A Neurobiologist Thinks Big — and Small

Ed Boyden, the leader of the Synthetic Neurobiology Group at the Massachusetts Institute of Technology, argues that when it comes to brain research, “We want to see everything and control everything.”
Ken Richardson for Quanta Magazine
Introduction
Ed Boyden has grand dreams. The aims of this neuroscientist at the Massachusetts Institute of Technology include decoding all of biology and achieving human enlightenment. But he also has his eye on the path that will get him to each goal. As he dives into an explanation of a world-changing idea, he frequently declares, “Step one!”
Seeing the micro and macro at the same time is what Boyden’s latest invention, expansion microscopy, is all about. Researchers start by embedding a piece of brain or other tissue in an expanding polymer, similar to the absorbent stuff inside disposable diapers. The polymer forms a dense web; its threads, only a nanometer or two apart, are anchored to points within the sample. Then the scientists add water. The material blows up as much as a hundredfold in volume, pushing the tissue’s molecules apart like an expanding balloon while maintaining their relative positions. This technique lets scientists zoom in on nanoscale details with ordinary microscopes. In a slice of mouse brain, for example, a field of blobs resolves into individual hamburger-shaped synapses, the meeting points between neurons.
Before expansion microscopy, Boyden helped invent optogenetics, a tool that enables scientists to turn brain cells on and off with light. The journal Nature Methods chose optogenetics as its 2010 Method of the Year, and researchers have found wide-ranging applications for it. In a 2016 study, for example, Boyden and his neuroscience colleague Li-Huei Tsai, the director of MIT’s Picower Institute for Learning and Memory, used optogenetics to trigger brain waves called gamma oscillations in mice engineered to model Alzheimer’s disease. When they drove the brain waves at a frequency of 40 times per second for an hour, amyloid plaques — the protein clumps linked to Alzheimer’s disease — began to disappear from the brains of the mice.
Then the researchers showed they could create the same effect without optogenetics simply by having mice stare at a light flickering 40 times a second. Tsai and Boyden have founded a company called Cognito Therapeutics to try to create videos that treat Alzheimer’s disease in human patients. They’ve just begun human trials.
Optogenetics transformed neuroscience, but Boyden describes the original research on the technology as a “side collaboration” he worked on while earning his doctorate at Stanford University. The 38-year-old has a 60-page resume and three companies. A jumble of prizes sits on a high shelf in his office, including the Breakthrough Prize in Life Sciences from 2016; when I met with him, he had just returned from Philadelphia, where he had accepted the Drexel Prize in Biotechnology. Boyden has two young children (he is married to Xue Han, a noted neuroscientist at Boston University). When I asked if he has some secret to getting so much done — like maybe he doesn’t sleep? — he seemed confused by the question.
The team that Boyden leads at MIT’s Media Lab, the Synthetic Neurobiology Group, has a mission that’s both grandiose and straightforward: “We want to see everything and control everything,” Boyden said. “And we need new technology to do that.” I talked with Boyden about his very large visions for the very small. An edited and condensed version of our conversation follows.

In a multiwall specimen plate (left), expanded slices of brain tissue wait for study under an inverted microscope (right) in Boyden’s lab.
Ken Richardson for Quanta Magazine
You have a five-year-old and an eight-year-old. Let’s say you were trying to explain optogenetics to your eight-year-old—
Well, when he was five he tried to explain it to me.
What did he say?
His explanation went like this: “The brain has some plugs and some wires, and it goes beep beep beep and makes me do what I do.” And I was like, that’s pretty darn good!
But to be slightly more biophysically accurate: If you can control a brain cell, then you can see what it does. Does it trigger a response, or a sensation, or a decision or a memory? Brain cells compute using electricity. So how do you drive electricity? One of the ways is to put electricity in directly, like cochlear implants do for the deaf. But the electricity goes in all directions. It’s not very precise. Light, on the other hand, can be aimed at individual cells. If we can use light, that’d be much more precise than direct electrical stimulation. The problem is, brain cells don’t respond to light.
So we get molecules from single-celled microbes that are like little solar panels. They convert light into electricity. These molecules, called opsins, are genetically encoded. We can cut out the genes from the microbes and paste them into the brain cells we want to study. The brain cells will manufacture these little solar panels. And when you shine light on the cells, the opsins will convert light into electrical signals, and the electrical signals control the neuron, and then you figure out what triggering that neuron does.
What kinds of things can you do with that tool?
My co-inventor Karl Deisseroth [at Stanford University] and I started thinking this up when we were both students, in the year 2000. We had three initial goals. Number one, if you can activate specific cells, can you correct a brain disorder? Second, could you figure out what drives a behavior, or a want, or a movement or sensation? And third, could you reprogram the computations of the brain? Could you take a memory, enter it into the brain and then see what happens?
Has anyone done that?
Well, my colleague here at MIT, Susumu Tonegawa, did something like that with mice. His group used one of our molecules from algae that’ll activate a neuron under blue light. They engineered mice so that only recently active neurons would express the gene. The mice would learn a memory, and the neurons involved in the memory would become light-activated. Then they shined blue light [on those neurons] and found that they could induce the recall of the memory.
What are some of the coolest applications of optogenetics you’ve seen so far?
There are so many! New papers come out every day. One of my favorite studies was done by Dayu Lin [now at New York University] and David Anderson [at the California Institute of Technology]. They were studying violence and aggression in male mice, which are territorial. They found that certain neurons were active when male mice fought. Then they put our molecule into those cells. And when they activated the neurons they found that the mice would attack basically whatever was next to them, even a rubber glove.
Let’s talk about expansion microscopy. I watched your TED talk, where you demonstrated how much diaper fibers can expand. That was a good origin story — is it really how the idea came about?
No, not at all!
Around 2012 we started doing super-resolution and electron microscopy in our group. It was really hard. And it was very slow. Also, if you do electron microscopy you don’t get much molecular information. You know the shapes of things, but not what molecules are there. So almost as a joke we started thinking: Why don’t we expand the brain?
I wanted to expand brains to move the molecules farther apart so we could label them better. Biomolecules are all made of the same building blocks in different orders, right? To tell them apart you really have to tag them. But there’s no room to tag them in the brain because they’re all jam-packed together.
Two really great grad students, Fei Chen [now at the Broad Institute] and Paul Tillberg [now at the Janelia Research Campus], were in the lab then. We got inspired by the papers of Toyoichi Tanaka, a physicist who studied responsive polymers here at MIT in the late 1970s and early 1980s. He had worked on these polymers that expand a thousandfold, very similar to the stuff in baby diapers. We set out to take a specimen and embed it in these “hyperswellable” polymers. Once we figured out the path, it started to work pretty quickly.
But after you blow something up, your microscope still has the same resolution. Does just having the components farther apart make things clearer? Or is it all about creating enough space to label molecules?
Turns out that it’s both. Microscopes have something called a diffraction limit. You just can’t see things much smaller than the wavelength of light. And unfortunately, that limit is right where things get interesting in biology. But if we expand a tissue twentyfold, a microscope with a 200-nanometer diffraction limit will now effectively have a 10-nanometer resolution.
And in addition, you make room around the biomolecules. It’s like you’re surrounding each biomolecule with a little virtual test tube made of chemicals of your choosing. So you can run all sorts of interesting analyses.
What does this help reveal about the brain that other methods can’t?
Brain circuits are large objects, right? I mean, some brain cells in the human brain are centimeters in size. And some neurons in our spinal cord are the largest cells in our body. If we want to understand how information flows, from sensation, to decision making, to action, we have to see the entire thing.
And yet the neural connections are nanoscale. All the molecules that make neurons do what they do are nanoscale. If you want to learn enough about a neural circuit that you can understand how it works, you have to see a large, three-dimensional object with nanoscale precision. And that’s what expansion lets you do.
These images demonstrate the power of expansion microscopy to bring out nanoscale details in brain tissue. At the top (a) is a magnified section of the hippocampus from a mouse. The area marked by the box, when viewed without expansion (b), looks indistinct. Expansion of the same area allows the extremely fine axons and dendrites of neurons (marked by arrows) to be crisp and clear (c). Synthetic Neurobiology Group/MIT, The Media Lab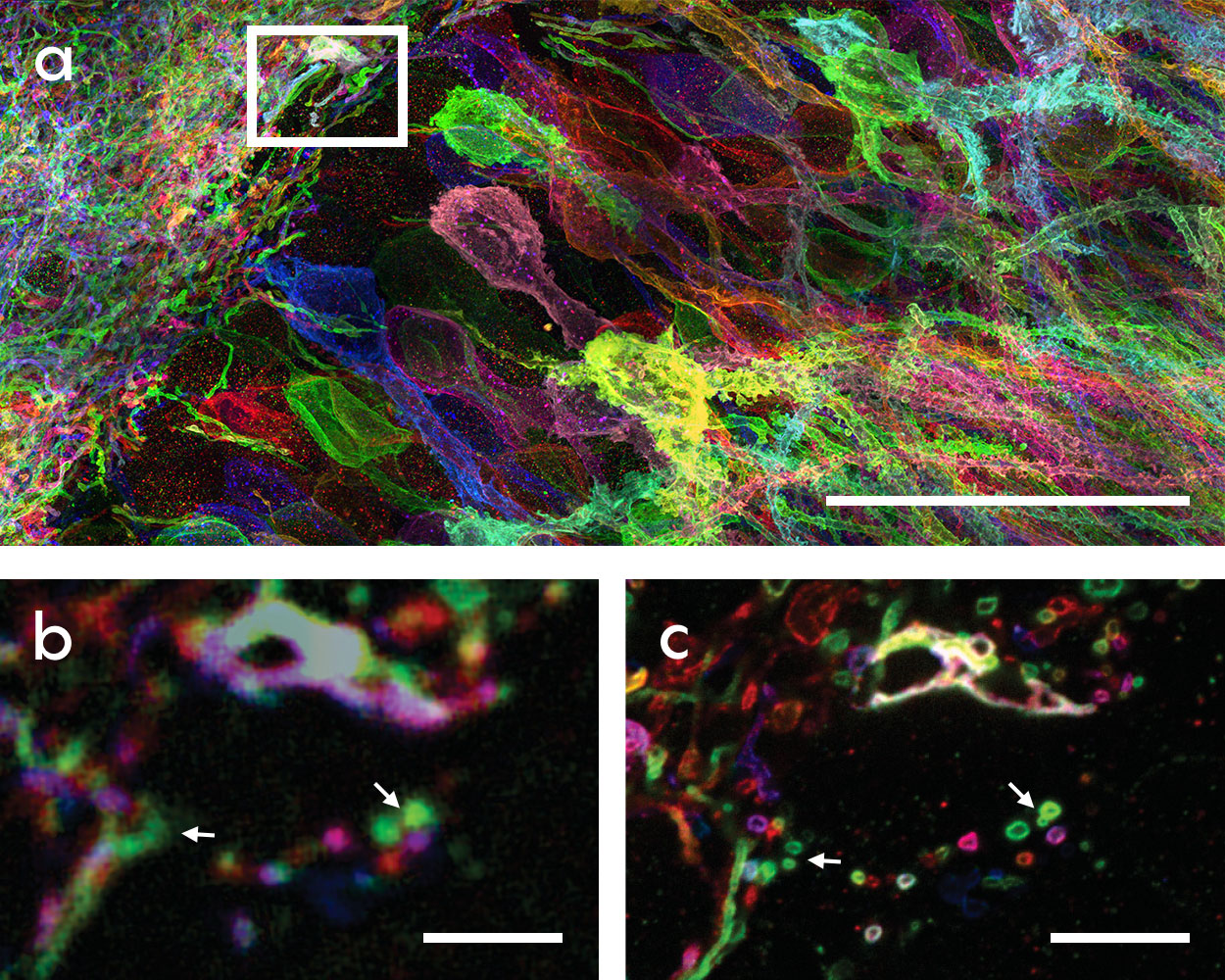
You’ve already applied this to cancer diagnosis.
We collaborated with several pathologists, including Andy Beck, then at Harvard, to see if, by expanding biopsies, we could diagnose early breast cancer better. Early disease detection is a big goal right now. But early diagnosis means seeing subtle things, and subtle often means nanoscale. These are diagnoses that really challenge human judgment. After we expanded the biopsies, we could train a machine-learning program to diagnose more accurately than with unexpanded biopsies.
What else is on the horizon for expansion microscopy?
Since our cancer paper came out, many people have emailed me — experts on prostate cancer, pancreatic cancer.… People have already published papers using expansion to map out how chromosomes look in cell division. We have a student working with our group and those of George Church and Ting Wu at Harvard to expand the genome and see it in its three-dimensional glory in an actual cell in a tissue, which has not been done before. We’re starting to talk to virologists about the idea of expanding a virus. People are applying expansion microscopy to all sorts of stuff. They’ve expanded human brain tissues from epilepsy patients. They’ve expanded circuits in the mouse brain involved with learning and Parkinson’s disease. They’ve mapped out tunnels used by viruses to spread from cell to cell. They’ve mapped out blood vessel structures involved with the blood-brain barrier. It’s very gratifying to see it take off so quickly.

Boyden’s accomplishments with optogenetics, expansion microscopy and other techniques have already brought him some of the highest honors in science.
Ken Richardson for Quanta Magazine
I’ve read that one of your group’s missions is sharing your methods with other researchers.
We’ve trained maybe around 300 groups in academia and industry on how to do expansion. We ran a summer school at the Janelia Research Campus in Virginia, and we ran two workshops at the Salk Institute in San Diego. We host visitors to come by our group nearly every week to watch us do expansion. And we’ve shared optogenetics tools with literally thousands of research groups in academia and industry now.
It’s weird because lots of people thank me for sharing. I guess it’s because in the old days some people built tools and tried to hoard them. But for me it’s like, Why are you thanking me? I should be thanking you for using our technology. If we build tools that people don’t use, then we have failed.
How does your group come up with these new tools?
The way science works is, if you are lucky and do something well, then people want to work for you. And if you do something well a second time, then everybody wants to work for you! Our group has now been lucky many, many times in a row, so the people working in the group are just amazing.
I think of us as a thinking-backward group. We think backward from a problem, and we survey all the disciplines of science and engineering to home in on the best possible technology we can think up. As a result, we often do the opposite of what other people do. If other people are trying to watch it, let’s control it. If other people are trying to zoom in, let’s blow it up. I think all the great problems in biology are obvious, but people kind of talk themselves out of confronting them.
What do you mean by that?
People often ask me, “What biological question are you interested in?” And I say, “I want to solve the brain.”
And to do that we need tools to map the molecules throughout the cells throughout the brain. Look at the history of science. Chemistry before the periodic table was very difficult. But now there’s the periodic table; there’s a list of basically all the building blocks you need to know. Physics has Maxwell’s equations and Newton’s laws and the equations of quantum mechanics; you could fit all the laws of physics on a single page. I think we can maybe have biology go through a similar transition, with the right kind of technology that lets us see the building blocks of life and how they interact.
And what will happen then?
My ultimate dream is, can we achieve a certain kind of enlightenment in the human condition? If we understand how the brain generates desires and irrational behaviors that cause suffering, could we point our minds toward a more suffering-free state?
That’s my primary motivation. Curing all these brain disorders is also a big goal for us, but it’s sort of a by-product of this bigger goal. Because if we don’t understand what we do and why, we might be able to live longer — but will we achieve meaning?
Let’s say it’s 30 years from now and you’ve figured everything out —
Hopefully not that long, but okay.
[Laughing] Okay, what’s your timeline for solving biology?
Well, I don’t know for sure, right? But I think things are moving much faster now than I anticipated. First of all, neurotechnology became cool. I started at the Media Lab in part because I had trouble getting a job. I was turned down by most of the departments I applied to for faculty positions, and I got hired because of sheer dumb luck that the Media Lab had a job opening at the time. The influx of talent into neurotechnology from other fields is starting to accelerate things.
So in less than 30 years …
Who knows? Who knows.
… when you’ve solved biology and you know how it all works, will that be disappointing at all? To have no more mystery about the mind, or why we do things?
The premise of the question is that the mystery would go away. But I don’t know if the premise is correct.
We know lots and lots about water, but does that erase the meditative quality of sitting on a beach and watching the waves? We know a heck of a lot about how stars work, through fusion of hydrogen and the creation of all the elements and so forth. But does that make it so nobody wants to look at the night sky? I think there are many kinds of wonder. And some are untouched, if not enhanced, by science.




