The Incredible Shrinking Sex Chromosome

Jennifer Marshall Graves, an Australian biologist, is probably best known for a dire prediction: the human Y chromosome, which makes males male, could disappear in the next 5 million years. In the last 190 million years, the number of genes on the Y has plummeted from more than 1,000 to roughly 50, a loss of more than 95 percent. The X chromosome, in contrast, stands strong at roughly 1,000 genes.
Media reaction has been predictable, with overheated headlines proclaiming “Men on road to extinction.” But Graves, a biologist at La Trobe University in Melbourne, notes that 5 million years is a long time for a species such as ours, which is only 200,000 years old. More importantly, the loss of the Y chromosome might not spell disaster. If the Y does disappear, we may well develop a new mechanism for making men.
Scientists are discovering that the mechanisms that organisms use to determine sex are in a remarkable state of flux. When one system is destroyed, evolution seems to easily come up with a new one. Birds, fish and snakes have found myriad ways of making males and females. Sex chromosomes are frequently lost or swapped. Even closely related species can determine sex in quite different ways, suggesting that the system is highly flexible and evolving rapidly.
Recent studies of these different animals are helping scientists understand what happens when sex chromosomes shrink and disappear. “Sex determination is probably the most fundamental decision you make; it has huge implications for morphology, behavior, life history,” said Katie Peichel, a biologist at the Fred Hutchinson Cancer Research Center in Seattle. “Given that this is a super-fundamental process, how come it seems like every organism came up with its own mechanism for doing it?”
Weird Sex
A sex chromosome is simply a chromosome that determines whether an organism will be male or female. Scientists can often narrow this function down to one particular gene on the chromosome. That sex-determining gene will activate a sex-specific developmental cascade, turning on the biological factories that produce, for instance, the testes in males.

Humans and most other mammals use the familiar XY system. A gene on the Y chromosome triggers the development of sperm, determining male sex. Birds use a ZW system, where the pairing pattern is reversed: Males are ZZ and females are ZW. (The letters indicate the mode of inheritance; Y chromosomes are inherited strictly from father to son, the W from mother to daughter.)
All mammals use the same gene to control sex; all birds use another. And for many years, scientists underestimated the variety of other systems across the animal kingdom. Over the last decade, however, researchers have been able to read genomes from a much broader variety of animals. Graves and others have found sex-determining systems that are surprisingly diverse. “We had been hamstrung because we could only look at humans and mice,” Graves said. “But now we can look at bearded dragons and the half-smooth tongue sole, and there’s lots of variation out there.”
Lizards, amphibians and fish, in particular, frequently change the systems used to control sex. One species of tropical frogs has three different sex chromosomes: Y, W and Z. Males can be YZ, YW or ZZ, and females can be ZW or WW. How these strange systems function is poorly understood, said Doris Bachtrog, a biologist at the University of California, Berkeley. “We know little beyond the model organisms.”
The bearded dragon, a lizard named for the spiky scales circling its head, is perhaps the most striking example of sexual fluidity. This creature typically uses a genetic system to determine sex — ZZs develop as males and ZWs as female. But in 2007, Graves and collaborators showed that they could convert the lizards’ genetically controlled system to a temperature-driven one. Lizard eggs raised at higher temperatures developed into females, regardless of their genetic identity.
Temperature-controlled sex determination wasn’t itself a surprise — many reptiles, such as crocodiles, follow this method. But before Graves’ study, scientists thought that individual species used one mechanism or the other. The switch with bearded dragons revealed an unexpected level of interchangeability.
In a study published in Nature last summer, researchers pushed the system even further. They found dragons in the wild that were female even though they were genetically male (ZZ). They then mated these females with typical ZZ males. These odd couples produced fertile ZZ offspring whose sex relied solely on temperature. In effect, researchers eliminated the W chromosome in a single generation.
The human Y’s demise will take longer, but Graves believes that its fate was likely sealed at its origin. After it acquired the first sex-determining gene, other sex-specific genes — those that are helpful to males but not to females — began clustering around the new sex-determination site. Pieces of the chromosome flipped around, which eventually blocked the Y from pairing with its mate, the X. That in turn prevented a form of genetic housecleaning known as recombination, which helps rid chromosomes of mistakes. Unable to repair itself, the Y began its journey of decay. “Sex chromosomes are sort of self-destructing,” Graves said. “Adding a sex-determining gene to any chromosome puts it in great danger.”
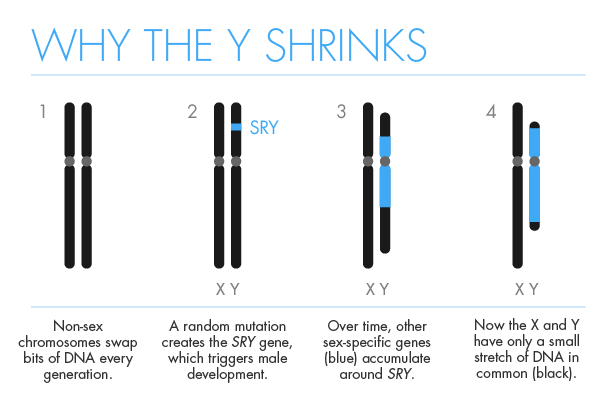
Graves notes that a number of rodents seem to be experimenting with new sex-determination systems. Several no longer possess an active version of SRY, the gene that triggers male development in most mammals. Two mice populations living on islands in Japan have lost their Y chromosomes altogether.
In all of these cases, individual populations have come up with new ways of making males, although researchers aren’t sure how they do it. Probably another gene on another chromosome has assumed the responsibility. (Eventually, this new sex chromosome will likely degrade just like our Y, Graves said.) What’s more, these changes may drive development of new species, which seems to be happening in stickleback fish.
Dancing Fish
Three-spine sticklebacks have a strange mating ritual. First, male sticklebacks build a nest and perform a dance to attract a mate. Then the males, whose backs are dotted with spines, swim beneath the females and prick them. Thus entranced, a female will lay eggs in her beau’s nest. He fertilizes them and chases the female away, and then provides the eggs with parental care.
Sticklebacks are a favorite among evolutionary biologists because they have evolved enormous diversity in both appearance and behavior on a relatively short evolutionary timescale. This diversity extends to their sex chromosomes. In some species, the male has two different chromosomes, as with humans. In other species, the female has them.
Two closely related species of sticklebacks in Japan have proved particularly interesting. The groups diverged about two million years ago, when some fish were trapped in the Sea of Japan by an icy barrier. The two species can be found breeding in the same locale — waters around the island of Hokkaido — but not with each other.
Both populations perform the pricking part of the mating dance, but with some significant differences. Males from the Pacific gently prick their would-be mates, while males from the Sea of Japan give them a great shove. “As soon as the male does the aggressive pricking behavior, the Pacific female says forget it, I’m out of here,” Peichel said. (The reverse pair — Pacific males and females from the Sea of Japan — will mate in the lab, but their male offspring are sterile.)
In addition, fish from the Sea of Japan have a chromosomal oddity. The Y chromosome is fused to the paternal copy of chromosome 9. The maternal copy of chromosome 9 becomes a new sex chromosome, dubbed the neo-X. And on this neo-X lie the genes that drive the fish’s aggressive behavior.
The findings link a new sex chromosome with a mating barrier, and ultimately a new species. But which came first? Did the chromosome fusion make it impossible for the two groups to mate, eventually leading to differences in their mating dance? Or did the new mating behavior precede the chromosomal change? No one knows. But recent data show that genetic differences in the fish are concentrated on the sex chromosomes. According to Peichel, that strongly suggests that sex-chromosome evolution leads to new species. “There are really no cases in which we know what caused speciation because it’s really hard to go back in time to figure it out,” she said. “But it is one of the rare cases where there is a direct link between chromosome rearrangement and speciation mechanism.”
Bad Design
Lizards, fish and rodents seem to survive major changes to their sex chromosomes. But what about humans? Are we in danger of losing the Y? That’s a matter of debate. For Graves, the answer is yes. Based on the number of genes on the Y chromosome and the rate of genes lost per million years, she estimates it will disappear in 4.6 million years.
Other researchers have challenged Graves’ dire predictions for the Y. A study published in Nature in 2012 found very little change over the last 25 million years. Since we diverged from old-world monkeys, the Y chromosome has lost just one gene. (Graves’ response is that sex-chromosome changes occur in fits and starts, so it’s impossible to predict whether the current pattern of stability will last.)
For many scientists studying sex chromosomes, the long-term status of the Y chromosome isn’t the most interesting issue. They want to understand more fundamental questions, such as why sex chromosomes exist at all. In puffer fish, for example, sex is determined by a single letter of DNA. If such a simple system works, “why have we progressed to the massive differences between the human X and Y?” said Judith Mank, an evolutionary biologist at University College London. Moreover, scientists have found animals whose sex chromosomes seem to resist decay, including some frog species with ancient sex chromosomes that have undergone little change over the millennia.
Mank, Peichel, Bachtrog and others have begun to assemble a database of sex-chromosome information, dubbed the Tree of Sex, which they hope will answer some of these big questions. “By mapping out sex determination across the tree of life,” Mank said, “we hope to understand how sex determination evolves, and to try to test theories about what sort of selection pressures might be driving the change.”