Inside Ancient Asteroids, Gamma Rays Made Building Blocks of Life
Kristina Armitage/Quanta Magazine
Introduction
In 2021, the Hayabusa2 space mission successfully delivered a morsel of the asteroid 162173 Ryugu to Earth — five grams of the oldest, most pristine matter left over from the solar system’s formation 4.5 billion years ago. Last spring, scientists revealed that the chemical composition of the asteroid includes 10 amino acids, the building blocks of proteins. The discovery added to the evidence that the primordial soup from which life on Earth arose may have been seasoned with amino acids from pieces of asteroids.
But where did these amino acids come from? The amino acids flowing through our ecosystems are products of cellular metabolism, mostly in plants. What nonbiological mechanism could have put them in meteorites and asteroids?
Scientists have thought of several ways, and recent work by researchers in Japan points to a significant new one: a mechanism that uses gamma rays to forge amino acids. Their discovery makes it seem even more likely that meteorites could have contributed to the origin of life on Earth.
Despite their cachet as an essential part of life’s chemistry, amino acids are simple molecules that can be cooked up artlessly from carbon, oxygen and nitrogen compounds if there’s sufficient energy. Seventy years ago, famous experiments by Stanley Miller and Harold Urey proved that an electrical discharge in a gaseous mixture of methane, ammonia and hydrogen (which at the time was incorrectly thought to mimic Earth’s early atmosphere) was all it took to make a mixture of organic compounds that included amino acids. Later laboratory work suggested that amino acids could also potentially form in sediments near hydrothermal vents on the seafloor, and a discovery in 2018 confirmed that this does sometimes occur.
The possibility that the original amino acids might have come from space began to catch on after 1969, when two large meteorites — the Murchison meteorite in southeastern Australia and the Allende meteorite in Mexico — were recovered promptly after their impacts. Both were carbonaceous chondrites, a rare class of meteorites resembling Ryugu that scientists think accreted from smaller icy bodies after the solar system first formed. Both also contained small but significant amounts of amino acids, although scientists couldn’t rule out the possibility that the amino acids were contaminants or byproducts of their impact.
Still, space scientists knew that the icy dust bodies that formed carbonaceous chondrites were likely to contain water, ammonia and small carbon molecules like aldehydes and methanol, so the elemental constituents of amino acids would have been present. They needed only a source of energy to facilitate the reaction. Experimental work suggested that ultraviolet radiation from supernovas could have been strong enough to do it. Collisions between the dust bodies could also have heated them enough to produce a similar effect.
“We know a lot of ways to make amino acids abiologically,” said Scott Sandford, a laboratory astrophysicist at NASA’s Ames Research Center. “And there’s no reason to expect that they didn’t all happen.”
Now a team of researchers at Yokohama National University in Japan led by the chemists Yoko Kebukawa and Kensei Kobayashi have shown that gamma rays could also have produced the amino acids in chondrites. In their new work, they showed that gamma rays from radioactive elements in the chondrites — most probably aluminum-26 — could convert the carbon, nitrogen and oxygen compounds into amino acids.
Of course, gamma rays can destroy organic compounds as easily as it can make them. But in the Japanese team’s experiments, “the enhancement of amino acid production by the radioisotopes was more effective than decomposition,” Kebukawa said, so the gamma rays produced more amino acids than they destroyed. From the rates of production observed in their experiments, the researchers calculated very roughly that gamma rays could have raised the concentration of amino acids in a carbonaceous chondrite asteroid to the levels seen in the Murchison meteorite in as little as 1,000 years or as many as 100,000.
Since gamma rays, unlike ultraviolet light, can penetrate deep into the interior of an asteroid or meteorite, this mechanism could have extra relevance to origin-of-life scenarios. “It opens up a whole new environment in which amino acids can be made,” Sandford said. If meteorites are big enough, “the middle part of them could survive atmospheric entry even if the outside ablates away,” he explained. “So you’re not only making [amino acids] but you’re making them on the path to get to a planet.”
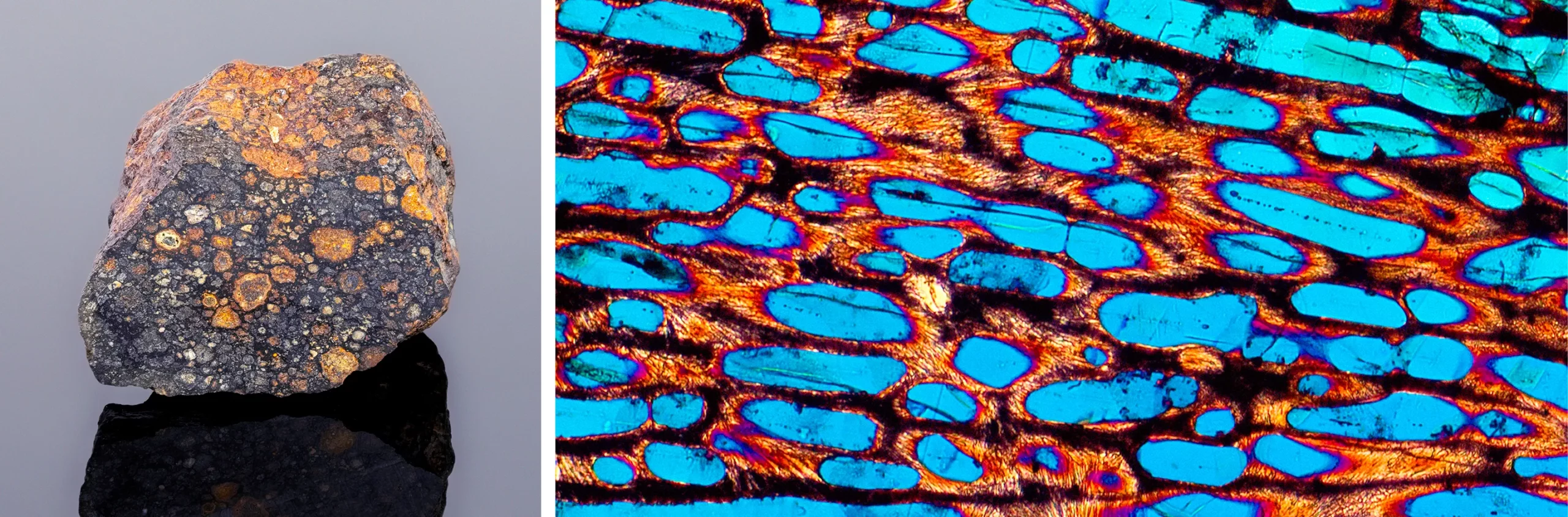
The meteorites called carbonaceous chondrites, such as the one at left, accreted from smaller icy bodies that contained mixtures of compounds rich in carbon, nitrogen and oxygen. Their conglomerated structure is visible in a magnified cross section.
Susan E. Degginger/Alamy Stock Photo (left); Laurence Garvie/ Buseck Center for Meteorite Studies, Arizona State University
One requirement of the new mechanism is that small amounts of liquid water must be present to support the reactions. That might seem like a significant limitation — “I can easily imagine that people think liquid water hardly exists in space environments,” Kebukawa said. But carbonaceous chondrite meteorites are full of minerals such as hydrated silicates and carbonates that only form in the presence of water, she explained, and tiny amounts of water have even been found trapped inside some of the mineral grains in chondrites.
From such mineralogical evidence, said Vassilissa Vinogradoff, an astrochemist at Aix-Marseille University in France, scientists know that young asteroids held significant amounts of liquid water. “The aqueous alteration phase of these bodies, which is when the amino acids in question would have had a chance to form, was a period of about a million years,” she said — more than long enough to produce the quantities of amino acids observed in meteorites.
Sandford notes that in experiments he and other researchers have conducted, irradiation of icy mixtures like those in the primordial interstellar molecular clouds can give rise to thousands of compounds relevant to life, including sugars and nucleobases, “and amino acids are virtually always there in the mix. So the universe seems to be kind of hard-wired to make amino acids.”
Vinogradoff echoed that view and said that the diversity of organic compounds that can be present in meteorites is now known to be vast. “The question has pivoted more to be: Why are these molecules the ones that have proved important for life on Earth?” she said. Why, for example, does terrestrial life use only 20 of the scores of amino acids that can be produced — and why does it almost exclusively use the “left-handed” structures of those molecules when the mirror-image “right-handed” structures naturally form in equal abundance? Those may be the mysteries that dominate chemical studies of life’s earliest origins in the future.
Correction: January 5, 2022
The impact site of the Murchison meteorite was originally described as western Australia. The meteorite landed in the southeastern state of Victoria near the village of Murchison.



