Bacterial Complexity Revises Ideas About ‘Which Came First?’

Bacteria look simple, but their internal structures may be surprisingly compartmentalized.
Koma Zhang for Quanta Magazine
Introduction
Open a basic biology textbook published decades ago or one published a few months ago, and both will define the two major categories of cells in the same way: Eukaryotes have membrane-bound compartments called organelles, including a nucleus where they store their genetic information, while prokaryotes do not. The distinction is even embedded in their names: In Greek, the word “eukaryote” means “true kernel” (a reference to the nucleus), while “prokaryote” means “before kernel.”
As the evolutionary story is usually told, first came the prokaryotes: the archaea and bacteria, which are often envisioned as simple bags of enzymes without an intricate structure. Then, more than 1.5 billion years ago, eukaryotes evolved, marking the advent of unprecedented cellular complexity and permanently transforming life on Earth, allowing for the rise of animals, plants, fungi and protists. The eukaryotes represented a substantial departure from their predecessors, and the transition from an all-prokaryote world to one that contained eukaryotes is often described as abrupt and explosive.
But this version of events ignores the fact that, for the past few decades, researchers have been quietly uncovering many complex structures within prokaryotes, including membrane-bound organelles. In contrast to eukaryotes, which all have a suite of organelles in common, different groups of prokaryotes showcase their own specialized compartments. One kind of bacterial organelle, discovered in 1979, is essentially a little magnet wrapped in a lipid package; another hosts a series of reactions crucial for energy metabolism; still others serve as small storage units for nutrients.
5W Infographics for Quanta Magazine
And that list is only growing as scientists discover more and more compartments within supposedly simple bacterial cells. “Bacteria are a lot more complex, in other words, and may have a lot more similarities in their biology to eukaryotes than people have assumed in the past,” said John Fuerst, a microbiologist at the University of Queensland in Australia. The very existence of organelles in these bacteria, coupled with intriguing parallels to the more familiar ones that characterize eukaryotes, has prompted scientists to revise how they think about the evolution of cellular complexity — all while offering new ways to probe the basic principles that underlie it.
A New Catalog
“Historically, people have known about compartments in bacterial cells that carry out specific functions for a long, long time, going back to the 1800s,” said Arash Komeili, a microbiologist at the University of California, Berkeley. Yet, while eukaryotic organelles have been studied in great detail for many decades, it has only recently become possible to do so in prokaryotes. Bacteria are tiny: orders of magnitude smaller than typical eukaryotic cells, and sometimes even smaller than eukaryotes’ organelles. That made it extremely difficult to isolate and analyze bacterial compartments to get a sense of what they were — and what they were doing. (Archaea, which were only recognized as a distinct prokaryotic kingdom in the 1970s, have received even less scrutiny than bacteria.) Better imaging techniques eventually started to make such research easier.
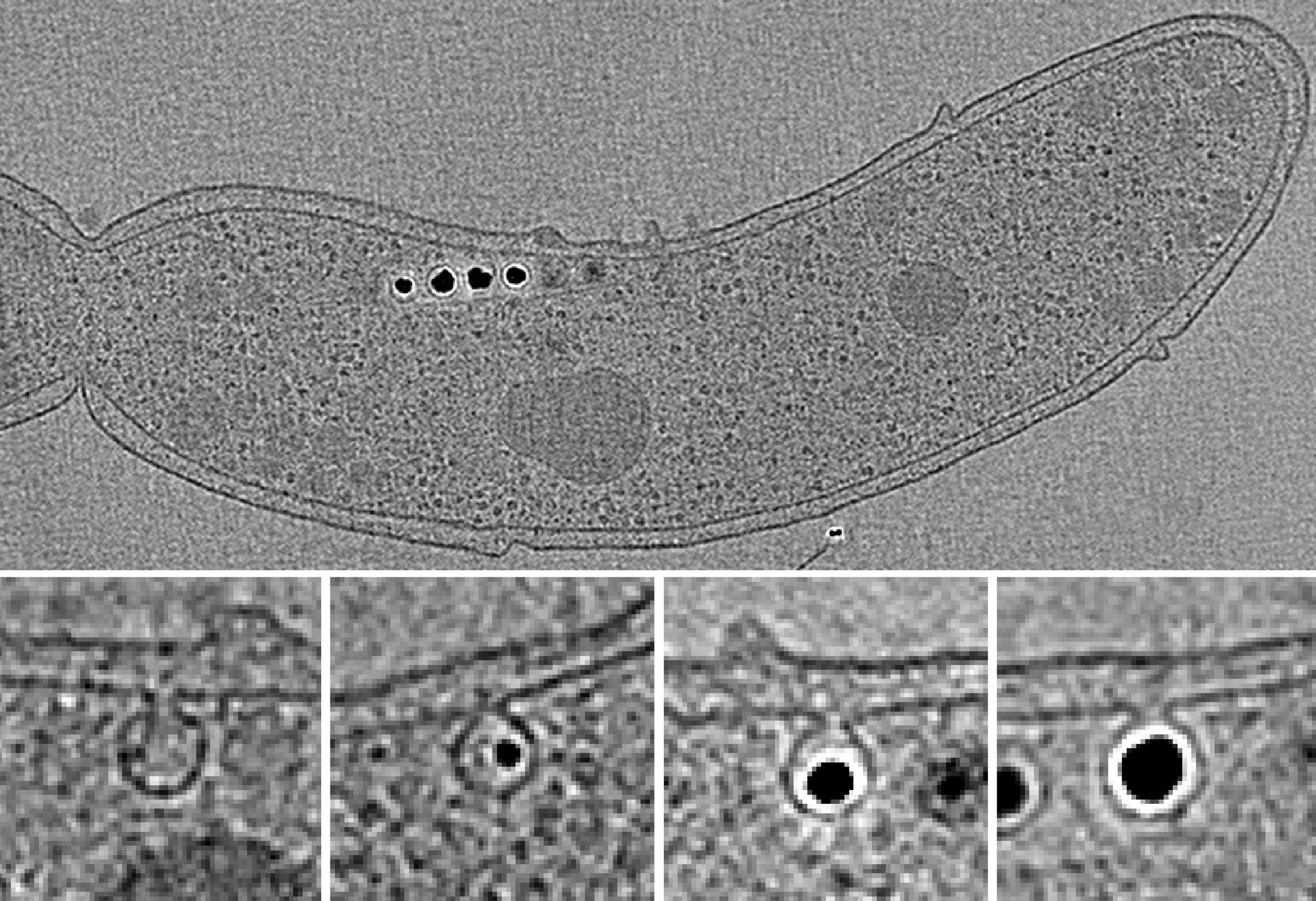
An electron micrograph of the bacterium Magnetospirillum magneticum (top) reveals the chain of magnetosomes it uses to navigate. A lipid membrane surrounds each magnetic particle (closeups at bottom). These structures, which are among the best-studied prokaryotic organelles, allow the bacterium to navigate through its aquatic environment.
Courtesy of Grant Jensen
Among the best studied of the bacterial organelles are the magnetosomes, round structures that build magnetic particles within their lipid bilayer membranes. The organelles allow aquatic “magnetotactic” bacteria to navigate vertically along the Earth’s magnetic fields toward the low-oxygen depths in which they thrive. Komeili and his colleagues have been identifying the genes and proteins involved in how magnetosomes are built, maintained and later divided among cellular offspring.
“On a superficial level,” Komeili said, “a lot of those activities, and even the way they look, are reminiscent of the ways that eukaryotic cells build organelles.” At the very least, their function parallels the ability of some animals, including salmon and homing pigeons, to detect magnetic fields — and in a paper published in April, researchers reported that one species of protist achieved this through a symbiotic relationship with magnetotactic bacteria.
But magnetosomes are not alone. Scientists have stumbled on a plethora of other bizarre bacterial compartments, often while searching for something else. Although many of these might not be considered organelles by the strictest definitions — organelles have to be lipid-bound structures completely separated from the cell membrane — some of them do fit the bill.
Tantalizing examples appear in a group of oval-shaped aquatic bacteria known as planctomycetes. Some species of planctomycetes contain a membrane-bound organelle called an anammoxosome, which sequesters a chemical reaction that produces nitrogen along with toxic intermediaries. Anammoxosomes act like energy factories for the bacteria, much as mitochondria do in eukaryotes, though anammoxosomes do not seem to be remnants of symbionts as mitochondria are.
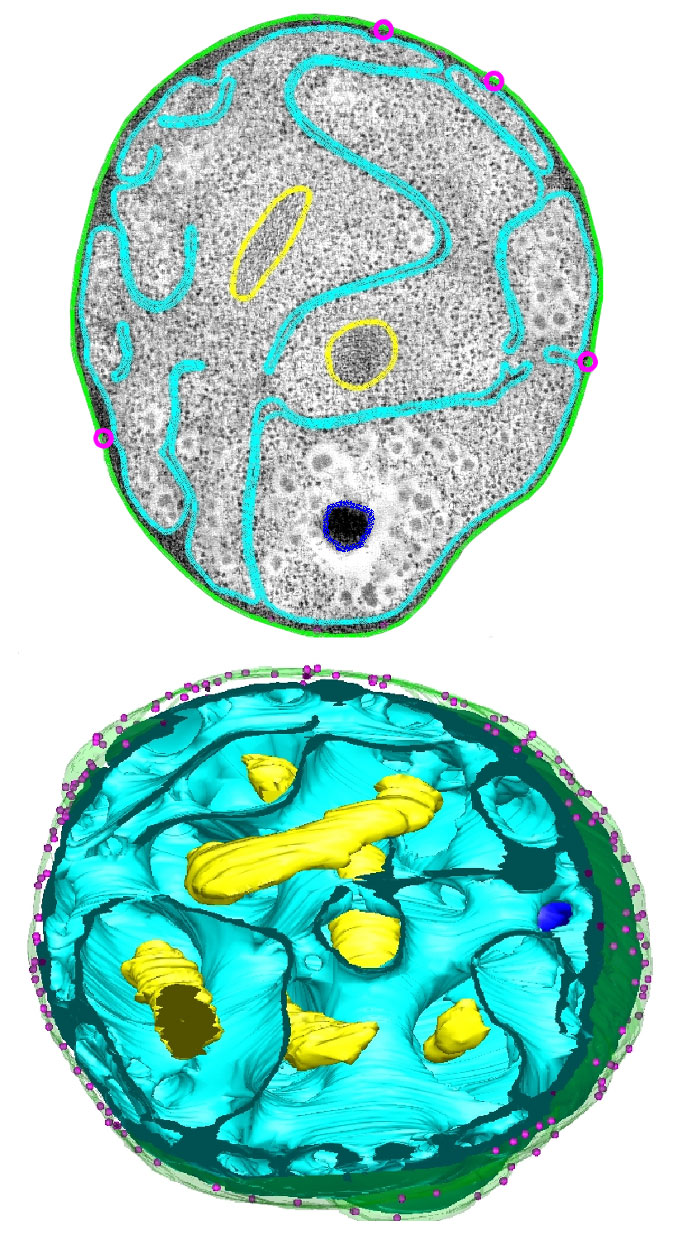
An electron micrograph (top) and three-dimensional reconstruction (bottom) of the bacterium Gemmata obscuriglobus show deep pouches in its cell membrane (blue) that seem to sequester its genetic material (yellow), inviting comparisons to the eukaryotic nucleus. This species and other planctomycete bacteria have the most complex internal membrane system yet observed in prokaryotes.
Rachel Santarella-Mellwig, Sabine Pruggnaller, Norbert Roos, Iain W. Mattaj, Damien P. Devos
Another kind of planctomycete has been a source of controversy for years. A couple of decades ago, two-dimensional imaging by Fuerst and others seemed to indicate that the DNA of the bacterium Gemmata obscuriglobus was surrounded by a membrane, instantly raising comparisons to the eukaryotic nucleus. Those results have been called into question — imaging seems to indicate that the compartment isn’t entirely closed, meaning it does not satisfy the definition of an organelle — but experts remain excited about these bacteria. They have the most complex internal membrane system seen in prokaryotes to date, and they contain proteins that structurally resemble those that shape and maintain eukaryotic membranes. They also seem capable of processes that were thought to be unique to eukaryotes, such as digesting nutrients inside their cells and synthesizing molecules called sterols.
“The problem is, we basically don’t know anything about [this membrane system],” said Damien Devos, a microbiologist at the Andalusian Center for Developmental Biology in Spain who studies planctomycetes. “We still have a very, very limited view on what it does, how it does it, and what are the molecules involved.”
Bacteria also seem to have a wide variety of enclosed structures that are bound not by a lipid membrane but by a protein coat. Take carboxysomes, which evolved in bacteria twice, independently, to fix carbon. They and smaller, self-assembling nanocompartments have a polyhedral structure that looks shockingly like a viral capsid, the protein shell that encloses viral genomic material.
The catalog keeps getting longer: Komeili and his colleagues recently discovered a new lipid-bound organelle that accumulates iron, which they’ve dubbed the ferrosome. Bacteria seem to have a cornucopia of such organelles, with more waiting to be discovered. Scientists are now starting to explore what that means in the context of eukaryotic evolution. They hope either to establish direct evolutionary relationships among the growing list of structures, or to pinpoint factors that are unique and necessary for compartmentalization and complexity.
The Nucleus as Evolutionary Latecomer
Questions about how eukaryotes evolved tend to revolve around the order in which various cellular innovations emerged, and “how many features are actually ancient and existed in archaea and bacteria, before the evolution of the first eukaryote,” Fuerst explained.
Two major milestones defined the origin of eukaryotes. One, in keeping with the “true kernel” meaning of their name, was the appearance of the nucleus as a container for their DNA. The other was the formation of mitochondria, which are thought to have once been free-living bacteria that were engulfed by an ancestor of the archaea. Experts have disagreed about the most likely order and relative importance of those events. Some posit that gaining mitochondria was the essential change that set off eukaryotic evolution. Others theorize that eukaryotic evolution was well underway, and that an intricate membrane apparatus (which likely included a nucleus) was already present and helped to enable the uptake of the mitochondrial precursors.
No one knows whether the structures seen in bacteria represent primitive, intermediate steps in the evolution of eukaryotic organelles, or separate innovations that evolved independently of those of eukaryotes. It’s possible that the answer varies with each organelle. But even if the bacterial and eukaryotic organelles did evolve completely independently, the prokaryotic structures may be useful for understanding the eukaryotic ones.
This possibility was highlighted in April, when a pair of researchers offered an intriguing argument that the nucleus was a much later addition than had generally been believed. “We’re proposing that in fact, for much of eukaryotic evolution, the nucleus as we know it today didn’t exist,” said Michael Rout, a cell biologist at the Rockefeller University and one of the paper’s co-authors.
He and his colleague Mark Field at the University of Dundee in the U.K. noted that the nuclear pore complex (the gateway between the nucleus and the cytoplasm) consists of a blend of two protein types that are each found separately in other membrane structures. Based on further analysis, they proposed a chronology in which “the endomembrane system was already diverging and specializing before the nucleus as we know it came about,” Rout said. Meanwhile, the acquisition of mitochondria would have occurred in parallel — all adding up to a slow, stepwise progression from the archaea to the last eukaryotic common ancestor. During this process, many intermediate eukaryotes would have lacked a nucleus and other complex features.
“The eukaryotic lineage may have been well on its way to being a separate entity before what we would consider to be the recognizable feature of the nucleus came about,” said Joel Dacks, an evolutionary cell biologist at the University of Alberta who did not participate in the study.
And that implied that “the ancestors of modern eukaryotes may well have had simple internal membrane structures that may not be a million miles removed from the simple structures that are being identified in prokaryotes,” said Anthony Poole, a biologist at the University of Auckland in New Zealand.
“It does raise the question of whether any of the [prokaryotic structures] bear some functional similarities to this picture of the early stages of eukaryote evolution,” he added.
Moreover, if prokaryotes build and maintain these structures differently than eukaryotes do, then scientists can more confidently determine how and why compartmentalization might arise. “It brings eukaryogenesis out of the realm of extraordinary one-off circumstances,” Dacks said, “and into the realm of a really great scientific question that can be tractably addressed.”
A Step Beyond Speculation
When compartmentalization was thought of as a singular feature of eukaryotes, experts were often forced to speculate about how it came about, what the biophysical constraints were, and what selective advantages it might have. “That’s where these prokaryotes become really interesting,” Poole said. “If they show some features that are even mildly similar to what we see in eukaryotes, it allows us to broaden the question and attack from a different angle: Under what conditions might compartmentation provide some benefit? Or is it just the case that there’s no benefit whatsoever?”
The bacterial cases “suggest that there are multiple ways to do this, and that there could be a strong evolutionary advantage to doing so,” Dacks said.
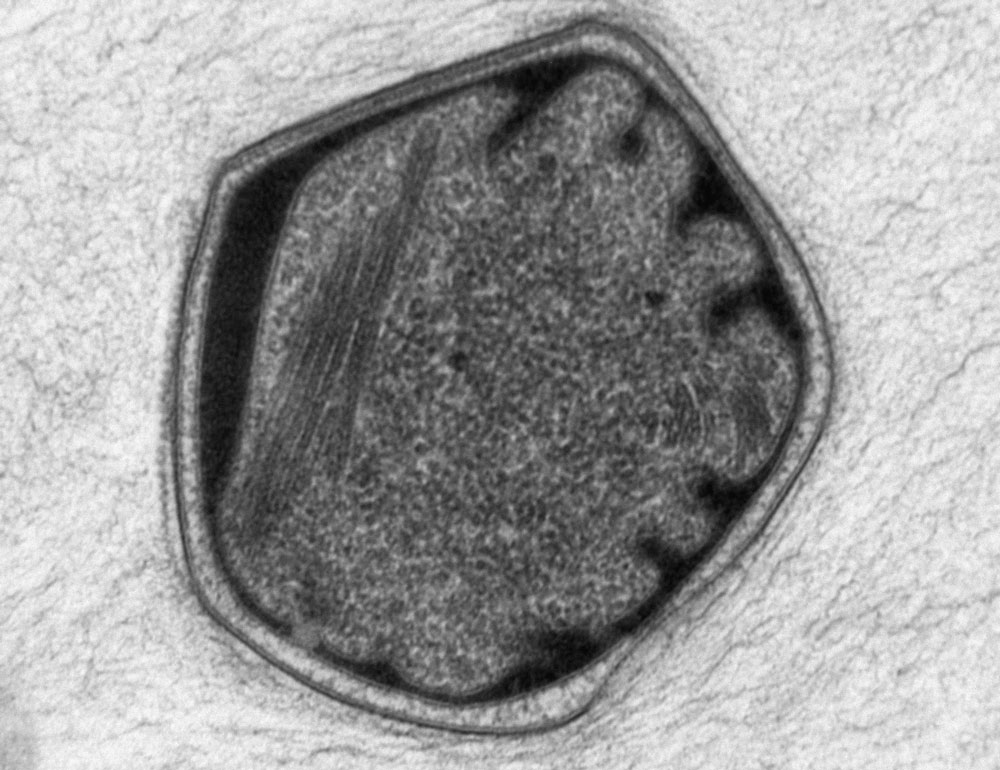
The anammoxosome, pictured here, is an organelle that produces energy for bacteria much as the mitochondrion produces energy for eukaryotes. Despite their similar function, the origins of the two organelles are completely different.
Laura van Niftrik, Willie J. C. Geerts, Elly G. van Donselaar, Bruno M. Humbel, Alevtyna Yakushevska, Arie J. Verklei, Mike S. M. Jetten, Marc Strous
That certainly seems to have been the case with energy production: The independent evolution of anammoxosomes in some kinds of bacteria and mitochondria in eukaryotes signify that “the compartmentalization of energy metabolism is beneficial to the cell,” said Laura van Niftrik, a microbial cell biologist at Radboud University in the Netherlands who studies the anammoxosome. “You see a trend in both prokaryotes and eukaryotes to compartmentalize certain traits or certain functions in order to better control them.”
Poole wants to see if this trend extends to the compartmentalization of genomic information. “We don’t really know why and whether compartmentation of genetic material is a good idea,” he said. But by examining it in different types of organisms — in the eukaryotic nucleus, in the planctomycete membrane system and even in giant viruses — he thinks researchers can start to outline drivers of compartmentalization and the conditions that might give rise to it. (At the very least, there appear to be certain biophysical constraints: A specific kind of protein fusion, for instance, seems to be required to manipulate membranes.)
This work not only suggests that compartmentalization is more prevalent among the various branches of the tree of life than people thought; it also indicates that this kind of complexity was not the critical innovation needed to trigger eukaryotic evolution. Komeili speculates that compartmentalization wasn’t necessarily “the lock that needed to be opened to give you more complicated cells in eukaryotes.” And therefore, “it didn’t have to be done in a particular way.”
Rather, eukaryotic characteristics likely emerged as part of a long, gradual trend, just as Rout’s work on the nuclear pore complex demonstrated. “It’s showing us that stepwise evolution is possible,” Devos said, “as opposed to a big explosive change from nothing to everything.”
Alternative Paths Toward an Answer
Some researchers are taking a different route. Kate Adamala, a synthetic biologist at the University of Minnesota, and her colleagues are building synthetic cells with some basic internal organization. So far, she said, “I can tell you a lot of things that don’t give rise to organelles.” Others are pursuing similar projects, such as attempting to make cells that consist only of protein components.
For their part, Komeili and his colleagues are hoping to use their research on magnetosomes and other bacterial organelles to figure out how to manipulate membranes and change their functions for novel applications, which could include identifying tumors and cleaning up toxic materials.
Meanwhile, though, “I teach introductory microbiology, and the textbook I’m tasked with using for the class still says that bacteria don’t have organelles,” said Anthony Vecchiarelli, a molecular biologist at the University of Michigan. It’s time to “fix the dogma that membrane-bound organelles are restricted to eukaryotes. That’s just not true anymore.”
Given that all of life is connected — whether in the deep evolutionary past, or in current symbiotic relationships (just think about all the bacteria residing in the human gut) — this new understanding of evolutionary history can give us more clues about where we came from. At the very least, “people are recognizing that there’s more diversity out there in the environment,” Dacks said, “and that the nice clean stories just don’t cut it anymore.”