Viruses Find a New Way to Hijack Cells

Viruses travel light. Most carry just their genetic material and a few tools to break into the cells of their hosts — after that, they hijack the host’s own machinery to manufacture thousands of copies of themselves. In recent decades, biologists have gained a clearer picture of just how this heist is pulled off. Many viruses, it turns out, suppress the messages that cells send to control their daily operations. This information interference shuts down some cellular functions that the attacking virus doesn’t need, and boosts others.
But some viruses do something subtler and more complex, as biologists at the University of California, San Diego, reported recently. The scientists looked at cells infected with cytomegalovirus, a common cause of birth defects. CMV infection doesn’t block cellular messages; instead, it changes their content, the team found. In a new paper in Nature Structural and Molecular Biology, they detail thousands of changes in these host communications, which may be the virus whispering sedition to remodel the cell.
To understand the importance of what is going on here, first consider how the cell functions normally. At the heart of the whole affair is the DNA — a sort of code book of instructions for how to make everything in the cell — which is kept under lock and key in the nucleus. When the cell needs to manufacture a protein, the relevant portion of the DNA is transcribed. That transcript, called a messenger RNA, leaves the nucleus and heads to the machinery that will use it as a template to make the new protein.
But along the way, the RNA can be edited in a number of ways. It might acquire tags that give extra instructions on how to handle it; it might have parts snipped out; it might gain or lose end pieces that make it easier or harder to use. In the normal way of things, the cell employs all of these strategies to control its own functions. In fact, the altering of these RNA messages, so different versions of proteins are made at different times, is key to the process of development. The same gene can be used to make one version of a protein in a human fetus and a different version in an adult.
But as with so many things, once those tools are under the control of an enemy, it’s a different story. A virus that can edit a host’s RNA messages would be able to create versions of proteins that favor a virus’s goals, without ever having to break into the nucleus. And because CMV is known to be somewhat peculiar — it is one of those viruses that don’t suppress hosts’ messages the way many others do — the UCSD group, led by Gene Yeo, a molecular biologist, and Deborah Spector, a virologist, decided to see if it was doing something else to the host’s RNA.
First, the team infected human cells with CMV. Then they extracted the RNA made by the cells at different time points over the course of the infection. The extractions revealed the sum of the chatter between the nucleus and the protein-making machinery. They looked to see how many edits — extra tags, altered end pieces and so on — there were, as compared with healthy, control cells. And while the early stages of infection didn’t show dramatic differences, the late stages, when the infected cells were gearing up to burst and release tons of new viruses, were a different story.
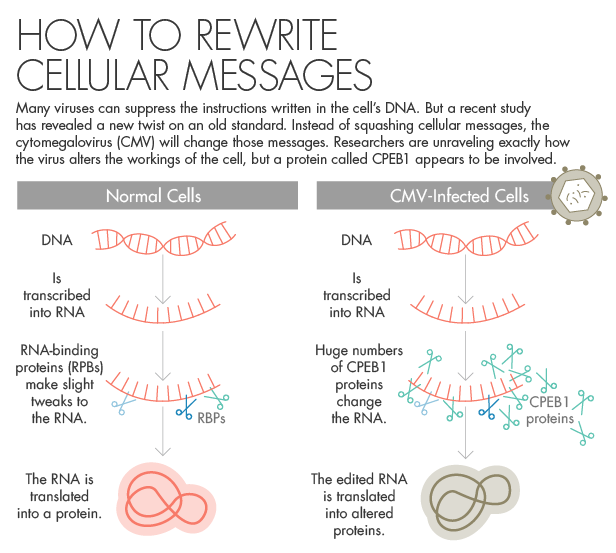
The team searched for the triggers of the changes by identifying proteins known to alter RNA. The researchers found that one in particular, called CPEB1, was also present at very high levels in infected cells. CPEB1 is made at low levels by human cells normally, and its job is making tweaks to the cell’s RNA. But when CMV arrives, CPEB1’s numbers go way, way up. The CPEB1 levels are remarkable, said Joan Steitz, a biochemist at Yale University who has been a pioneer in the study of RNA. She said that while she’s ordinarily skeptical of claims that moderate changes in protein levels are a mark of something important, this study shows that infected cells have 30 times the normal amount of CPEB1. “I mean, this is huge,” she emphasized. “So something is going on here.”
To see what this excess CPEB1 was doing, the team added some to healthy cells. These cells went on to develop many alterations seen in their infected siblings. The researchers then took infected cells and artificially reduced their CPEB1 levels. The number of viruses in the cells plummeted, and the cells also looked healthier; they didn’t have the swollen, bulbous look of late-stage infection. Similar experiments with a few related proteins did not produce the same effects. “The virus is handicapped unless it has CPEB1” specifically, Yeo said, “in the host cell at the level that it needs.”
So what, exactly, is the link between the virus and this protein? It’s still unclear. CPEB1 is known to modify RNA. But is CMV really using CPEB1 to edit RNA to manipulate the cell into making special versions of host proteins that directly benefit the virus — for instance, immune proteins that will leave it alone, or packaging proteins that are just its size? Or does the virus drive up levels of CPEB1 because it needs the protein as part of its own self-maintenance process, and all these changes to the host are a byproduct?
At first glance, it appears that many of the proteins altered by CMV infection involve cellular packaging, which fits with the idea that CPEB1 might be remaking the cell for the virus’s benefit. But to find out if that’s really the case, researchers will need to look closer at what exactly the edits are and how they change what the host cell does.
There is a wealth of possible follow-up studies, said Britt Glaunsinger, a professor at the University of California, Berkeley, who studies related viruses. “If they’re able to follow up, or if other people — even better — are able to follow up mechanistically and get some of the finer details,” Glaunsinger said, “then that will tell us.”
But looking to the cell’s messages and the proteins that manipulate them is a promising strategy, said Steitz, who has studied how viruses use such proteins. “There are thousands of RNA-binding proteins,” she said. “They are really important to the life of the messenger RNA.” And how that RNA is interpreted can make all the difference between a healthy cell and a diseased one. From the perspective of a virus, these messages traveling through the cell, ripe for easy editing, and the myriad proteins capable of changing them, are likely a prime way to achieve their ends. In fact, the UCSD team also found reworked host-cell messages that had been infected with herpes simplex virus-2, which is related to CMV. CPEB1 didn’t seem involved in those cases, but many host proteins could be called into action to make these changes.
“It’s just another example of a virus going in and targeting a particular protein that then has a profound effect on both its own viral RNAs and host RNAs and is part of the whole viral devastation plan,” Steitz said. “What’s so fascinating is that different viruses do it in slightly different ways.”
With all the RNA-binding proteins potentially at a virus’s disposal, our enemies may be making profound revisions to our cells’ messages — but only time, and more work, will tell for sure. Steitz is curious whether high levels of CPEB1 will crop up in any of the herpes viruses she is studying. It’s another lead to follow in the ever-shifting case of viral takeover.
This article was reprinted on TheAtlantic.com.



