Can Our Brains Be Taken Over?
Myriam Wares for Quanta Magazine
Introduction
In The Last of Us, a video game series and recent television show, fungal pathogens are to blame for a zombie-like plague. Once infected, humans lose control over their bodies and become increasingly aggressive, seeking to infect others through violence. It’s a familiar trope: The same fungus, Ophiocordyceps, torments humanity in the movie The Girl With All the Gifts, while viruses do the work in the film 28 Days Later and the novel World War Z.
But the concept of a pathogen that can manipulate its host’s behavior — against their will and often to their detriment — is not purely the work of fiction. In these zombie-like cases, the pathogen (whether it’s a virus, bacteria or fungus, or something else) acts specifically to change the behavior of its host.
While we know a decent amount about these pathogens — including the very real Ophiocordyceps fungus, which does turn insects into unwitting agents of societal collapse — there’s still much to learn.
So the Cordyceps fungus is real?
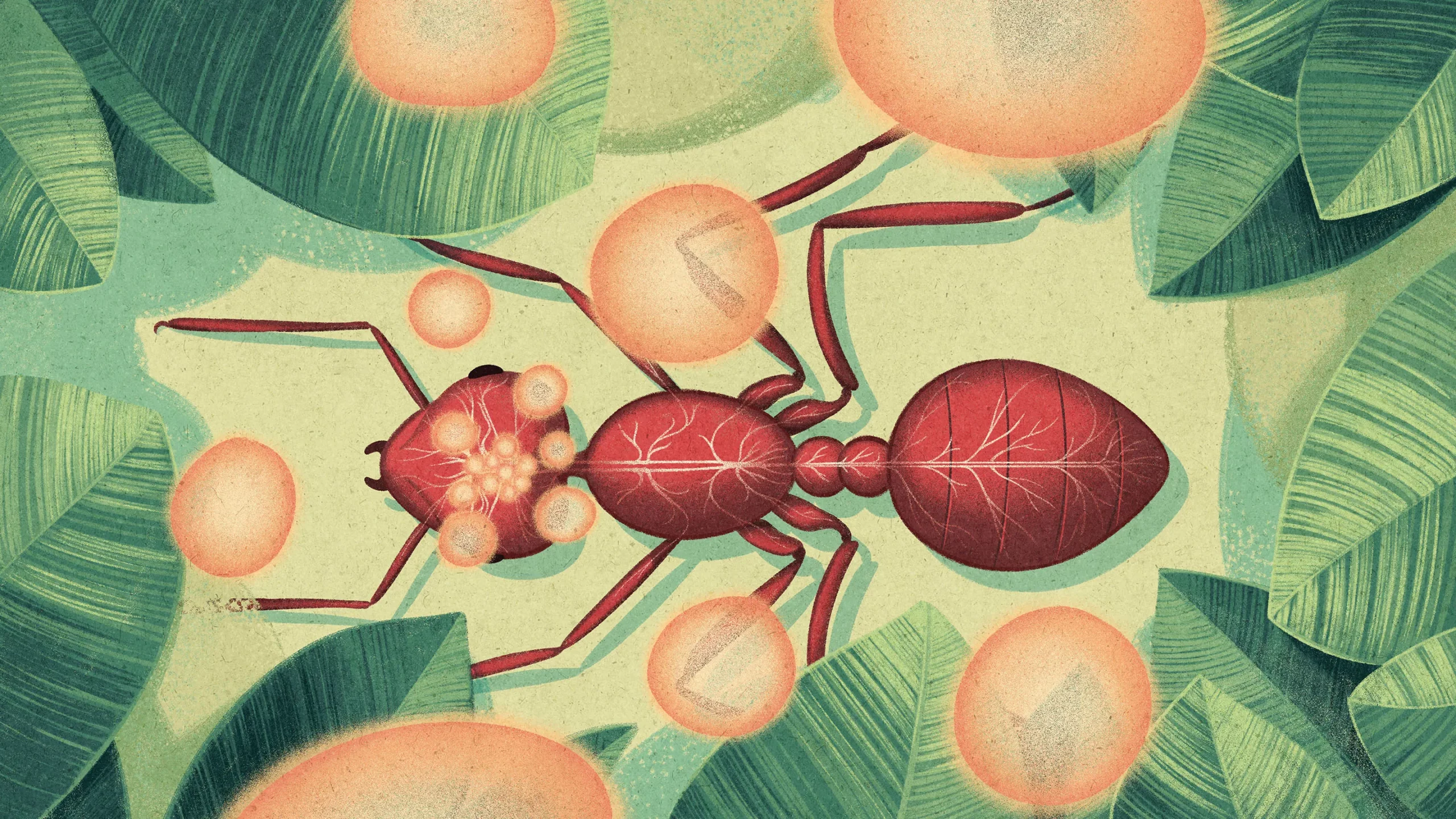
“Cordyceps” has become a common catch-all name for a group of fungi that infect insects. This grouping includes the species Ophiocordyceps unilateralis, better known as the “zombie ant fungus.” It spreads by sprouting fungal structures that erupt through the ant’s head after its death.
The challenge for this reproductive strategy is that ants are social insects, and so they act to protect the colony from infections. As part of this behavior, ants typically remove dead ants from the nest. A lone dead ant outside the nest won’t spread the fungus.
To get around this, the fungus induces an infected ant to leave the nest, climb above it, and bite into nearby vegetation as it is dying — a practice known as summiting. A day or two after the ant’s death, the parasitic fungal spores erupt out of the ant’s head to shower the colony with the pathogen.
Ophiocordyceps-infected ants aren’t the only ones who exhibit this climbing behavior upon infection. It is widespread across different categories of insects and can be caused by viruses, parasites called trematodes, and various species of fungi unrelated to Ophiocordyceps. It even has a common name: summit disease.
How do these infections work?
We don’t know for sure. Recent studies have found chemical signals from the fungi in infected insects. Some of these signals — likely secreted proteins — may target the host’s behavioral systems and control behavior like summiting and nest desertion.
Scientists hypothesize that pathogens may be taking advantage of preexisting behaviors governing molting and sleep, which evolved millions of years ago. Molting — the process by which an insect sheds its protective exoskeleton to grow a new, bigger one — is a time of vulnerability, since it takes a day or two for the new exoskeleton to harden enough to provide safety. Insects have evolved behaviors for this time that can include wandering from the nest and summiting. Sleep is also a vulnerable time, so some insect species have learned to sleep while grasping onto grass or leaves.
Molting and sleep are controlled by the insect’s circadian rhythm, and summiting appears to be as well, as many species of infected insects do this at predictable times. In Ophiocordyceps-infected carpenter ants, for example, summiting always happens around solar noon. The biological machinery associated with an insect’s circadian rhythm therefore seems like a good place to explore.
Could Cordyceps infect humans?
No. Because they are so specialized, individual species of Ophiocordyceps fungi only infect a limited range of insects and do not infect humans at all. (In fact, Cordyceps supplements are sold as a health food, despite a lack of scientific evidence for their supposed health benefits.)
Does that mean we have nothing to worry about from these kinds of infections?
Not quite. We’ve long known that some compounds from fungi can change our behavior, like the hallucinogenic chemical psilocybin from Psilocybe mushrooms and ergot from Claviceps-contaminated grain.
Other fungal infections can be deadly, if not phantasmagoric. Last year the World Health Organization published its first-ever list of fungal “priority pathogens” for humans, noting their health implications and increasing resistance to treatment. The fungal pathogen Candida auris, for example, can cause bloodstream infections that can be fatal in as many as 60% of those infected.
What about non-fungal infections?
Multiple pathogens can change behavior in mammals — and potentially humans. One famous example is the single-celled parasite Toxoplasma gondii, which reproduces in cats and infects about a third of the world’s human population.
The infection that it causes, known as toxoplasmosis, typically isn’t a big deal in humans, producing only mild flu symptoms at the most. But for immunocompromised people it can be devastating, and it can cause serious issues in a developing fetus. This is why pregnant people are supposed to avoid contact with cat litter.
Like Ophiocordyceps, Toxoplasma can also manipulate host behavior. In multiple rodent studies, including one from last year, researchers showed that Toxoplasma-infected rodents no longer fear the scent of cat urine. Instead, they become physically attracted to it. The parasite seems to cause behavior that gets its rodent host eaten by a cat — the parasite’s final host.
For the past 70 years, researchers have investigated the behavioral effects of Toxoplasma infection in humans. Multiple studies have linked it to mood disorders and mental health issues, including schizophrenia. The parasite may also induce more subtle behavioral changes, such as promoting risky behaviors and perhaps even influencing political beliefs.
Again, we don’t know how the parasite does this. Researchers suspect it subtly tweaks the chemicals produced by our cells, which can result in a change in behavior. Studies also suggest that chronic infection with the parasite can affect the amount of neurotransmitters — chemicals that carry signals between neurons — associated with reward and motivation, including dopamine. By manipulating host neurochemicals, the parasite could alter our actions.
What else can potentially change a human’s behavior against their will?
Rabies is a mammalian virus transmitted via infected saliva, generally through biting. In the United States, bats are the leading cause of rabies infections, although raccoons, skunks and foxes also spread the virus. Outside the U.S., rabid dogs are the leading cause of infection in humans, which results in about 60,000 deaths per year globally. Infections make the host aggressive and more prone to bite others — a potentially manipulating behavior that promotes transmission of the virus.
Descriptions of rabies infections date back hundreds of years, but we are still learning about the molecular pathways it uses. The virus is neurotropic, meaning it attacks the nervous system, but it’s still not clear how this leads to specific symptoms. Research suggests that the rabies glycoprotein — a large molecule on the surface of the virus that attaches to host cells — may interact with host proteins that help produce muscular contractions. If the rabies glycoprotein binds to and occupies the receptor, it could disrupt how neurons communicate with other cells. Artificially inhibiting this receptor with a part of the rabies glycoprotein was found to produce rabies-like behavior in mice.
Do we know for sure that pathogens can actively change human behavior?
Infections can change a host’s behavior in several possible ways. Maybe the pathogen coaxes the host to act in a way that increases the likelihood of reproduction, like Ophiocordyceps does for ants. Or maybe the host’s behavioral changes are just a side-effect of feeling sick — something of a coincidence. It’s hard to study this relationship in humans because of ethical concerns raised by clinical trials. Instead, we must use epidemiological studies that compare the behavior of infected individuals with typical behavior. Or we rely on animal studies, which do not always accurately replicate human behaviors.
Even in the absence of studies that test how behavioral changes occur, it’s clear that some loss of control can happen. So while it’s unlikely that we’ll see a zombie-like plague, or a virus that can gain full control over a human’s actions, it’s unwise to underestimate nature. We remain at the mercy of many pathogens, brain control or no.
Correction: February 28, 2023
An earlier version of this article incorrectly described trematodes as “single-celled.”