really like anyone’s prepared to say, ‘Here’s a line nobody should cross.’” Although he does want to use their discussions to guide future policy, for now it’s about “the need for ethics and science to develop together, to not have ethics always be a reaction to the science, but be involved early on to help shape the direction of the research in a socially responsible way.”
Although the emergence of organoid consciousness, experience of pain, self-recognition and other worrisome traits inevitably comes up in Brainstorm Project meetings, that’s not their central focus — those developments are still too far off. Instead, Lunshof, Hyun and their colleagues want to pinpoint more immediate ethical issues worth considering. Discussions of consciousness are crucial, “but keeping your eye solely on that problem distracts you from other things that may be a lot more pressing and important and surprising,” Hyun said. “So that’s what we’re looking at now.”
Benchmarking the Brain
Foremost on Hyun’s list is validating that the science is where it says it is. Many of the questions involved might not immediately seem to belong in the realm of ethics. But determining the practical effectiveness of these approaches is important to the ethical standing of the work because if they are useless, then any harm they cause becomes unacceptable. Are the organoids actually modeling what researchers claim? What are the most appropriate uses for such entities, once they’ve been created? Is it all right to base therapies on experiments done in different organoid systems, or to use organoids to diagnose certain conditions?
As research moves forward, it’ll be critical to determine the differences between brain organoids and actual brains, beyond the most obvious features, like size and connectivity. “We know that these [organoid] neurons are functioning. We know that these neurons are connected,” said Bruna Paulsen, a postdoctoral fellow in the laboratory of the prominent organoid researcher Paola Arlotta at Harvard. “But what does that actually mean?”
“There are even significant questions as to what types of basic electrical activity are there,” Chen said. For example, Muotri and several colleagues recently caused quite a stir, and some concern, when they reported last fall that they’d recorded electrical signals resembling the brain waves of a newborn baby in a cortical organoid. But some scientists took issue with that interpretation of the results: In a human brain, they argued, that electrical activity depends on anatomical structures and cell types that the organoid doesn’t have. In the absence of those features, the comparison might be misleading and could generate needless worry. (Muotri, for his part, took these debates to signal a need for further discussion, and for teaming up with experts on human development, consciousness, and other areas that stem cell researchers might not be familiar with.)
Also up for discussion are questions regarding how long to keep organoids alive, what trade-offs should be made between creating a complex organoid and keeping it manageably simple, and how to talk about the work publicly. (Lunshof and Greely noted that the popular description of brain organoids as “mini-brains,” while formerly embraced by many scientists, has raised unnecessary alarms.)
How researchers ask for consent in organoid experiments is under scrutiny now, too. Biologists and ethicists wrestle with whether study participants should be told that their skin cells might be transformed into pluripotent stem cells and used to create a brain organoid — and if so, how much information to reveal. In light of his recent work, Muotri revised his consent form to include a discussion of brain organoids and their ability to generate certain kinds of electrical signals. “I had for the first time a family that decided not to participate in the research because of that,” he said. “Not everybody is comfortable now in donating their cells if we are going in that direction.”
Questioning What Signals Consciousness
Even so, Muotri isn’t waiting for organoids to get more complex before pursuing ambitious research questions. The work he’s planning could help to map the biological and ethical terrain for future organoid research to explore. In particular, Muotri wants to monitor organoids’ electrical activity through new experiments designed specifically to look for traces of potential consciousness.
Until now, most of the proposed options for such studies have been analogues of tests administered to human patients. For example, some scientists have suggested taking certain algorithms developed to evaluate the brain waves of coma patients for signs of awareness and tweaking them to apply to the study of activity in organoids.
But there’s no consensus about whether those strategies would be fruitful, Chen said. “To be honest, there aren’t even that many ideas as to how to go about this” — particularly given the minimal amounts of activity occurring in the organoids. Moreover, since such measurements have never been validated for cells in a dish, or for animals, there’s no way to compare their efficacy.
Even so, according to Chen, the focus of work on organoids has gradually shifted from their cellular makeup, their gene expression and the protocols used to create them, toward understanding their electrical activity. “The only thing that we have available to us right now is just to be measuring the electrical activity of these organoids, and see what we see,” he said. “I think that over the next five years, we’re going to see a lot more papers that look specifically at characterizing that electrical activity.”
Muotri intends to be one of the major contributors to that research. One experiment he wants to perform would involve giving an anesthetic to an organoid and testing how it reacts: If certain electrical signals disappear from the recordings under anesthesia but reappear afterward, the implications could be interesting (though Muotri is careful to note that this is by no means strong evidence of consciousness, but rather just a first step). He also hopes to start feeding more sensory information to the organoids by creating neurons that carry pain receptors and testing whether those neurons react to stimulation. “We are not 100 percent sure that this is the right way to go,” he said. But it “might suggest something” or point them in a helpful direction.
He’s also currently creating a “map that will tell people where their organoids are on the road to consciousness,” he said. At the moment, it’s a set of yes-or-no questions that could help researchers keep track of how far they’ve gone in their work. They include: Does the organoid have a cortex? Does that cortex generate signals that resemble brain waves? Are there structures in addition to the cortex, and how are they connected? Can the organoid control an attached body part? Can it receive an input?
Finding a boundary between the ethical and the unacceptable on such a road map, or really any concrete answer or set of clear guidelines, “is much harder than it may seem,” Lunshof said. “The more involved you are in this research, the less certain you are about the answers you could give.” But she has hopes for the future of the collaborative efforts between scientists and ethicists. “I’m now at 15 years of putting this into practice,” she said. “And yes, it works.”
Correction: January 24, 2020
The photograph of Jeantine Lunshof was originally misattributed to the Wyss Institute at Harvard University; the actual photographer is Aram Boghosian.
This article was reprinted on Wired.com.



