Unexpected ‘Germline’ Plant Cells May Shield New Generations
Introduction
Karel Říha’s mutant plants were too healthy. The molecular biologist, a postdoctoral researcher in Texas in the year 2000, was breeding botany’s leading model organism, Arabidopsis, an unremarkable weed in the mustard family. He had carefully chosen a strain with a mutation that robbed the plants of their ability to repair the caps at the end of their DNA. With every cell division, these protective telomeres grew shorter, hastening the plants’ inevitable genetic meltdown.
But two months became six, and successive generations of Arabidopsis continued to grow like the weeds they were. “It took me over a year to get the first signs of problems with telomeres,” said Říha, who now runs a lab at CEITEC (the Central European Institute of Technology) at Masaryk University in the Czech Republic. “This was the first sign there was something strange going on.”
The mutants were aging too slowly, and a decade would pass before he could figure out why. In recent years, Říha and other biologists have scrutinized how plants grow at the cellular level; some of the important details are unexpectedly murky. Even though the plants appear to make flowers from normal tissue, they may somehow be blocking mutations in body cells from getting into future generations — a strategy typically associated with animals. If it pans out, the controversial finding could overturn a century of conventional wisdom about how the plant kingdom evolves, lives and dies.
Cells accumulate genetic damage throughout their lives from environmental wear and tear, and especially from mishaps that occur while DNA is being copied for cell division. During the 19th century, even before DNA was recognized as the material of heredity, the German evolutionary biologist August Weismann theorized that organisms might protect their genetic heritage by establishing a “germline” tissue, kept isolated from exposure and replication. Today, biologists know that most animals earmark the cells they’ll someday use for procreation early, squirreling them away for safekeeping in what will become the reproductive organs. Decades of sunlight might degrade your skin’s DNA, but since you don’t make babies with your elbow, that damage goes with you to the grave.
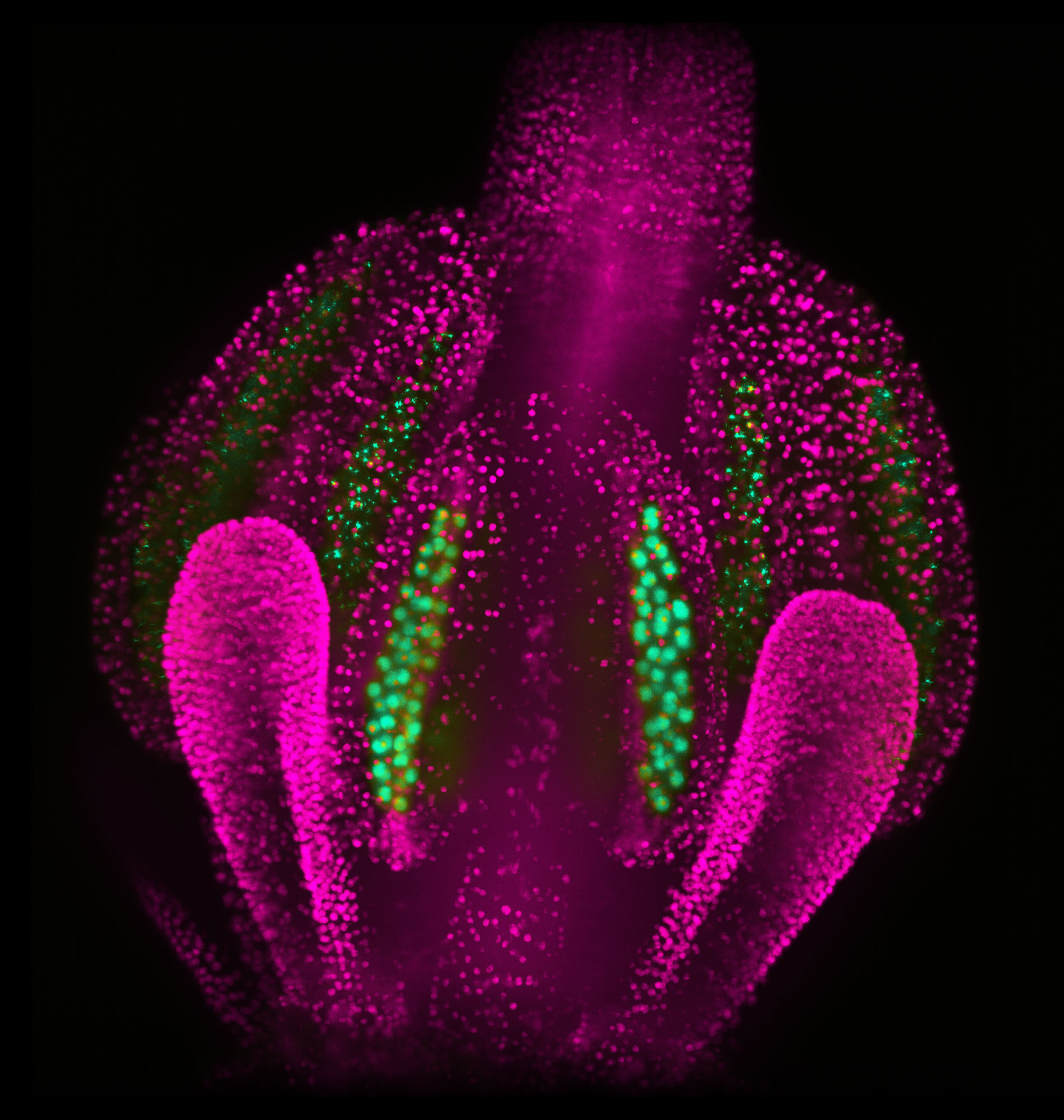
In this micrograph of an Arabidopsis flower, the nuclei of cells making up the pistils (seed-making female organs) and anthers (pollen-making male organs) appear magenta. The nuclei of male germline cells at different stages of development appear green.
Courtesy Říha Lab
Plants, however, are different. Reproductive structures, such as flowers or cones, can sprout from just about anywhere on a plant, which led botanists to conclude that plants don’t produce egg analogues until late in the game, just before reproducing. The cells eventually earmarked for reproduction are presumably drawn from standard body tissues, which means that any mutations arising in those cell lineages during the organism’s life could pass into the next generation, according to this thinking.
To some biologists, plants’ apparent lack of a germline seems like an advantageous feature that lets plant populations test out many mutations to find the ones that best match their changing environments. To others, however, it’s a puzzling bug that clashes with the observed longevity of many trees. If all the mutations in ancient, towering trees can live on, the trees should have trouble producing generation after generation of healthy seedlings. Now, a handful of researchers seek to shift the debate back to its foundational assumption: What if plants do have a mutation-throttling germline after all?
A few years ago, Robert Lanfear, who studies molecular evolution at the Australian National University, was mulling over the surprisingly low number of mutations that had turned up in the body cells of eucalyptus plants during a genomic analysis. Wondering how the plants might be keeping mutations in check, he decided to track down the origins of the no-germline idea. When he found that the supporting evidence was thin, he summarized his findings in a 2018 article for PLOS Biology.
“This took me at least 18 months of reading hundreds of papers, some dating all the way back to the 1920s,” he said. “I do think we were standing on shakier ground than we realized.”
One particular pair of results from 2016 spurred Lanfear to write his paper. If branches, like flowers, can grow from any cell, then the number of cell divisions between two points on a plant should depend on the amount of material between them. However, by painstakingly tracking the microscopic splitting of cells, a team from the University of Bern in Switzerland found that Arabidopsis and tomato branches, at least, are a bit pickier.
All plant cells ultimately derive from a crown of hundreds of cells sitting at the head of the plant’s main stalk — the apical meristem. Botanists have known for decades that slowly dividing stem cells in the center drive growth by producing daughter cells, which replicate faster on the periphery, pushing the meristem higher and eventually becoming leaves, branches and flowers. But it was unclear exactly how plants choose which cells should start these organs.
The Bern group’s tracing efforts suggested that new branches sprout directly from the stem cells, not from specialized leaf or branch cells. Just seven to nine cell divisions separated one branch from the next, regardless of how long the branch was or how big the plant grew. At that rate, the central stem cells at the tip of the furthest branch would have divided just dozens of times over the plant’s entire life span, a tiny figure compared with the organism’s trillions of cells. “That’s pretty weird,” Lanfear said. “The number is a lot lower than anyone thought.”
Months later, Říha’s Arabidopsis results finally came to light, doing for life span what the tomato research had done for growth. Skeptical reviewers had rejected the article in 2011, but the winds shifted after the Bern research. Following up on Říha’s Texas observations, his team had raised Arabidopsis mutants under normal and extended-light conditions and measured the telomere deterioration from one generation to the next as a proxy for how many times the plants had copied their DNA during their lifetime. Despite living three times longer than extended-light individuals, the normal-light Arabidopsis passed on DNA with telomeres only about 15% shorter — a much smaller reduction than the difference in their longevity had implied.
Říha finally understood why his plants in Texas had thrived for so many generations: Whatever cell lineage was shepherding Arabidopsis’s reproductive DNA, it was dividing at a glacial pace that had little to do with size or life span. In light of these results and those of the Bern group, the slowly dividing stem cells in the apical meristem seemed like prime candidates for the lineage in question. Říha now suspects that the organization of the apical meristem, with a few cells at the top dividing only when absolutely necessary while daughter cells below account for the lion’s share of the plant’s growth, may have evolved to safeguard reproductive DNA.
The strategy isn’t exactly the same as sealing off eggs in ovaries, as animals do, but keeping stem cells relatively dormant in the apical meristem might bring the same benefit — while body cells divide like crazy, accumulating errors, a few quiet stem cells keep their DNA clean for eventual reproduction. “It’s not like there is a strict segregation, which would put aside the germline very early and keep it aside,” Říha said. “It’s more like a pool of cells that are protected or shielded from extensive replication.”
Some biologists are starting to consider this tranquil pool of cells a “functional germline,” in a twist on a mid-20th-century French theory called the méristème d’attente, or “waiting meristem,” which suggested that plant embryos sequester germ cells early like many animals, and that the cells remain perfectly silent in the meristem until flowering. Botanists eventually discredited the theory by proving that all embryonic cells do divide as a plant grows, but Lanfear thinks the rejection went too far. There’s a lot of room, he says, between the extremes of setting germ cells aside immediately so they never divide again, and frenetic division all the way to reproduction. What’s more, he says, germline handling and timing vary from animal to animal, and expecting that plants all do it the same way would be naive.
But does the functional germline actually correlate with depressed mutation rates? Recent studies suggest it might. Trees send out branches as they grow taller, so the lower branches serve as snapshots of the plant’s genome when it was young. And affordable gene sequencing techniques are letting researchers assemble slideshows that reveal how DNA changes as trees age.
Struck by the majesty of an old oak on the University of Lausanne’s campus in Switzerland, Philippe Reymond, a molecular biologist, wondered how many mutations its upper reaches had accumulated after more than 200 years of growth under the sun. Sequencing the DNA of leaves from the oak’s bottom and top, his group used algorithms to search the genetic codes for typos — an effort he compares to copyediting two stacks of 100 Oxford English Dictionaries. In the end, they estimated that the uppermost leaves had accumulated no more than three dozen mutations — only one-tenth of what Reymond had expected based on the Arabidopsis research.
A functional germline with both genetic and physical shielding could explain how an ancient tree keeps itself so healthy genetically. “There must be a genetic control on why a few cells don’t divide — a strict genetic program,” Reymond said. “These layers of leaves could also be protection against UV light” for the stem cells beneath them.
Arabidopsis, tomatoes and oaks all belong to the flowering-plant group called angiosperms, but the first mutation rate estimated for individual conifer trees, published in June, tells a similar story. A team from the University of British Columbia (UBC) had professional tree climbers collect needles and bits of trunk from 200- to 500-year-old Sitka spruce trees. Sequencing select genome sections, they found a per-year mutation rate lower than that of many animals. Assuming that spruces botch DNA replication about as often as Arabidopsis does, the group concluded that the trees’ stem cells may lie quiet for years between divisions. “That is akin to a germline,” said Sally Aitken, a co-author and a professor of forestry at UBC.


A forestry team from the University of British Columbia, led by the researcher Sally Aitken, collects samples from Sitka spruce trees in Carmanah Walbran Provincial Park to study mutation rates in different parts of the plants.
Some, however, have reservations. Sarah Otto, an evolutionary biologist at UBC who also worked on the conifer study, points out that stem cells may have evolved to divide slowly for reasons unrelated to controlling mutations. A shoot apical meristem that remains small and quiet, for instance, might be the only type that stays balanced above the comparatively explosive growth of the plant below. To her, a more in-depth molecular analysis of the stem cells would be more convincing proof.
Laurence Hurst, an evolutionary geneticist at the Milner Center for Evolution at the University of Bath, also counsels caution because it’s hard to compare mutation rates across groups with different populations, life spans and sizes. To get to the root of mutations within individuals, he recently co-authored a paper, published in April in PLOS Biology, comparing hundreds of genomes taken from different parts of plants from the same eight species. “We’re trying to strip away all those variables,” he said. “It’s all from the same damn plant.”
Hurst finds Lanfear’s hypothesis intriguing, however, in part because one of the experiments in Hurst’s recent study goes a step beyond the functional germline idea, revealing the most suggestive evidence yet for an honest-to-goodness, animal-style separated germline.
Strawberries reproduce by putting out runners that grow into new plants when they hit the ground. Since these runners carry the DNA for future strawberry plants, germline theory would insist that the plant must keep runner mutations low. But these runners had twice the mutations of the reproductively inconsequential leaves. “It really had us scratching our heads,” Hurst said.
After carefully tracing the five mutations, however, the team found that only one made it into each of the offspring plants, and always the same one. The odds of each consecutive plant randomly drawing the same mutation were less than 1 in 1,000, Hurst calculated, suggesting another interpretation.
“What we’ve got is the most beautiful cell line tracer by accident,” he said. “This [mutation] must be sitting in something like a germline.” If strawberries physically separate the cells in their runners, dedicating molecular resources to keeping the mutation rate low in the cells destined for reproduction, they could ignore the rest of the runner without many ill effects for their progeny.
“I think this is the best evidence that anyone’s ever come up with that some plants really do have a germline,” Hurst said. Next, he plans to get some runner samples under a microscope and look for physical differences.
Lanfear finds such results exciting but stresses that they are preliminary. At this point, most experiments involve small groups of plants and were not explicitly designed to search for plant germlines. Numerous technical caveats also complicate interpretation of the genetic results. “None of this evidence is totally bulletproof,” he said.
Putting the functional germline hypothesis on firmer ground calls for studying how the apical meristem divides, measuring mutation rates in more species, and tracking how many mutations make it into the next generation. Proving a separated germline will be even tougher. Ideally, researchers would trace a plant’s entire cell lineage and look for one meristem population that leads only to flowers, but plants are long lived, opaque and developmentally plastic, and the meristem’s leaf covering hides it from scientists as well as ultraviolet rays.
Despite the challenges, Lanfear thinks upcoming work will go a long way toward resolution. “I wouldn’t be surprised,” he said, “if within the next five years we have some pretty good answers to this question in lots of different species.”
Since germlines represent the genetic gates between generations, understanding to what degree species harness them could illuminate evolutionary trends across swaths of the tree of life. Hurst studies plants to test what he calls the “future shadow effect,” a hypothesis that the more important a stretch of DNA will be to future generations, the more an organism works to keep it safe. “The [sequestered] germline is just a crystallization of that,” he said.
Biologists have long puzzled over why lifetime cancer rates hold somewhat constant across species from mice to elephants, even though the latter has so many more cells to go haywire. Plant cancers don’t metastasize because their cells don’t move, but random mutations cause harm in other ways. It stands to reason, then, that our leafy companions have developed techniques to keep their genetic machinery humming smoothly enough to allow them to grow so large. When it comes to managing their genes, every species seeks a balance between thrifty corner cutting and costly perfectionism. The question now is how much our respective strategies may overlap.
“The more we study plants, the more we find there are either similar strategies or similar findings in humans or animals,” Reymond said. “They’re different solutions but maybe for the same reasons or the same goal.”



