The Shape-Shifting Squeeze Coolers
Introduction
After losing his sight to smallpox in 1759 at the age of 2, John Gough developed a heightened sense of touch. The budding naturalist soon learned to identify plants by feel, touching their hairs with his lower lip and their stamens and pistils with his tongue. So when as an adult he quickly stretched a piece of natural rubber and felt its sudden warmth on his lip — and its subsequent coolness as it relaxed — he gained what he considered the most direct and convincing proof of a curious phenomenon.
He described his observations in 1802, providing the first record, in English at least, of what’s now known as the elastocaloric effect. It’s part of a broader category of caloric effects, in which some external trigger — a force, pressure, a magnetic or electric field — induces a change in a material’s temperature.
But caloric effects have become more than a curiosity.
Over the past couple of decades, researchers have identified increasingly mighty caloric materials. The ultimate goal is to build environmentally friendly refrigerators and air conditioners — caloric cooling devices won’t leak harmful refrigerants, which can be thousands of times more potent than carbon dioxide as a greenhouse gas. But better cooling devices require better materials.
The more a material can change its temperature, the more efficient it can be. And in the last year, researchers have identified two unique types of materials that can change by an unprecedented amount. One responds to an applied force, the other to pressure. They are both capable of temperature changes — “delta T” for short — of a dramatic 30 degrees Celsius or more.
“Who would’ve thought you would get a material to give you a delta T of 30 by itself?” said Ichiro Takeuchi, a materials scientist at the University of Maryland, College Park, who wasn’t part of the new research. “That’s enormous.”
Hot Flash
Gough didn’t know it, but when he stretched his piece of rubber more than two centuries ago, he lined up the long molecules inside. The alignment reduced the disorder in the system — disorder measured by a quantity called entropy.
According to the second law of thermodynamics, the total entropy of a closed system must increase, or at least remain constant. If the entropy of the rubber’s molecular configuration decreases, then the entropy must increase elsewhere.
In a piece of rubber like Gough’s, the increase in entropy happens in the vibrational motion of the molecules. The molecules shake, and this boost in molecular movement manifests itself as heat — a seemingly hidden heat called latent heat. If the rubber is stretched quickly enough, the latent heat stays in the material and its temperature goes up.
Many materials have at least a slight elastocaloric effect, warming up a bit when squeezed or stretched. But to reach temperature changes large enough to be useful in a cooling system, the material would need a much larger corresponding change in entropy.
The best elastocaloric materials so far are shape memory alloys. They work because of a phase change, akin to liquid water freezing into ice. In one phase, the material can warp and stay warped. But if you crank up the heat, the alloy’s crystal structure transitions into a more rigid phase and reverts to whichever shape it had before (hence the name shape memory alloy).

A prototype elastocaloric heat pump at DTU Energy.
Jaka Tušek, originally published in
Nature Energy
The shift in the crystal structure between these two phases causes an entropy change. While entropy is related to a system’s disorder, it’s more precisely described as a measure of the number of configurations a system can have. The fewer the configurations, the less entropy there is. Think about a shelf of books: There’s only one way for the books to be alphabetized, but many ways for them to be un-alphabetized. Thus, a shelf of alphabetized books is more orderly and has less entropy.
In a shape memory alloy like nickel-titanium —which has shown one of the biggest elastocaloric effects — the crystal structure of the rigid phase is cubic. The pliable phase forms rhomboids, which are diamondlike elongated cubes.
These rhomboids have fewer possible configurations than the cubes. Consider that a square will remain unchanged if rotated through four possible angles: 90, 180, 270 or 360 degrees. A rhombus, on the other hand, will look the same only after two such rotations: 180 and 360 degrees.
Since the pliable phase has fewer possible configurations, it has less entropy. When an external force pushes on the alloy while it’s in its rigid phase, the metal transitions to its pliable, lower-entropy phase. As with Gough’s rubber, an entropy drop in the metal’s structure requires a rise in the entropy of its atomic vibrations, which heats the material.
In an air conditioner or refrigerator, you would then have to quickly remove this heat while keeping the alloy in its pliable, low-entropy phase. Once the force is removed, the alloy returns to its rigid, higher-entropy phase. But for that to happen, the atomic structure must acquire entropy from the alloy’s vibrating atoms. The atoms vibrate less, and because such vibrations are simply heat, the alloy’s temperature drops. The cold metal can then cool its surroundings.
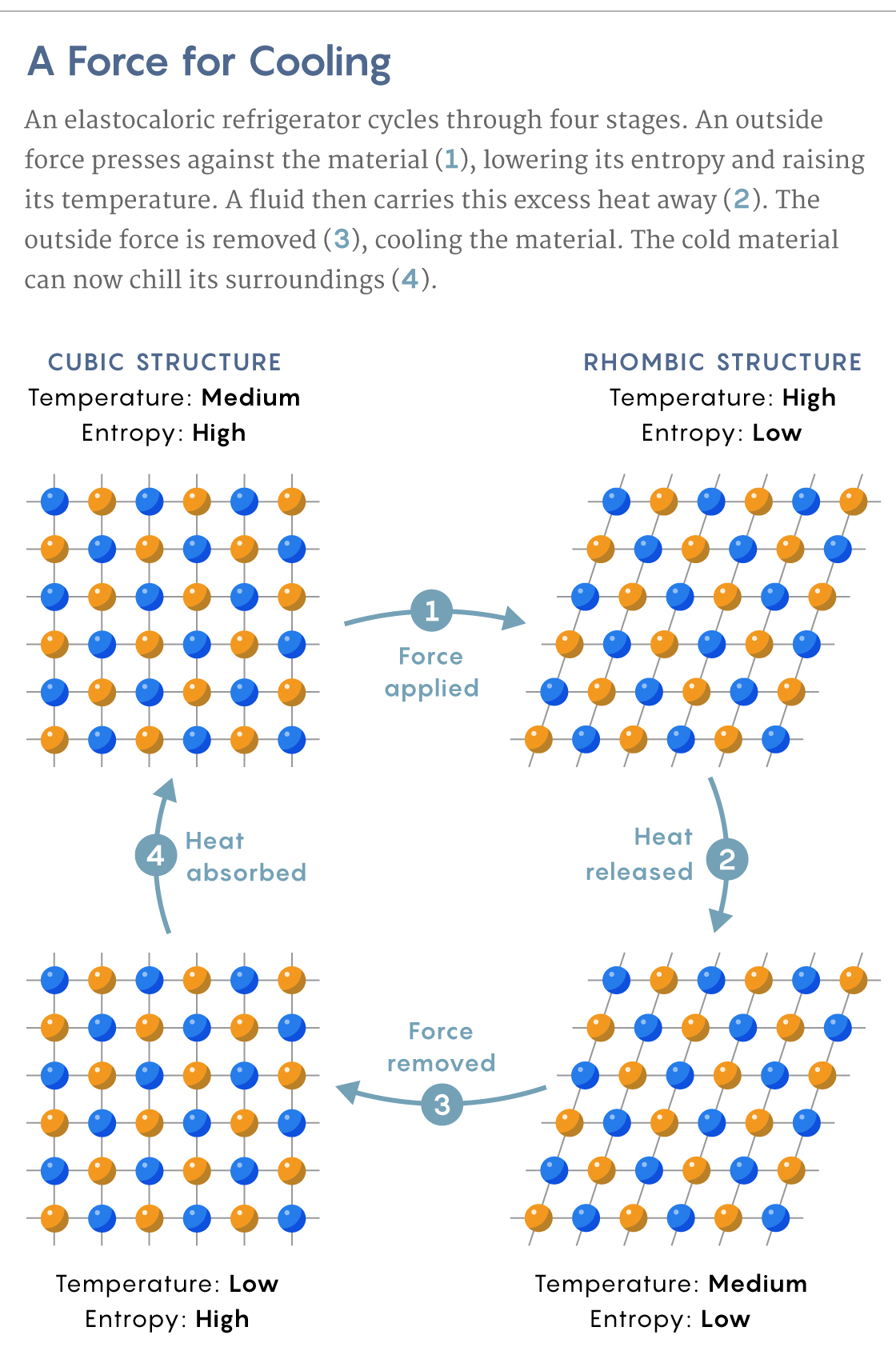
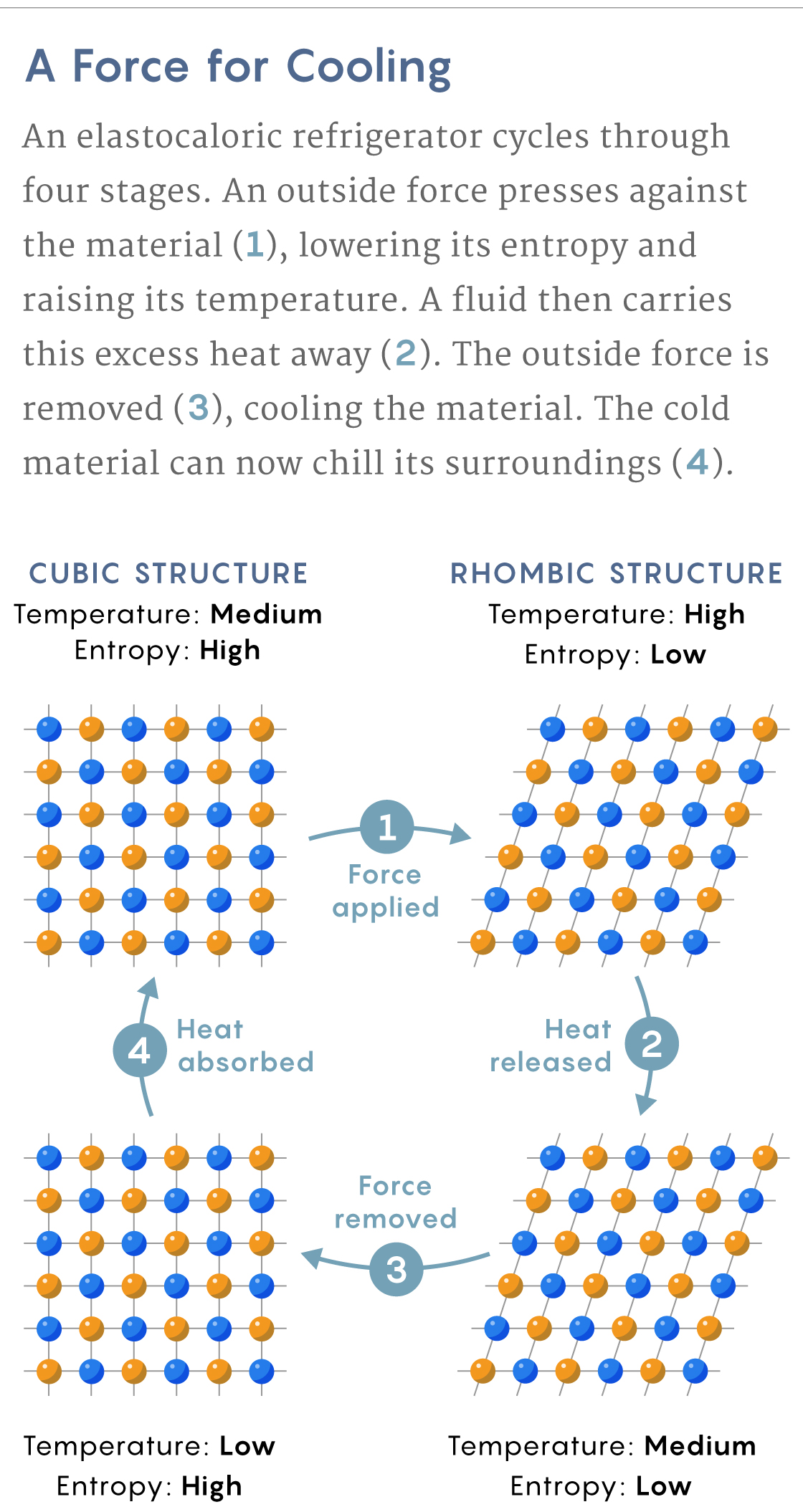
Samuel Velasco/Quanta Magazine; source: DOI: 10.1063/1.4913878
Progress on these materials has been steady. In 2012, Takeuchi and colleagues measured a temperature change of 17 degrees Celsius in nickel-titanium wires. Three years later, Jaka Tušek of the University of Ljubljana and others observed a change of 25 degrees in similar wires.
Then last year, a group based at the University of Science and Technology Beijing discovered a new shape memory alloy of nickel-manganese-titanium, which boasts what they called a “colossal” temperature change of 31.5 degrees. “So far, this material is the best,” said Antoni Planes, a solid-state physicist at the University of Barcelona who was part of the team.
What makes it so good? During a phase transition, nickel-manganese alloys shrink. Because volume corresponds to the number of possible atomic configurations of the material, a reduction in volume leads to a further reduction in entropy. “This extra contribution is what makes this material interesting,” Planes said.
Cool Under Pressure
Shape memory alloys have limitations, though. Notably, if you squeeze a piece of metal over and over, the material is going to fatigue.
Partly for this reason, researchers have also pursued “barocaloric” materials, which heat up when you apply pressure. It’s the same basic principle: Pressure induces a phase change, lowering entropy and heating the material.
One intriguing material is neopentylglycol, a type of plastic crystal. This material is soft and deformable, consisting of molecules loosely bound in a crystal structure.
Neopentylgylcol’s molecules are round and arranged in a three-dimensional lattice. They interact with one another only weakly and can swivel into roughly 60 different orientations. But apply enough pressure and the molecules become stuck. With fewer possible configurations, the material’s entropy drops.
A plastic crystal’s squishiness means that squeezing it reduces its volume, decreasing entropy even more. “Because they are, in a way, between solid and liquid, they can display larger changes in entropy when you apply pressure,” said Xavier Moya, a solid-state physicist at the University of Cambridge.
Last year, two teams achieved the largest barocaloric effects on record. Neither team directly measured a temperature change, but a European team that included Planes and Moya reported an entropy change of 500 joules per kilogram per kelvin — the largest ever for a solid, on a par with entropy changes in commercial fluid refrigerants. They calculated a corresponding temperature change of at least 40 degrees. Another team based at the Shenyang National Laboratory for Materials Science in China reported an entropy change of 389 J/kg/K.
But many practical challenges remain. While barocaloric materials are less susceptible to fatigue than elastocaloric materials, the new milestones required colossal pressures of thousands of atmospheres. Such pressures also require the material to be sealed. “It’s difficult to exchange heat between this material and the surroundings if you seal the whole system,” Tušek said.
Indeed, heat exchange isn’t straightforward, Moya said. But he’s working on some proprietary systems for a barocaloric refrigeration company he co-founded called Barocal, which is a finalist for the Global Cooling Prize, an international competition to find sustainable cooling technologies. Takeuchi, meanwhile, founded Maryland Energy and Sensor Technologies in 2009 to commercialize elastocaloric cooling. The commercial products are being developed with copper-based shape memory alloys, which are softer and don’t need as much force as nickel-titanium alloys.
By contrast, Planes and his longtime collaborator Lluís Mañosa are focusing on multicalorics, which respond to multiple stimuli, such as both force and a magnetic field. Multicaloric devices would likely be more complex, but multiple stimuli could drive even greater entropy and temperature changes with higher efficiency. “Prospects for the future are very good,” Planes said. “But for the moment we are at the beginning.”
This article was reprinted on Wired.com.



