The New Science of Evolutionary Forecasting

Michael Lässig can be certain that if he steps out of his home in Cologne, Germany, on the night of Jan. 19, 2030 — assuming he’s still alive and the sky is clear — he will see a full moon.
Lässig’s confidence doesn’t come from psychic messages he’s receiving from the future. He knows the moon will be full because physics tells him so. “The whole of physics is about prediction, and we’ve gotten quite good at it,” said Lässig, a physicist at the University of Cologne. “When we know where the moon is today, we can tell where the moon is tomorrow. We can even tell where it will be in a thousand years.”
Early in his career, Lässig made predictions about quantum particles, but in the 1990s, he turned to biology, exploring how genes evolved. In his research, Lässig was looking back in time, reconstructing evolutionary history. Looking ahead to evolution’s future was not something that biologists bothered doing. It might be possible to predict the motion of the moon, but biology was so complex that trying to predict its evolution seemed a fool’s errand.
But lately, evolution is starting to look surprisingly predictable. Lässig believes that soon it may even be possible to make evolutionary forecasts. Scientists may not be able to predict what life will be like 100 million years from now, but they may be able to make short-term forecasts for the next few months or years. And if they’re making predictions about viruses or other health threats, they might be able to save some lives in the process.
“As we collect a few examples of predictability, it changes the whole goal of evolutionary biology,” Lässig said.
Replaying the Tape of Life
If you want to understand why evolutionary biologists have been so loathe to make predictions, read “Wonderful Life,” a 1989 book by the late paleontologist Stephen Jay Gould.
The book is ostensibly about the Cambrian explosion, a flurry of evolutionary innovation that took place more than 500 million years ago. The oldest known fossils of many of today’s major animal groups date to that time. Our own lineage, the vertebrates, first made an appearance in the Cambrian explosion, for example.
But Gould had a deeper question in mind as he wrote his book. If you knew everything about life on Earth half a billion years ago, could you predict that humans would eventually evolve?
Gould thought not. He even doubted that scientists could safely predict that any vertebrates would still be on the planet today. How could they, he argued, when life is constantly buffeted by random evolutionary gusts? Natural selection depends on unpredictable mutations, and once a species emerges, its fate can be influenced by all sorts of forces, from viral outbreaks to continental drift, volcanic eruptions and asteroid impacts. Our continued existence, Gould wrote, is the result of a thousand happy accidents.
To illustrate his argument, Gould had his readers imagine an experiment he called “replaying life’s tape.” “You press the rewind button and, making sure you thoroughly erase everything that actually happened, go back to any time and place in the past,” he wrote. “Then let the tape run again and see if the repetition looks at all like the original.” Gould wagered that it wouldn’t.
Although Gould only offered it as a thought experiment, the notion of replaying the tape of life has endured. That’s because nature sometimes runs experiments that capture the spirit of his proposal.
Predictable Lizards
For an experiment to be predictable, it has to be repeatable. If the initial conditions are the same, the final conditions should also be the same. For example, a marble placed at the edge of a bowl and released will end up at the bottom of the bowl no matter how many times the action is repeated.
Biologists have found cases in which evolution has, in effect, run the same experiment several times over. And in some cases the results of those natural experiments have turned out very similar each time. In other words, evolution has been predictable.
One of the most striking cases of repeated evolution has occurred in the Caribbean. The islands there are home to a vast number of native species of anole lizards, which come in a staggering variety. The lizards live in the treetops, on forest floors and in open grassland. They come in a riot of colors and shapes. Some are blue, some are green and some are gray. Some are huge and bold while others are small and shy.
To understand how this diversity evolved, Jonathan Losos of Harvard University and his students gathered DNA from the animals. After they compared the genetic material from different species, the scientists drew an evolutionary tree, with a branch for every lizard species.
When immigrant lizards arrived on a new island, Losos found, their descendants could evolve into new species. It was as if the lizard tape of life was rewound to the same moment and then played again.
If Gould were right, the pattern of evolution on each island would look nothing like the pattern on the other islands. If evolution were more predictable, however, the lizards would tend to repeat the same patterns.
Losos and his students have found that evolution did sometimes veer off in odd directions. On Cuba, for example, a species of lizard adapted to spending a lot of time in the water. It dives for fish and can even sprint across the surface of a stream. You won’t find a fishing lizard on any other Caribbean island.
For the most part, though, lizard evolution followed predictable patterns. Each time lizards colonized an island, they evolved into many of the same forms. On each island, some lizards adapted to living high in trees, evolving pads on their feet for gripping surfaces, along with long legs and a stocky body. Other lizards adapted to life among the thin branches lower down on the trees, evolving short legs that help them hug their narrow perches. Still other lizards adapted to living in grass and shrubs, evolving long tails and slender trunks. On island after island, the same kinds of lizards have evolved.
“I think the tide is running against Gould,” Losos said. Other researchers are also finding cases in which evolution is repeating itself. When cichlid fish colonize lakes in Africa, for example, they diversify into the same range of forms again and again.
“But the question is: What’s the overall picture?” Losos asked. “Are we cherry-picking the examples that work against him, or are we going to find that most of life is deterministic? No one is going to say Gould is completely wrong. But they’re not going to say he’s completely right either.”
Evolution in a Test Tube
Natural experiments can be revealing, but artificial experiments can be precise. Scientists can put organisms in exactly the same conditions and then watch evolution unfold. Microbes work best for this kind of research because scientists can rear billions of them in a single flask and the microbes can go through several generations in a single day. The most spectacular of these experiments has been going on for 26 years — and more than 60,000 generations — in the lab of Richard Lenski at Michigan State University.
Lenski launched the experiment with a single E. coli microbe. He let it divide into a dozen genetically identical clones that he then placed in a dozen separate flasks. Each flask contained a medium — a cocktail of chemicals mixed into water — that Lenski created especially for the experiment. Among other ingredients, it contained glucose for the bacteria to feed on. But it was a meager supply, which ran out after just a few hours. The bacteria then had to eke out their existence until the next morning, when Lenski or his students transferred a little of the microbe-laced fluid into a fresh flask. With a new supply of glucose, they could grow for a few more hours. Lenski and his students at Michigan State have been repeating this chore every day since.
Lenski thought the tape of life would replay differently with each rewind. But that’s not what happened.
At the outset, Lenski wasn’t sure what would happen, but he had his suspicions. He expected mutations to arise randomly in each line of bacteria. Some would help the microbes reproduce faster while others would be neutral or even harmful. “I imagined they’d be running off in one direction or another,” Lenski said.
In other words, Lenski thought the tape of life would replay differently with each rewind. But that’s not what happened. What Lenski witnessed was strikingly similar to the evolution that Jonathan Losos has documented in the Caribbean.
Lenski and his students have witnessed evolutionary oddities arise in their experiment — microbial versions of the Cuban fishing lizards, if you will. In 2003, Lenski’s team noticed that one line of bacteria had abruptly switched from feeding on glucose to feeding on a compound called citrate. The medium contains citrate to keep iron in a form that the bacteria can absorb. Normally, however, the bacteria don’t feed on the citrate itself. In fact, the inability to feed on citrate in the presence of oxygen is one of the defining features of E. coli as a species.
But Lenski has also observed evolution repeat itself many times over in his experiment. All 12 lines have evolved to grow faster on their meager diet of glucose. That improvement has continued to this day in the 11 lines that didn’t shift to citrate. Their doubling time — the time it takes for them to double their population — has sped up 70 percent. And when Lenski and his students have pinpointed the genes that have mutated to produce this improvement, they are often the same from one line to the next.
“That’s not at all what I expected when I started the experiment,” Lenski said. “I evidently was wrong-headed.”
Getting Complex Without Getting Random
Lenski’s results have inspired other scientists to set up more complex experiments. Michael Doebeli, a mathematical biologist at the University of British Columbia, wondered how E. coli would evolve if it had two kinds of food instead of just one. In the mid-2000s, he ran an experiment in which he provided glucose — the sole staple of Lenski’s experiment — and another compound E. coli can grow on, known as acetate.
Doebeli chose the two compounds because he knew that E. coli treats them very differently. When given a choice between the two, it will devour all the glucose before switching on the molecular machinery for feeding on acetate. That’s because glucose is a better source of energy. Feeding on acetate, by contrast, E. coli can only grow slowly.
Something remarkable happened in Doebeli’s experiment — and it happened over and over again. The bacteria split into two kinds, each specialized for a different way of feeding. One population became better adapted to growing on glucose. These glucose-specialists fed on the sugar until it ran out and then slowly switched over to feeding on acetate. The other population became acetate-specialists; they evolved to switch over to feeding on acetate even before the glucose supply ran out and could grow fairly quickly on acetate.
When two different kinds of organisms are competing for the same food, it’s common for one to outcompete the other. But in Doebeli’s experiment, the two kinds of bacteria developed a stable coexistence. That’s because both strategies, while good, are not perfect. The glucose-specialists start out growing quickly, but once the glucose runs out, they slow down drastically. The acetate-specialists, on the other hand, don’t get as much benefit from the glucose. But they’re able to grow faster than their rivals once the glucose runs out.
Doebeli’s bacteria echoed the evolution of lizards in the Caribbean. Each time the lizards arrived on an island, they diversified into many of the same forms, each with its own set of adaptations. Doebeli’s bacteria diversified as well — and did so in flask after flask.
To get a deeper understanding of this predictable evolution, Doebeli and his postdoctoral researcher, Matthew Herron, sequenced the genomes of some of the bacteria from these experiments. In three separate populations they discovered that the bacteria had evolved in remarkable parallel. In every case, many of the same genes had mutated.
Although Doebeli’s experiments are more complex than Lenski’s, they’re still simple compared with what E. coli encounters in real life. E. coli is a resident of the gut, where it feeds on dozens of compounds, where it coexists with hundreds of other species, where it must survive changing levels of oxygen and pH, and where it must negotiate an uneasy truce with our immune system. Even if E. coli’s evolution might be predictable in a flask of glucose and acetate, it would be difficult to predict how the bacteria would evolve in the jungle of our digestive system.
And yet scientists have been surprised to find that bacteria evolve predictably inside a host. Isabel Gordo, a microbiologist at the Gulbenkian Institute of Science in Portugal, and her colleagues designed a clever experiment that enabled them to track bacteria inside a mouse. Mice were inoculated with a genetically identical population of E. coli clones. Once the bacteria arrived in the mice’s guts, they started to grow, reproduce and evolve. And some of the bacteria were carried out of the mouse’s body with its droppings. The scientists isolated the experimental E. coli from the droppings. By examining the bacteria’s DNA, the scientists could track their evolution from one day to the next.
The scientists found that it took only days for the bacteria to start evolving. Different lineages of E. coli picked up new mutations that made them reproduce faster than their ancestors. And again and again, they evolved many of the same traits. For example, the original E. coli couldn’t grow if it was exposed to a molecule called galactitol, which mammals make as they break down sugar. However, Gordo’s team found that as E. coli adapted to life inside a mouse, it always evolved the ability to withstand galactitol. The bacteria treated a living host like one of Lenski’s flasks — or an island in the Caribbean.
Evolution’s Butterfly Effect
Each new example of predictable evolution is striking. But, as Losos warned, we can’t be sure whether scientists have stumbled across a widespread pattern in nature. Certainly, testing more species will help. But Doebeli has taken a very different approach to the question: He’s using math to understand how predictable evolution is overall.
Doebeli’s work draws on pioneering ideas that geneticists like Sewall Wright developed in the early 1900s. Wright pictured evolution like a hilly landscape. Each point on the landscape represents a different combination of traits — the length of a lizard’s legs versus the width of its trunk, for example. A population of lizards might be located on a spot on the landscape that represents long legs and a narrow trunk. Another spot on the landscape would represent short legs and a narrow trunk. And in another direction, there’s a spot representing long legs and a thick trunk.
The precise combinations of traits in an organism will influence its success at reproducing. Wright used the elevation of a spot on the evolutionary landscape to record that success. An evolutionary landscape might have several peaks, each representing one of the best possible combinations. On such a landscape, natural selection always pushes populations up hills. Eventually, a population may reach the top of a hill; at that point, any change will lead to fewer offspring. In theory, the population should stay put.
The future of evolution might seem easy to predict on such a landscape. Scientists could simply look at the slope of the evolutionary landscape and draw a line up the nearest hill.
“This view is just simply wrong,” said Doebeli.
That’s because the population’s evolution changes the landscape. If a population of bacteria evolves to feed on a new kind of food, for example, then the competition for that food becomes fierce. The benefit of specializing on that food goes down, and the peak collapses. “It’s actually the worst place to be,” Doebeli said.
Over short periods of time, it is predictable, if you have enough information. But you can’t predict it over long periods of time.
To keep climbing uphill, the population has to veer onto a new course, toward a different peak. But as it travels in a new direction, it alters the landscape yet again.
Recently, Doebeli and Iaroslav Ispolatov, a mathematician at the University of Santiago in Chile, developed a model to understand how evolution works under these more complicated conditions. Their analysis suggests that evolution is a lot like the weather — in other words, it’s difficult to predict.
In the early 1960s, a scientist at the Massachusetts Institute of Technology named Edward Lorenz developed one of the first mathematical models of weather. He hoped that they would reveal repeatable patterns that would help meteorologists predict the weather more accurately.
But Lorenz discovered just the opposite. Even a tiny change to the initial conditions of the model led, in time, to drastically different kinds of weather. In other words, Lorenz had to understand the model’s initial conditions with perfect accuracy to make long-term predictions about how it would change. Even a slight error would ruin the forecast.
Mathematicians later dubbed this sensitivity chaos. They would find that many systems — even surprisingly simple ones — behave chaotically. One essential ingredient for chaos is feedback — the ability for one part of the system to influence another, and vice versa. Feedback amplifies even tiny differences into big ones. When Lorenz presented his results, he joked that the flap of a butterfly’s wings in Brazil could set off a tornado in Texas.
Evolution has feedbacks, too. A population evolves to climb the evolutionary landscape, but its changes alter the landscape itself. To see how these feedbacks affected evolution, Doebeli and Ispolatov created their own mathematical models. They would drop populations onto the evolutionary landscape at almost precisely the same spot. And then they followed the populations as they evolved.
In some trials, the scientists only tracked the evolution of a few traits, while in others, they tracked many. They found that in the simple models, the populations tended to follow the same path, even though they started out in slightly different places. In other words, their evolution was fairly easy to predict.
But when the scientists tracked the evolution of many traits at once, that predictability disappeared. Despite starting out under almost identical conditions, the populations veered off on different evolutionary paths. In other words, evolution turned to chaos.
Doebeli and Isplolatov’s research suggests that for the most part, evolution is too chaotic to be predicted with any great accuracy. If they are right, then the successes that scientists like Losos and Lenski have had in finding predictable evolution are the exceptions that prove the rule. The future of evolution, for the most part, is as fundamentally unknowable as the future of the weather.
This conclusion may seem strange coming from Doebeli. After all, he has conducted experiments on E. coli that have shown just how predictable evolution can be. But he sees no contradiction. “It’s just a matter of time scales,” he said. “Over short periods of time, it is predictable, if you have enough information. But you can’t predict it over long periods of time.”
Darwin’s Prophets
Even over short periods of time, accurate forecasts can save lives. Meteorologists can make fairly reliable predictions about treacherous weather a few days in advance. That can be enough time to evacuate a town ahead of a hurricane or lay in supplies for a blizzard.
Richard Lenski thinks that recent studies raise the question of whether evolutionary forecasting could also provide practical benefits. “I think the answer is definitely yes,” he said.
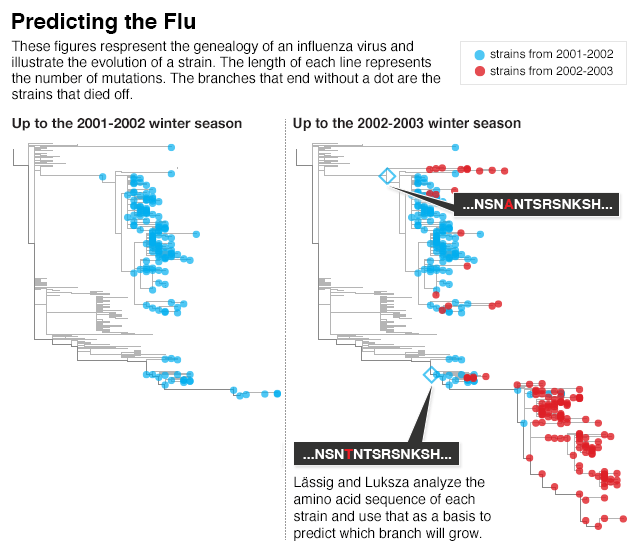
One of the most compelling examples comes from Lässig. Using his physics background, he is working on a way to forecast the flu.
Worldwide, the flu kills as many as 500,000 people a year. Outside of the tropics, infections cycle annually from a high in winter to a low in summer. Flu vaccines can offer some protection, but the rapid evolution of the influenza virus makes it a moving target for vaccination efforts.
The influenza virus reproduces by invading the cells in our airway and using their molecular machinery to make new viruses. It’s a sloppy process, which produces many new mutants. Some of their mutations are harmful, crippling the viruses so that they can’t reproduce. But other mutations are harmless. And still others will make new flu viruses even better at making copies of themselves.
As the flu virus evolves, it diverges into many different strains. A vaccine that is effective against one strain will offer less protection against others. So vaccine manufacturers try to provide the best defense each flu season by combining the three or four most common strains of the flu.
There’s a problem with this practice, however. Manufacturing a new season’s flu vaccines takes several months. In the United States and other countries in the Northern Hemisphere, vaccine manufacturers must decide in February which strains to use for the flu season that starts in October. They often make the right prediction. But sometimes a strain that’s not covered by the vaccine unexpectedly comes to dominate a flu season. “If something goes wrong, it can cost thousands of lives,” Lässig said.
A few years ago, Lässig started to study the vexing evolution of the flu. He focused his attention on the rapidly evolving proteins that stud the shell of the flu virus, called hemagglutinin. Hemagglutinin latches on to receptors on our cells and opens up a passageway for the virus to invade.
When we get sick with the flu, our immune system responds by building antibodies that grab onto the tip of the hemagglutinin protein. The antibodies prevent the viruses from invading our cells and also make it easier for immune cells to detect the viruses and kill them. When we get flu vaccines, they spur our immune system to make those antibodies even before we get sick so that we’re ready to wipe out an infection as soon as it starts.
Scientists have been sequencing hemagglutinin genes from flu seasons for more than 40 years. Poring over this trove of information, Lässig was able to track the evolution of the viruses. He found that most mutations that altered the tip of the hemagglutinin protein helped the viruses reproduce more, probably because they made it difficult for antibodies to grab onto them. Escaping the immune system, they can make more copies of themselves.
Each strain of the flu has its own collection of beneficial mutations. But Lässig noticed that the viruses also carry harmful mutations in their hemagglutinin gene. Those harmful mutations make hemagglutinin less stable and thus less able to open up cells for invasion.
It occurred to Lässig that these mutations might determine which strains would thrive in the near future. Perhaps a virus with more beneficial mutations would be more likely to escape people’s immune systems. And if they escaped destruction, they would make more copies of themselves. Likewise, Lässig theorized, the more harmful mutations a virus had, the more it would struggle to invade cells.
If that were true, then it might be possible to predict which strains would become more or less common based on how many beneficial and harmful mutations they carried. Working with Columbia University biologist Marta Łuksza, he came up with a way to score the evolutionary potential of each strain of the flu. For each beneficial mutation, a strain earned a point. For each harmful one, Lässig and Łuksza took a point away.
The scientists examined thousands of strains of the flu that have been sampled since 1993. They would calculate the score for every strain in a given year and then use that score to predict how it would fare the following year. They correctly forecast whether a strain would grow or decline about 90 percent of the time. “It’s a simple procedure,” Lässig said. “But it works reasonably well.”
Lässig and his colleagues are now exploring ways to improve their forecast. Lässig hopes to be able to make predictions about future flu seasons that the World Health Organization could consult as they decide which strains should be included in flu vaccines. “It’s just a question of a few years,” he said.
The flu isn’t the only disease that evolutionary forecasting could help combat. Bacteria are rapidly evolving resistance to antibiotics. If scientists can predict the path that the microbes will take, they may be able to come up with strategies for putting up roadblocks.
Forecasting could also be useful in fighting cancer. When cells turn cancerous, they undergo an evolution of their own. As cancer cells divide, they sometimes gain mutations that let them grow faster or escape the immune system’s notice. It may be possible to forecast how tumors will evolve and then plan treatments accordingly.
Beyond its practical value, Lässig sees a profound importance to being able to predict evolution. It will bring the science of evolutionary biology closer to other fields like physics and chemistry. Lässig doesn’t think that he’ll be able to forecast evolution as easily as he can the motion of the moon, but he hopes that there’s much about evolution that will prove to be predictable. “There’s going to be a boundary, but we don’t know where the boundary is,” he said.
This article was reprinted on BusinessInsider.com.