New Brain Maps Can Predict Behaviors
Introduction
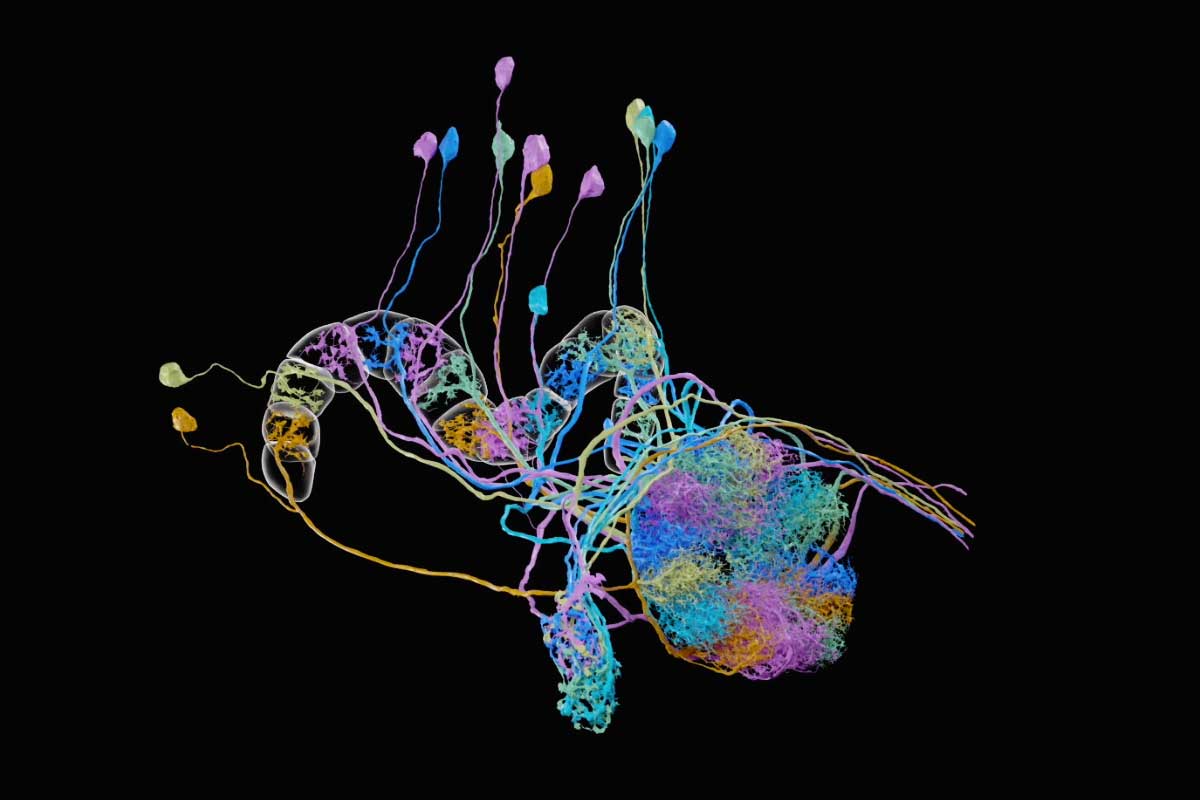
Last summer a group of Harvard University neuroscientists and Google engineers released the first wiring diagram of a piece of the human brain. The tissue, about the size of a pinhead, had been preserved, stained with heavy metals, cut into 5,000 slices and imaged under an electron microscope. This cubic millimeter of tissue accounts for only one-millionth of the entire human brain. Yet the vast trove of data depicting it comprises 1.4 petabytes’ worth of brightly colored microscopy images of nerve cells, blood vessels and more.
“It is like discovering a new continent,” said Jeff Lichtman of Harvard, the senior author of the paper that presented these results. He described a menagerie of puzzling features that his team had already spotted in the human tissue, including new types of cells never seen in other animals, such as neurons with axons that curl up and spiral atop each other and neurons with two axons instead of one. These findings just scratched the surface: To search the sample completely, he said, would be a task akin to driving every road in North America.
Lichtman has spent his career creating and contemplating these kinds of neural wiring diagrams, or connectomes — comprehensive maps of all the neural connections within a part or the entirety of a living brain. Because a connectome underpins all the neural activity associated with a volume of brain matter, it is a key to understanding how its host thinks, feels, moves, remembers, perceives, and much more.
Don’t expect a complete wiring diagram for a human brain anytime soon, however, because it’s technically infeasible: Lichtman points out that the zettabyte of data involved would be equivalent to a significant chunk of the entire world’s stored content today. In fact, the only species for which there is yet a comprehensive connectome is Caenorhabditis elegans, the humble roundworm.
Nevertheless, the masses of connectome data that scientists have amassed from worms, flies, mice and humans are already having a potent effect on neuroscience. And because techniques for mapping brains are getting faster, Lichtman and other researchers are excited that large-scale connectomics — mapping and comparing the brains of many individuals of a species — is finally becoming a reality.

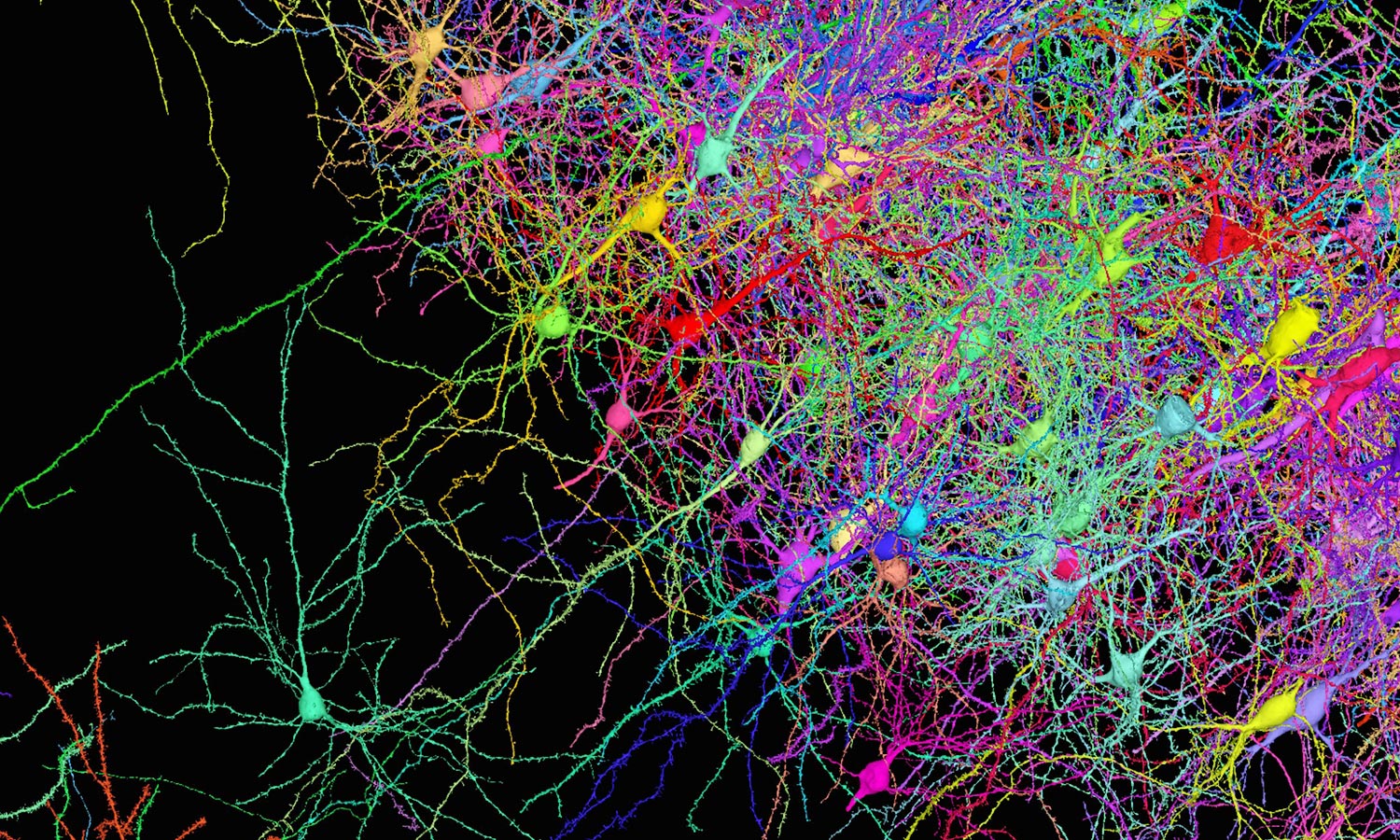
This pair of images is drawn from the connectome that Harvard University and Google researchers compiled from just one cubic millimeter of human cerebral cortex. At 1.4 petabytes, it is the most detailed and complex connectome ever created.
Google/Lichtman Laboratory
Biological individuals typically vary so much in so many ways that scientists need to examine assortments of them to draw conclusions. Large-scale connectomics could give neuroscience the same kind of huge boost that fast, simple genome sequencing brought to genomics.
Recent work with C. elegans has demonstrated the power of large-scale connectomics. One experiment showed that it’s sometimes possible for scientists to predict the behavior of an animal from a knowledge of its connectome; another hinted at rules governing the linkage of neurons into working circuits. Those successes, however, also underscore how far large-scale connectomics still needs to go before it can tackle much more complex creatures. Researchers are also cautious about built-in limitations to what connectomics can tell us.
Predicting Behaviors From Connections
About 35 years ago, the first full-brain wiring diagram was completed for the roundworm. For its time, the effort was heroic, even though the animal has only 302 neurons in its brain. It was carried out through the painstaking process of hand-drawing neuronal connections on printouts of electron microscopy images. It took more than 15 years to complete.
Today, advances in imaging technology augmented by artificial intelligence make it possible for scientists to complete a single C. elegans brain in about a month. Neuroscientists can map several worms for a single experiment to make comparisons between individuals with different traits or in different stages of life. They overlay functional imaging on the maps to learn how the network of connections fires during complex behaviors.
This approach is leading to impressive progress in understanding these animals. In a report in Cell published in September, scientists used the worm connectome to describe one of the most complex behaviors in the natural world: sex. Using video and calcium imaging — which measures and traces activity in brain cells — they recorded C. elegans during the act of mating. Videos showed the worms slithering around one another in serpentine patterns while white light from fluorescent proteins indicating neuronal activity flickered on and off along the length of their slender bodies.
Scientists broke down the complex mating behaviors into subcategories such as partner searching, copulation and rest. Then they mapped the neuronal activities onto the worm connectome to identify the brain mechanisms that process information from the environment during mating. The resulting map of brain activity was so pronounced and consistent between the eight worms used in the study that they could use it to predict the behaviors of a ninth worm, explained Vladislav Susoy, a neuroscientist and first author of the study.
So the scientists decided to put their understanding to the test experimentally. Taking a new worm, they precisely eliminated one of the five neurons involved in a movement called “turning,” in which the worm wraps around its mate just prior to copulation. Without that neuron, the worm lost its ability to turn. “It was really striking how clear the link was,” Susoy said.
Florian Engert, a Harvard neuroscientist working on a brain map of the zebra fish, another widely used laboratory species, called the C. elegans paper “a milestone” in the field for its use of the connectome to generate insights into a complex behavior. The connectome is “becoming a critical resource,” he said.
“The way that the entire field wants to use connectomics now is as a tool and a database for investigating how neuronal circuits operate,” said Gregor Schuhknecht, a neuroscientist and a postdoctoral fellow in Engert’s lab.
But in addition to explaining the underpinnings of behaviors, connectomics studies can also reveal subtle details about how those behaviors are wired into brains.
For example, it has been known for some time that in C. elegans, the connections between neurons dramatically reorganize themselves between birth and adulthood. To understand how the brain changes throughout development, in a recent paper in Nature, the labs of Mei Zhen at the Lunenfeld-Tanenbaum Research Institute of Mount Sinai Hospital in Toronto, and Aravinthan D.T. Samuel and Lichtman at Harvard compared the connectomes of eight genetically identical roundworms ranging between larval and adult stages.
The most interesting finding of the study, according to first author Daniel Witvliet, was that even though the worms were genetically identical, as much as 40% of the connections between nerve cells in their brains differed. Moreover, the connections that varied between individuals were weaker than those that were similar. Stronger connections that contained 100 synapses or more were consistent across multiple organisms.
For Witvliet and Lichtman, this finding points to the power of brain-map comparisons in bulk. “You can’t just say, ‘We’ve mapped the worm connectome,’ because each connectome is slightly different,” Witvliet said.
Lichtman says the finding points to the existence of two classes of connections: variable ones and consistent ones. If it turns out that animals make more consistent connections to support neural activity essential for survival, then he thinks that the level of variation in connections could become an important marker for significant features in the connectome.
“If you have multiple connectomes, the functionally important parts may pop out from that sea of randomness,” Lichtman said. He hopes that in the future, connectomics will regularly analyze the brains of multiple individuals, comparing healthy and unhealthy animals, young and old ones, and so on. “I think that’s where things will head once mapping the brain becomes routine.”
Neuroscience’s “n of 1” Problem
Getting large-scale connectomics to be routine, however, will be easier said than done. The field of connectomics still suffers from what Lichtman calls an “n of 1” problem: Even with all the recent improvements in neural mapping technology, diagramming the brain wiring of even one individual, especially in a species more complex than a worm, is not a task to be undertaken lightly.
His colleague Samuel, a co-author with Susoy of the worm mating paper, agrees. “I tend to think of connectomics in most contexts like Mount Everest expeditions,” Samuel said. “You do it once and say you’re done.”
That challenge represents a significant handicap for research, particularly on complex creatures. When Lichtman and his colleagues mapped the snippet of human brain, for example, they had no idea whether the strange things they saw were normal or one-offs due to the unique history and genetic makeup of the person. If they could map equivalent samples from 100 human brains, then they would get some clarity on these unknowns, but at 1.4 petabytes per brain, that is unlikely to happen anytime soon.
Nevertheless, connectomics is making important progress even where it can’t yet be large scale and where only partial connectomes exist. Work in the fruit fly, Drosophila melanogaster, is particularly far along, both in the larva (which has about 10,000 neurons) and in the adult (with about 135,000 neurons). Last year, researchers at the Janelia Research Campus of the Howard Hughes Medical Institute released a synapse-level “hemi-brain” connectome that mapped many important control centers in the fly’s brain. This led to an important announcement in October, when neuroscientists uncovered dozens of new neuron types and circuits that seem to aid in fly navigation. The work was hailed as a major milestone in revealing how flies incorporate sensory information and translate it into action.
There can also be great value in being able to compare the genomes of different species, even if each of their connectomes is essentially a one-off. In July, a collaboration of neuroscientists at the Allen Institute for Brain Research, Princeton University and Baylor College of Medicine released a data set containing the fine structures and connectivity of 200,000 brain cells in the mouse visual neocortex. The project took five years to complete and represents the largest data set in mice to date.
That’s just a start on the whole brain, but even this much means that for the first time, researchers can compare parts of the connectomes of two mammals — mice and humans. When a connectome becomes available for the zebra fish, it will be possible to look at three different vertebrates.
What Connections Can’t Do
The successes of connectomics can be bittersweet. For many years, a central criticism of connectomics has been that it is insufficient to explain how the brain functions. Despite having had a map of the brain of C. elegans for decades, scientists still struggle to draw meaningful conclusions about its neural functions. To Lichtman, parsing the seemingly limitless interconnectivity of more complex brains is a challenge that tests the limits of human and artificial intelligence.
Another limitation of the connectome is that it doesn’t tell us anything about the quality of the connections: whether they are strong or weak. It simply tells us that there is a connection. “If you have 20 connections between two neurons but they’re all extremely weak,” Engert said, “then you need to know that if you want to know how information flows through the brain.”
Neuroscientists have often assumed that if a neuron makes a connection, it must be making the neuron it touches do something. But not every connection is significant, Lichtman points out, because neurons make thousands of connections with other neurons in vast networks full of redundancies and pathways with overlapping functions. This is why someone can suffer a stroke that wipes out thousands of neurons and not lose their memories. “There is an infinite number of ways the brain could do things,” Lichtman said. “And my guess is, almost in every case, the nervous system rarely takes the simplest path because it is not designed to be simple.”
Connectomics also tells us almost nothing about the brain chemicals called neuromodulators, which circulate through the fluid surrounding neurons, unlike the neurotransmitter chemicals released precisely within the synaptic connections between neurons. They represent another way that cells in the brain communicate with one another.
Many neural circuits change their behavior in the presence of a neuromodulator, said Louis Scheffer, a neuroscientist and computational biologist at the Janelia Research Campus. He cites the example of the stomatogastric ganglion, a circuit of just three neurons in lobsters and crabs that controls the rhythmic movements of the muscles in their stomach. Scientists have identified about 20 neuromodulators that make this ganglion change its firing pattern, which makes it difficult to draw concrete conclusions about how much the motor functions are defined purely by the connections between the neurons. Scheffer calls this neural circuit the “poster child” for why a connectome alone can’t explain brain function.
Engert thinks there is widespread agreement that the connectome will not be sufficient for understanding a brain. But the successes to date of the brain wiring maps in explaining behaviors in roundworms and flies, he said, show that the connectome “certainly is going to be very helpful to inform function, and it might even be necessary.”