Toward a Grand Unified Theory of Snowflakes
Introduction
Kenneth Libbrecht is that rare person who, in the middle of winter, gleefully leaves Southern California for a place like Fairbanks, Alaska, where wintertime temperatures rarely rise above freezing. There, he dons a parka and sits in a field with a camera and a piece of foam board, waiting for snow.
Specifically, he seeks the sparkliest, sharpest, most beautiful snow crystals nature can produce. Superior flakes tend to form in the chilliest places, he says, like Fairbanks and snowy upstate New York. The best snow he ever found was in Cochrane, in remote northeastern Ontario, where there is little wind to batter snowflakes as they fall through the sky.
Ensconced in the elements, Libbrecht scans his board with an archaeologist’s patience, looking for perfect snowflakes and other snow crystals. “If there’s a really nice one there, your eye will find it,” he said. “If not, you just brush that away, and you do that for hours.”
Libbrecht is a physicist. His lab at the California Institute of Technology has investigated the internal structure of the sun and developed advanced instruments for gravitational-wave detection. But for 20 years, Libbrecht’s passion has been snow — not only its appearance, but also what makes it look the way it does. “It’s a little embarrassing when stuff falls out of the sky, and it’s like, ‘Why does it look like that? Beats me,’” he said.
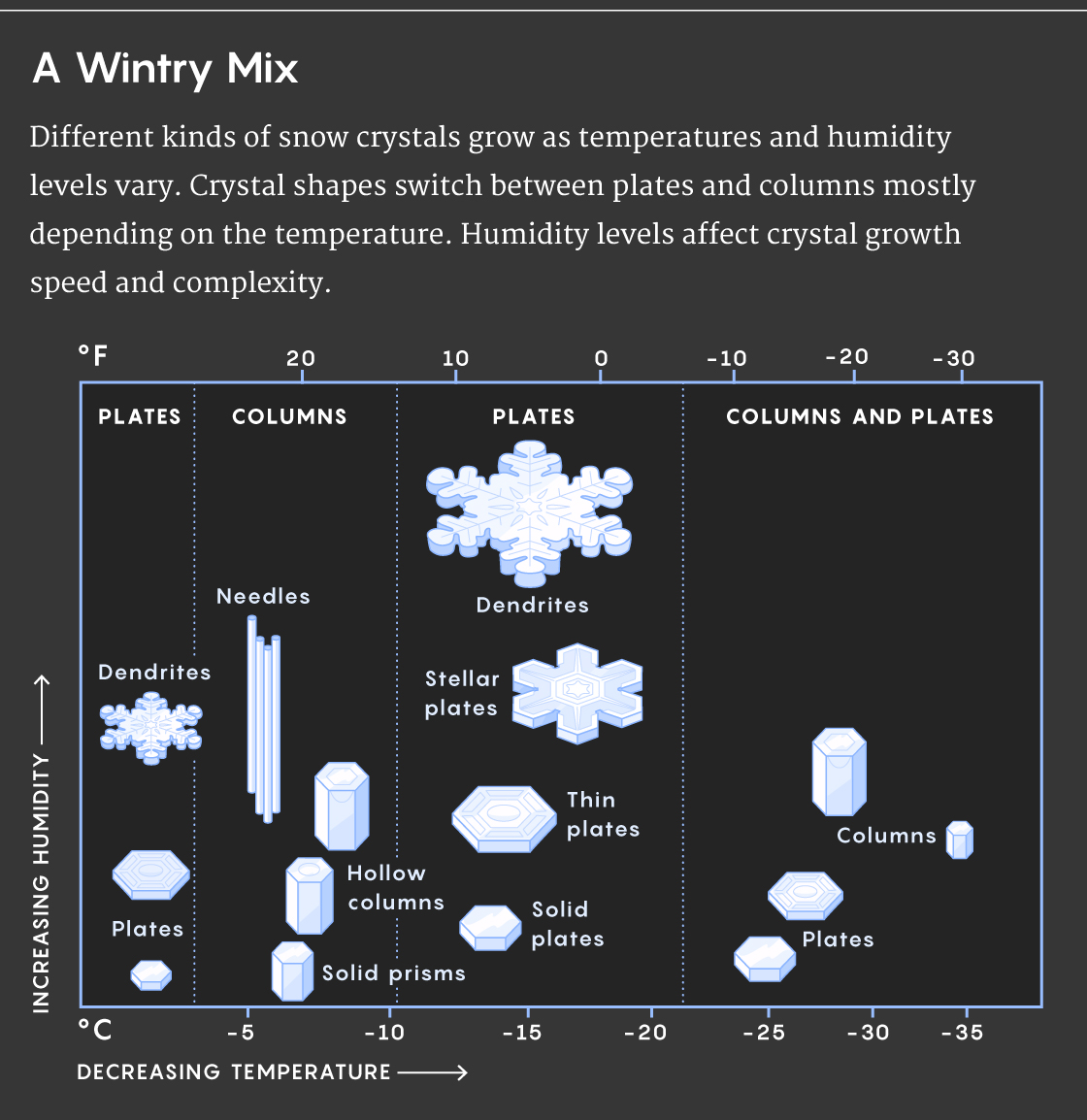
For 75 years, physicists have known that the tiny crystals in snow fit into two prevailing types. One is the iconic flat star, with either six or 12 points, each decorated with matching branches of lace in a dizzying array of possibility. The other is a column, sometimes sandwiched by flat caps and sometimes resembling a bolt from a hardware store. These different shapes occur at different temperatures and humidities, but the reason for this has been a mystery.
Over the years, Libbrecht’s painstaking observations have yielded insights into the snow crystallization process. “He surely is the pope in the domain,” said Gilles Demange, a materials scientist at the University of Rouen in France who also studies snow crystals.
Now, Libbrecht’s work on snow has crystallized in a new model that attempts to explain why snowflakes and other snow crystals form the way they do. His model, detailed in a paper that he posted online in October, describes the dance of water molecules near the freezing point and how the particular movements of those molecules may account for the panoply of crystals that form under different conditions. In a separate, 540-page monograph, Libbrecht describes the full body of knowledge about snow crystals. Douglas Natelson, a condensed matter physicist at Rice University, called the new monograph “a tour de force.”
“As a piece of work,” Natelson said, “boy, it’s gorgeous.”
Six-Cornered Starlets
Everyone knows no two snowflakes are alike, a fact that stems from the way the crystals cook up in the sky. Snow is a cluster of ice crystals that form in the atmosphere and retain their shape as they collectively fall upon Earth. They form when the atmosphere is cold enough to prevent them from fusing or melting and becoming sleet or rain.
Although a cloud contains multitudes of temperatures and humidity levels, these variables are as good as constant across a single snowflake. This is why snowflake growth is often symmetrical. On the other hand, every snowflake is buffeted by changing winds, sunlight and other variables, notes Mary Jane Shultz, a chemist at Tufts University who published a recent essay on snowflake physics. As each crystal submits to the chaos of a cloud, they all take on slightly different forms, she explains.
Lucy Reading-Ikkanda/Quanta Magazine, adapted from Kenneth Libbrecht
The earliest recorded musings on these delicate shapes date to 135 B.C. in China, according to Libbrecht’s research. “Flowers of plants and trees are generally five-pointed, but those of snow, which are called ying, are always six-pointed,” wrote the scholar Han Yin. But the first scientist to try to understand why this happens was probably Johannes Kepler, the German scientist and polymath.
In 1611, Kepler offered a New Year’s gift to his patron, the Holy Roman Emperor Rudolf II: an essay called “The Six-Cornered Snowflake.” Kepler writes that he noticed a snowflake on his lapel as he crossed Prague’s Charles Bridge and could not help but muse on its geometry. “There must be a cause why snow has the shape of a six-cornered starlet. It cannot be chance,” he wrote.
He would have recalled a letter from his contemporary Thomas Harriot, an English scientist and astronomer who, among many roles, served as a navigator for the explorer Sir Walter Raleigh. Around 1584, Harriot sought the most efficient way to stack cannonballs on Raleigh’s ship decks. Hexagonal patterns seemed the best way to pack spheres closely together, Harriot found, and he corresponded about it with Kepler. Kepler wondered if something similar was taking place in snowflakes, and whether their six sides could be pinned on the arrangement of “the smallest natural unit of a liquid like water.”






Micrographs of plate-like snowflakes.
Courtesy of Kenneth Libbrecht
It was a remarkable early insight into atomic physics, one that wouldn’t be formalized for another 300 years. Indeed, molecules of water, with their two hydrogens and one oxygen, tend to lock together to form hexagonal arrays. Kepler and his contemporaries could not have known how much this matters. “Because of the hydrogen bonding, and the details of how the molecules interact with each other, you have this comparatively open crystal structure,” said Natelson. Aside from helping grow snowflakes, this hexagonal structure makes ice less dense than liquid water, which hugely affects geochemistry, geophysics and climate. According to Natelson, if ice did not float, “life on Earth would not be possible.”
After Kepler’s treatise, snowflake observation remained a hobby more than a science. In the 1880s, an American photographer named Wilson Bentley — from the cold, quality-snow-producing village of Jericho, Vermont — began making the first snow crystal images using photographic plates. He produced more than 5,000 images before eventually succumbing to pneumonia.

Drawings of diverse snowflakes by the Japanese physicist Ukichiro Nakaya, who conducted a decades-long study of the different types.
Ukichiro Nakaya
Then, in the 1930s, the Japanese researcher Ukichiro Nakaya began a systematic study of the different snow crystal types. By midcentury, Nakaya was producing snowflakes in a lab, using individual rabbit hairs to suspend frost crystals in refrigerated air where they could grow into full-fledged snowflakes. He tinkered with humidity and temperature settings to grow the two main crystal types and assembled his seminal catalog of possible shapes. Nakaya found that stars tend to form at −2 degrees Celsius and −15 C. The columns form at −5 C and again at about −30 C. In low humidity, stars form few branches and resemble hexagonal plates, but in high humidity, the stars grow more intricate, lacy designs.
According to Libbrecht, the reason for the various crystal shapes also began to come into focus after Nakaya’s pioneering work. Crystals grow into flat stars and plates (rather than three-dimensional structures) when the edges grow outward quickly while the faces grow upward slowly. Slender columns grow in a different way, with fast-growing faces and slower-growing edges.
But the underlying atomic processes that dictate whether snow crystals will be shaped like stars or columns remained opaque. “What changes with temperature?” Libbrecht said. “I’ve been trying to piece all that together.”
Snowflake Recipe
Libbrecht and the very small cadre of researchers who study this problem have been trying to come up with a snowflake recipe, as it were — a set of equations and parameters that can be fed into a supercomputer that would then spit out the splendid variety of snowflakes we actually see.
Libbrecht took up the pursuit two decades ago after learning of the exotic snowflake form called a capped column. It looks like an empty spool, or two wheels and an axle. As a North Dakota native, he was shocked, wondering, “How had I never seen one of these?” Fascinated with the endless forms of snow, he set about understanding their nature for a popular science book he later published, and he started taking pictures, too. Soon, he was tinkering with snowflake-growing equipment in his lab. His new model is the result of observations made over decades that he says recently began to gel.
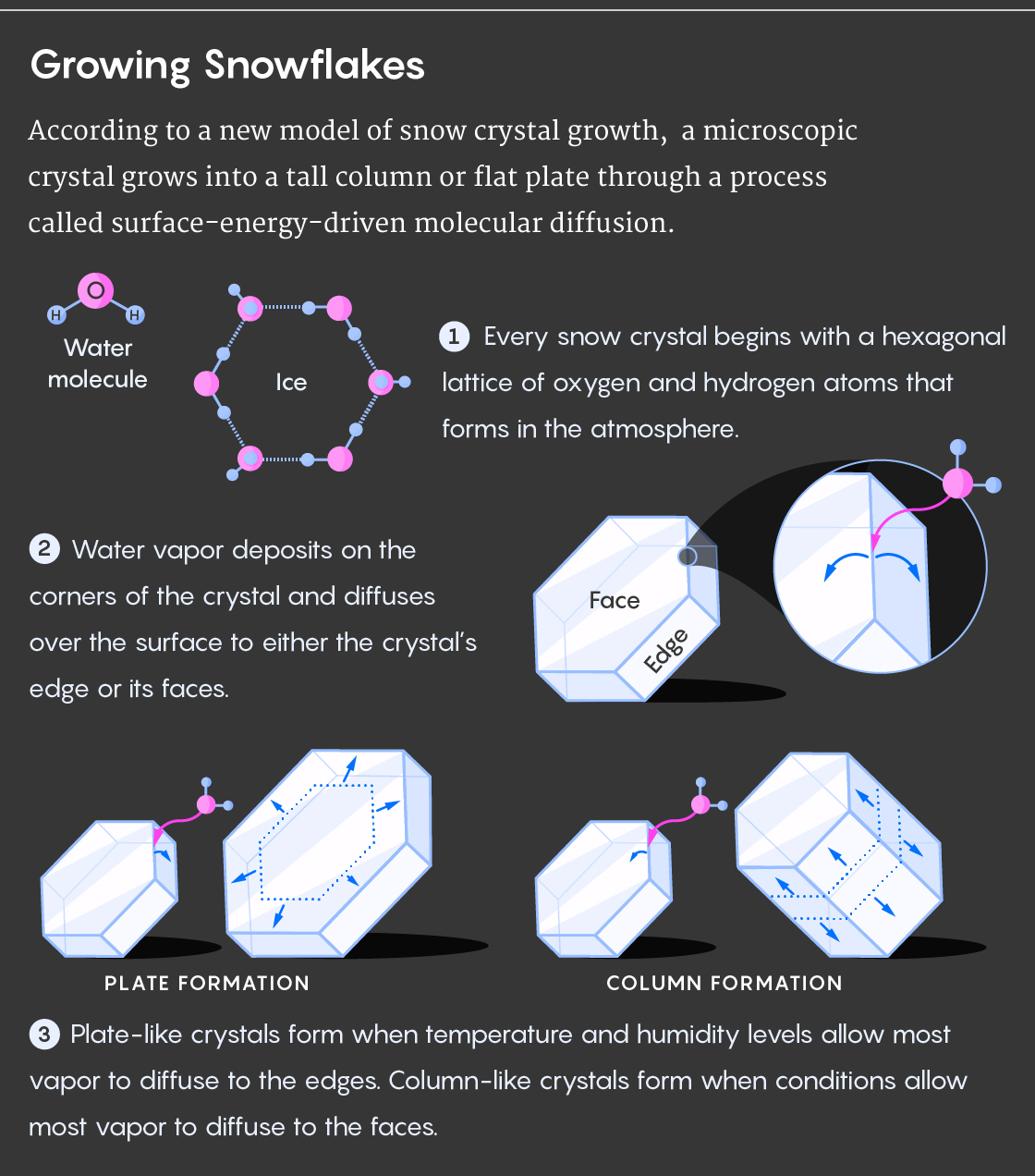
His key breakthrough was an idea called surface-energy-driven molecular diffusion, which describes how a snow crystal’s growth depends on the initial conditions and behavior of the molecules that form it.
Lucy Reading-Ikkanda/Quanta Magazine
Imagine molecules of water arranged loosely, as water vapor just begins to freeze. If you were somehow viewing this from a tiny observatory, you would see the freezing water molecules begin to form a rigid lattice, with each oxygen atom surrounded by four hydrogen atoms. These crystals grow by incorporating water molecules from the surrounding air into their pattern. They can grow in two main directions: up or out.
A thin, flat crystal (either plate-like or starlike) forms when the edges rope in material more quickly than the crystal’s two faces. The burgeoning crystal will spread outward. However, when its faces grow faster than its edges, the crystal grows taller, forming a needle, hollow column or rod.
According to Libbrecht’s model, water vapor first settles on the corners of the crystal, then diffuses over the surface either to the crystal’s edge or to its faces, causing the crystal to grow outward or upward, respectively. Which of these processes wins as various surface effects and instabilities interact depends mostly on temperature.
All this happens only in ice, an unusual mineral, because of a phenomenon called “pre-melting.” Because water ice is usually found close to its melting point, the top few layers are liquid-like and disordered. Pre-melting occurs differently on the faces and edges as a function of temperature, though the details of this are not completely understood. “This is the part of the model where I just make it up whole cloth,” Libbrecht said — though he says the overall physical picture seems plausible.






Examples of column-like snowflakes.
Courtesy of Kenneth Libbrecht
His new model is “semi-empirical,” partly tuned to match observations rather than explaining snowflake growth starting entirely from first principles. The instabilities and the interactions among countless molecules are too complicated to unravel entirely. But he hopes his ideas will form the foundation of a comprehensive model of ice growth dynamics that can be fleshed out by more detailed measurements and experiments.
Although ice is especially weird, similar questions arise in condensed matter physics more generally. Drug molecules, semiconductor chips for computers, solar cells and countless other applications rely on high-quality crystals, and entire groups of researchers focus on the basics of crystal growth.
Meenesh Singh is one such researcher, at the University of Illinois, Chicago. In a recent paper, Singh and a coauthor identified a new mechanism that might underlie crystal growth in solvents, as opposed to the phase-change crystallization of Libbrecht’s snow and ice. In solvent crystallization, solid materials are dissolved in a solution like water or another liquid. By tweaking the temperature and adding other solvents, manufacturers can crystallize new drug molecules or produce new crystals for solar cells, and so on.
“All the applications regarding crystal growth are dealt with empirically,” Singh said. “You have certain empirical data, and using that information, you try to explain how a crystal would grow.” But it’s not clear, he said, how a molecule in the solution integrates into a crystal. “What really drives a molecule to do that? Why would I go to a crystal? If you start wondering, it creates a lot of questions, and those questions are not addressed.”
Libbrecht believes better experiments and more sophisticated computer simulations will answer many questions about crystal growth in the coming years. “Someday, you will be able to make a whole molecular model right down to the atom and see these phenomena going on, right down to quantum mechanics,” he said.
While he tries to unravel the physics, he still enjoys snow crystal photography and the travel that comes with it. But lately, he has stayed put in sunny Southern California, where he has rigged up a sophisticated system for growing snowflakes in his lab. At 61, he’s nearing retirement, which means, he said, “I am throwing away the shackles of my other jobs. I’m just going to do ice from now on.”
This article was reprinted on Wired.com.



