Random Search Wired Into Animals May Help Them Hunt

A wide variety of predatory and foraging creatures may have evolved to follow a certain kind of random path — a Lévy walk — when no clues to the whereabouts of their food are available.
Natalie Dombois for Quanta Magazine
Introduction
It’s not surprising that the fruit fly larva in the laboratory of Jimena Berni crawls across its large plate of agar in search of food. “A Drosophila larva is either eating or not eating, and if it’s not eating, it wants to eat,” she said. The surprise is that this larva can search for food at all. Owing to a suite of genetic tricks performed by Berni, it has no functional brain. In fact, the systems that normally relay sensations of touch and feedback from its muscles have also been shut down.
Berni, an Argentinian neuroscientist whose investigations of fruit fly nervous systems recently earned her a group leader position at the University of Sussex, is learning what the tiny cluster of neurons that directly controls the larva’s muscles does when it’s allowed to run free, entirely without input from the brain or senses. How does the animal forage when it’s cut off from information about the outside world?
The answer is that it moves according to a very particular pattern of random movements, a finding that thrilled Berni and her collaborator David Sims, a professor of marine ecology at the Marine Biological Association in Plymouth, U.K. For in its prowl for food, this insensate maggot behaves exactly like an animal Sims has studied for more than 25 years — a shark.
In neuroscience, the usual schema for considering behavior has it that the brain receives inputs, combines them with stored information, then decides what to do next. This corresponds to our own intuitions and experiences, because we humans are almost always responding to what we sense and remember.
But for many creatures, useful information isn’t always available, and for them something else may also be going on. When searching their environment, sharks and a diverse array of other species, now including fruit fly larvae, sometimes default to the same pattern of movement, a specific type of random motion called a Lévy walk. This shared trait hints that evolution may have equipped nervous systems to spontaneously generate a foundational movement pattern, a solution that works better than any other strategy for a blind search.
The idea has provoked debate across the fields of ecology and animal behavior for more than two decades. The work of Berni and Sims now brings the phenomenon firmly into the realm of neuroscience and makes it hard to ignore the case for its importance.
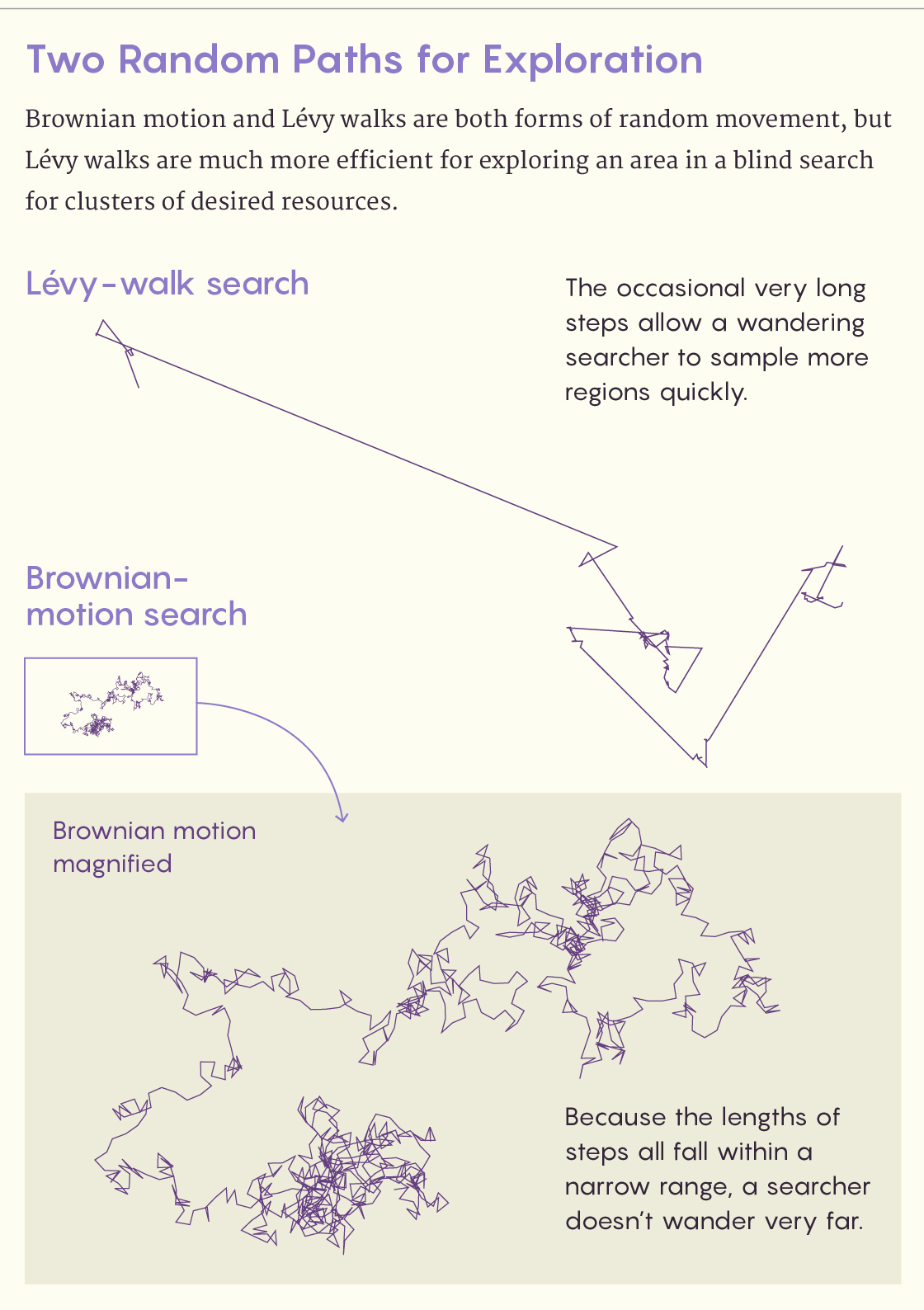
Lévy Walks on the Wild Side
When the Scottish botanist Robert Brown observed the chance jiggling of pollen grains in water in 1827, his name was permanently wedded to the most famous type of random movement. A body in Brownian motion moves in random directions for random distances over and over again. The distribution of the random distances is Gaussian, or normal: They fall symmetrically around a mean value — and, crucially, no step length deviates hugely from the mean. Brownian motion describes a great many physical phenomena, including diffusion.
In 1937, however, the French mathematician Paul Lévy described a second type of random movement. In this one, the distribution of random distances follows a power law and has a characteristically long righthand tail. This means that the lengths of most movements cluster around the average, but some movements go much, much farther. The fractal pioneer Benoit Mandelbrot, a former student of Lévy’s, named this distribution of step lengths a Lévy walk (or Lévy flight) in 1982.
5W Infographics for Quanta Magazine; source: Gandhi Viswanathan
Around this time, a few biologists began exploring the potential usefulness of this pattern to describe natural behaviors because a Lévy walk is very reminiscent of the path of an animal thoroughly exploring one space, then popping off to explore another. The idea did not achieve prominence until 1996, however, when Gandhi Viswanathan and colleagues proposed that albatross flights were a Lévy walk.
“My original motivation, as a Ph.D. student,” said Viswanathan, now a professor of physics at the Federal University of Rio Grande do Norte in Brazil, “was actually to better understand ‘free will.’” As a physicist, he wanted to look for statistical laws that described people’s seemingly indeterminate behavior, but he had no useful human data to examine. When the opportunity arose to analyze flight data from albatrosses — birds that fly freely over the open ocean in search of food — he gratefully took it.
Viswanathan was given a distribution of flight times derived from wet/dry sensors clipped to the birds’ legs. When a sensor was wet, it meant a bird was bobbing on the seas; when it was dry, the bird was in flight. The data revealed a Lévy distribution of flight times. Published in Nature in 1996, the finding piqued the interest of ecologists and led Viswanathan to explore its potential significance.
Working with a team of physicists that included Marcos da Luz of the Federal University of Parana and Ernesto Raposo at the Federal University of Pernambuco in Brazil, Viswanathan published a theoretical study in 1999 that showed the relevance of Lévy walks to foraging. When food is distributed in sparse patches that can regenerate quickly and an animal has no sensory guidance to where the food is, Lévy walks are the mathematically optimal search strategy for blindly discovering a meal.

Albatrosses fly over vast expanses of ocean in a search for fish. Sometimes when those searches seem particularly unguided, the birds seem to fly in Lévy walk patterns.
The paper evoked a level of controversy that still surprises Viswanathan. The mathematical ideas were embraced by some ecologists and physicists interested in animal behavior, but others flatly rejected them. “In retrospect,” he said, “I get the feeling that this way of thinking … was too shocking to some researchers.”
The field quickly acquired a reputation as fractious and torn by passionate arguments. The usefulness of Lévy walks was further supported by theoretical work, such as a study that showed they were the best blind search strategy when the target was rare and slower moving than the searcher — a scenario relevant to predators seeking prey, pollinators seeking flowers and mating creatures seeking partners. But empirical demonstrations that many species’ movements fit this theoretical framework were slow to come: Zoologists needed more complete and detailed observations of wild animals’ movements, and mathematicians needed more refined methods for analyzing such data.
One ecologist inspired to action by the Lévy walk idea was Sims. At the time he was trying to map the movements of fish as an aid to marine conservation. He thought, “I really want to test this in basking sharks ” — a species that had become endangered after many years of being hunted for their oil, meat and fins.
Basking sharks rove vast spans of the ocean for patches of plankton to graze — not unlike an albatross on the wing seeking schools of fish. If a Lévy walk was evident in a shark’s path, it might mean the animal was exploring rather than dining on plankton. And Sims thought that insight might help him better understand the sharks’ movements and work out how to protect the animals from fisheries.
The problem he faced was getting enough data. To detect a Lévy walk, researchers need to follow every move an animal makes for a considerable amount of time. Today, GPS systems can precisely track the movements of radio-enabled objects, such as our cell phones. But in the late 1990s, Sims was monitoring basking sharks by watching from a boat when they disturbed the surface, which meant he was seeing only a tiny fraction of the creatures’ movements. And the sharks he could see were almost certainly feeding amid plankton patches — not wandering on Lévy walks.
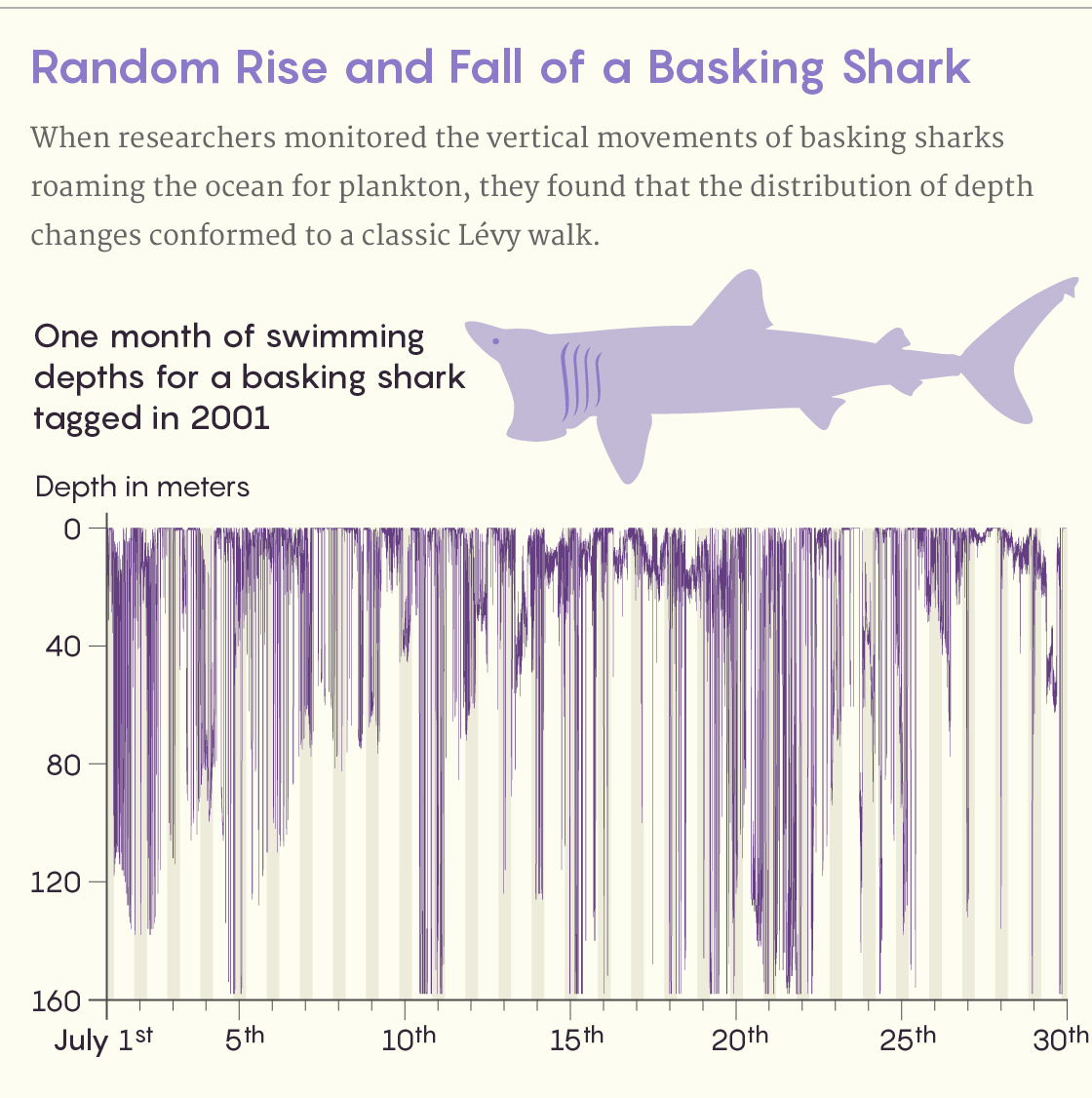
So Sims resorted to tagging the sharks. A pressure sensor on a tag attached to a shark’s fin recorded the animal’s swimming depth at one-second intervals as the shark moved up and down in the water column. At a programmed time, the tag separated from the shark, bobbed to the surface and relayed its data record to a satellite.
The pattern of these vertical movements was a classic truncated Lévy walk. (Truncated, that is, in a purely mathematical sense: In a perfect Lévy walk, there would be a nonzero probability of the shark occasionally taking a step equivalent to swimming to Mars.) Sims was so impressed, he looked at tracking data he had on blue sharks and ocean sunfish too, then wrote to colleagues and got comparable data describing leatherback turtles, penguins and tuna. All of them showed Lévy walks to greater or lesser extents.
Sims and his colleagues hypothesize that many animals evolved to use this search pattern when they could neither sense nor remember where food was. “Lévy movements arise when the animal really has no clue,” said Nick Humphries, a computational biologist and a postdoctoral researcher in Sims’ lab.
5W Infographics for Quanta Magazine; source: The Marine Biological Association
That description applies to sharks in the middle of the ocean. “The Atlantic Ocean is pretty much a desert,” Humphries said. “There are food patches, but they are sparse. The distance between the last food patch and the next is way beyond sensory range.” It’s also an ever-shifting environment where memory is useless. It’s exactly the type of environment where a Lévy walk-based search would most likely have evolved.
In a summary of their findings in Nature in 2008, Sims, Humphries and their colleagues showed that the movements of sharks and other marine predators varied depending on whether they were searching for food (when they did Lévy walks) or in the midst of a food patch (when Brownian randomness prevailed). Their work supported Sims’ original hunch that defining the type of movement might reveal whether a fish was consuming food or exploring for it.

A juvenile basking shark about three meters long feeds near the surface off the Cornish coast of the U.K.
2001 The Marine Biological Association
Ironically, their paper arrived only months after a study co-authored by Viswanathan, da Luz and Raposo showed a catastrophic error in Viswanathan’s original albatross study. The wet/dry sensors they had relied on weren’t dry only when the birds flew: They were also dry when the albatrosses sat on their nests. Some of the longest “flights” in the data set might have actually been protracted visits to the birds’ nests. The study also criticized certain analytical techniques and claimed that albatrosses don’t do Lévy walks after all, and that it was unclear whether many species did.
Sims acknowledges that the conflicting papers “put people on edge.” But his work used more up-to-date mathematical methods to demonstrate that a whole menagerie of marine predators take Lévy walks. And in retrospect, Sims’ paper was a turning point in the field. According to Raposo, it “called the attention of many scientists working in the area to the use of more powerful and modern statistical techniques of empirical data analysis of animal movement.” These improved methods quashed doubts over whether animals ever do Lévy walks. “The amount of data is overwhelming,” said da Luz.
Sims and Humphries even revisited albatrosses in a subsequent study in 2012. Working with more sophisticated GPS tracking devices, they found that albatrosses did not perform Lévy walks while searching for food above shallow water, where they might have more visual or olfactory information to guide them. But when they flew out over deeper water, the birds did in fact adopt Lévy searches.
Lévy walks are now seen as a movement pattern that a nervous system can produce in the absence of useful sensory or mnemonic information, when it is an animal’s most advantageous search strategy. Of course, many animals may never employ a Lévy walk: If a polar bear can smell a seal, or a cheetah can see a gazelle, the animals are unlikely to engage in a random search strategy. “We expect the adaptation for Lévy walks to have appeared only where they confer practical advantages,” Viswanathan said.
To Berni, Sims and Humphries, the situation is less definitive: Although Lévy walks have not been documented in many species yet, the scientists think too few species have been properly observed to justify statements about how common they might be.
Intrinsic or Extrinsic Guidance
The ongoing collaboration between Sims and Berni with the genetically modified Drosophila larvae emerged from the next debate that engulfed the field, which concerned whether Lévy walks are adaptive, intrinsic traits. Are Lévy walks a behavioral strategy that evolved because it made animals better at finding whatever they needed? Or do Lévy walks just emerge when an animal goes searching because the world’s resources and information follow that distribution?
Certain monkey species, for example, do Lévy walks when exploring, but they seem to be using their senses and memories to travel between food resources distributed that way. The monkeys are therefore neither generating the Lévy walks nor using them for blind searches.
Andy Reynolds, a theoretical physicist at Rothamsted Research in the U.K., notes that Lévy distributions have been found in assorted biological and physical phenomena, including the journeys of immune T cells in the brain, the operations of intracellular molecules, and the airborne dispersal of seeds and spores. “The picture is almost certainly a pluralistic one, as no one mechanism can account for all the observations,” Reynolds said.
For example, Reynolds believes that the albatrosses’ Lévy walks “appear to be an accidental byproduct of olfactory-cued navigation — of the birds following their noses.” He suspects that the movements of airborne scents are the true source of the Lévy distribution. (Sims says a number of observations are incompatible with this hypothesis, not least the distinction between albatrosses’ different searches above shallow and deep water.) The way to settle the matter, Reynolds says, is to temporarily disrupt some birds’ sense of smell to see if the Lévy patterns disappear. In short, experimental science needs to overtake the observational science that has dominated the field.
This is exactly what Sims and Berni set out to do in the fruit fly. By shutting down its brain and any sensory channels, they could determine whether a mechanism in the larvae generated Lévy walks in the absence of information about the world.
Sims says that when he started thinking about how to tackle the mechanistic underpinnings of Lévy walks, he was lucky to read Berni’s 2012 paper that described how genetically inactivating the brains of Drosophila larvae didn’t stop them from exploring their environment: A small cluster of neurons in the nerve cord running down the larva’s back could still usefully control the animal’s muscles without guidance from the brain. “I realized her methods were exactly what we needed to begin testing this idea of intrinsic versus extrinsic,” Sims said.
As it happened, Berni had also become interested in foraging and Lévy walks and was keen to collaborate with someone in the field. In 2013, Sims travelled to the University of Cambridge, where Berni then worked. “We met for a really good lunch,” he recalled. They hit it off and agreed to work together.
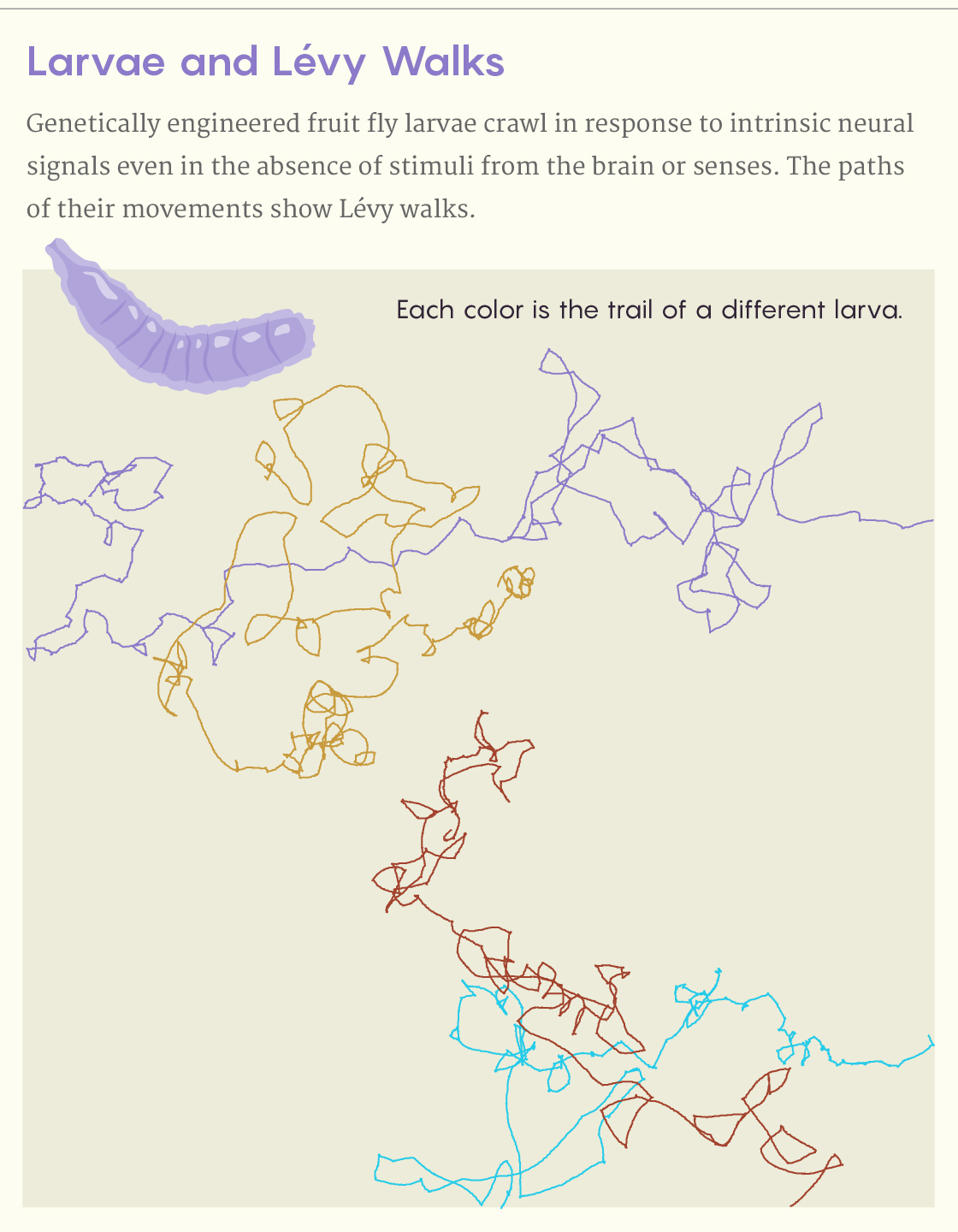
Berni bred new fruit flies and built much bigger agar arenas to run much longer tracking experiments. Her early studies of larval movement were akin to Sims’ method of watching a shark from a boat; now she needed to follow the animals as they roamed over much greater distances.
When Sims and Humphries analyzed Berni’s results in 2015, they found that normal larvae placed on an expanse of agar containing no food occasionally moved in a way that approximated a Lévy walk. But Lévy walks were still detectable even when the animals’ brains and senses were switched off. They concluded that the neural center intrinsically generates the pattern. “Wow — we were blown away!” Sims said.
5W Infographics for Quanta Magazine; source: Jimena Berni
The resultant paper was published last November in eLife, where it was warmly received by reviewers. One of these was Adam Calhoun, a neuroscientist at Princeton University, who roundly praises the study. He says that the biology of foraging has emerged as a serious concern for neuroscientists only over the last decade. His take was that “there’s no sensory information and they’re generating a power law — that’s really cool. They generate a Lévy walk as a base state.”
Viswanathan describes the work as amazing. “The question is no longer if but when animals exploit Lévy walks. Also, the intrinsic generation can only have an evolutionary origin.”
Reynolds, though, isn’t yet certain. “The results provide the strongest evidence so far for the intrinsic generation of Lévy-like movement patterns,” he said, but he warned that “it remains to be seen whether the Lévy walk behavior is an evolved trait of this animal.”
Berni is now trying to demonstrate exactly that. “We’re working on what circuits are generating these alternations between the straight runs and the turns,” she said. To do so, she isolates larval nervous systems and places them under a microscope. Because their genetically modified neurons glow when active, she can watch the flickering patterns of neurons firing. “They are producing rhythms spontaneously without any sensory inputs coming to them,” she said, “and we can see these rhythms of motor neuron behavior that are equivalent to what the behavioral output is.”
Berni and her group are also investigating what generates the Lévy distribution of walk lengths. In collaboration with the computational neuroscientist Julijana Gjorgjieva and the mathematician Marina Wosniack at the Max Planck Institute for Brain Research in Frankfurt, they are building computer models of the neural network. They are hoping such a model will enable them to explore how external sensory signals or instructions from the brain change the intrinsic activity pattern governing a larva’s baseline behavior.
Berni’s lab is also running further behavioral experiments in which larvae traverse agar plates dotted with different arrangements of food. When there are patches of food, interesting behavioral transitions happen: Like sharks moving within or beyond clouds of oceanic plankton, the larvae move in and out of Lévy walks.
Walking the Science Forward
There are other lab animals that neuroscientists could monitor for Lévy walk behavior. Evidence hints that nematodes and certain mud snails use this search strategy. Sims and Berni wonder how these ideas might be explored in vertebrates like mice and zebra fish. Humphries ponders whether simply asking some undergraduates to look for something hidden on a beach might reveal the characteristic pattern.
Comparisons of Lévy walk behavior between species could prove fascinating. Berni wonders if different animals arrive at the behavior in different ways, “or is there some principle of neural firing or neural activity or neuronal connectivity that is actually generating the same type of characteristics in different animals?” The idea that the mathematics for an optimal blind search strategy might be embedded in the very structure of a nervous system feels like a question that needs to be answered.
For now, though, Sims and Humphries are mostly turning away from the basic science of the phenomenon to focus again on conservation work: They will use Lévy walks along with other tools to classify shark behavior. But Berni hopes other neuroscientists will be inspired to look for generators of these movement patterns beneath everything else brains do.
“The field discovers something they can really work on,” she said of the neuroscientists. “Then they stay and explore this for a while, before they move on and look for something else.” She added with a smile, “In a little bit of a Lévy way.”



