Cells Form Into ‘Xenobots’ on Their Own
Introduction
Early last year, the biologist Michael Levin and his colleagues offered a glimpse of how versatile living matter can be. Levin and Douglas Blackiston, a member of his laboratory at the Allen Discovery Center of Tufts University, brought together nascent skin and muscle cells from a frog embryo and shaped the multicelled assemblies by hand. This sculpting process was guided by an algorithm developed by the computer scientists Josh Bongard and Sam Kriegman of the University of Vermont, which searched for simulated arrangements of the two cell types capable of organized movement. One design, for example, had two twitching leglike stumps on the bottom for pushing itself along.
The researchers let the cell clusters assemble in the right proportions and then used micro-manipulation tools to move or eliminate cells — essentially poking and carving them into shapes like those recommended by the algorithm. The resulting cell clusters showed the predicted ability to move over a surface in a nonrandom way.
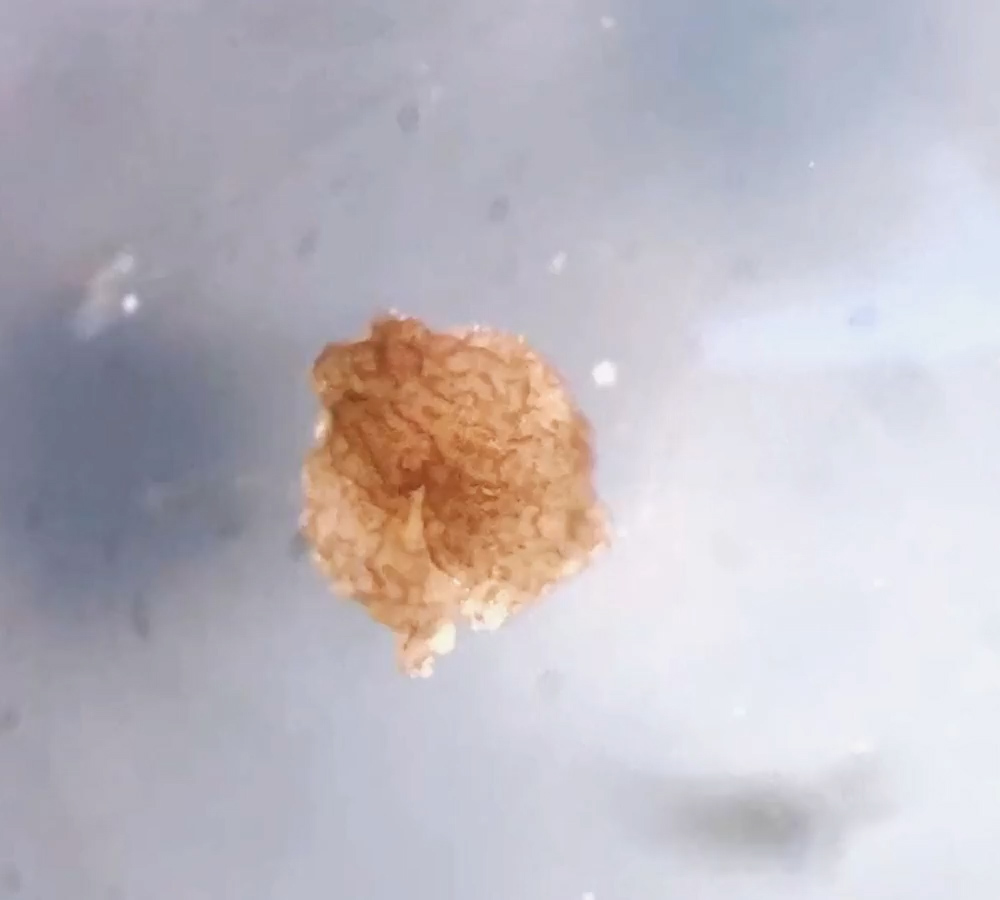
The team dubbed these structures xenobots. While the prefix was derived from the Latin name of the African clawed frogs (Xenopus laevis) that supplied the cells, it also seemed fitting because of its relation to xenos, the ancient Greek for “strange.” These were indeed strange living robots: tiny masterpieces of cell craft fashioned by human design. And they hinted at how cells might be persuaded to develop new collective goals and assume shapes totally unlike those that normally develop from an embryo.
But that only scratched the surface of the problem for Levin, who wanted to know what might happen if embryonic frog cells were “liberated” from the constraints of both an embryonic body and researchers’ manipulations. “If we give them the opportunity to re-envision multicellularity,” Levin said, then his question was, “What is it that they will build?”
Some of those answers are now being unveiled in work appearing today in Science Robotics. It describes a new generation of xenobots — ones that took shape on their own, entirely without human guidance or assistance.
At a glance, these xenobots might be mistaken for other microscopic aquatic animals — amoebas or plankton or Giardia parasites — swimming here and there with apparent agency. Some move in orbit around particles in the water, while others patrol back and forth as though on the lookout for something. Collections of them in a petri dish act like a community, responding to one another’s presence and participating in collective activities.
When he shows movies of these spontaneously grown xenobots to other biologists and asks them to guess what they are, Levin said that “People say, ‘It’s an animal you found in a pond somewhere.’” They are astounded when he reveals that “it’s 100% Xenopus laevis.” These microscopic entities are utterly unlike any stage in the normal development of a frog.
The xenobots are turning some conventional views in developmental biology upside down. They suggest that the frog genome doesn’t uniquely instruct cells about how to proliferate, differentiate and arrange themselves into a frog body. Rather, that’s just one possible outcome of the process that the genomic programming permits.
For the evolutionary biologist Eva Jablonka of Tel Aviv University, who was not involved in the work, xenobots are nothing less than a new type of creature — one “defined by what it does rather than to what it belongs developmentally and evolutionarily.” She suspects the findings might illuminate the very origins of multicellular life.
Levin believes that his cell-bots disclose something profound about how cells and development work. The results seem to imply that individual cells have a kind of decision-making capacity that creates a palette of possible bodies they could build — constrained and guided by the genome but not defined by it. Rules operating above the level of genes appear to specify biological form, and the way we see them embodied in xenobots can tell us something about how they operate. Ricard Solé, a complex systems theorist at Pompeu Fabra University in Spain, said that the new experiments “open a whole new window to interrogate development — and more generally, novel forms of complex life.”
It’s certainly not just about frogs. “If the organization we see in xenobots is the basic state of multicellular animal organization,” said Jablonka, then she anticipates that human cells will behave in the same way. Someday, if we can learn and guide the effect of these rules, Levin thinks, we might be able to achieve things that our cells don’t seem able to manage on their own, such as the regeneration of limbs.
Cells Find Their Own Solutions
The experiments described in the paper published today were remarkably simple. The same team of researchers, along with Emma Lederer of Levin’s lab, removed cells from developing frog embryos that had already specialized into epithelial cells and left them to develop in clusters on their own without the rest of the embryo, which normally provides the signals that guide cells to become the “right” type in the “right” place.
What the cells did first was unremarkable: They gathered into a ball, composed of dozens of cells or a few hundred. That kind of behavior was already well known and reflects the tendency of skin cells to make their surface area as small as possible after tissue damage, which helps wounds to heal.
Then things got weird. Frog skin is generally covered with a protective layer of mucus that keeps it moist; to ensure that the mucus covers the skin evenly, the skin cells have little hairlike protrusions called cilia, which can move and beat. We have them, too, on the lining of our lungs and respiratory tract, where their beating motion helps sweep away dirt in the mucus.
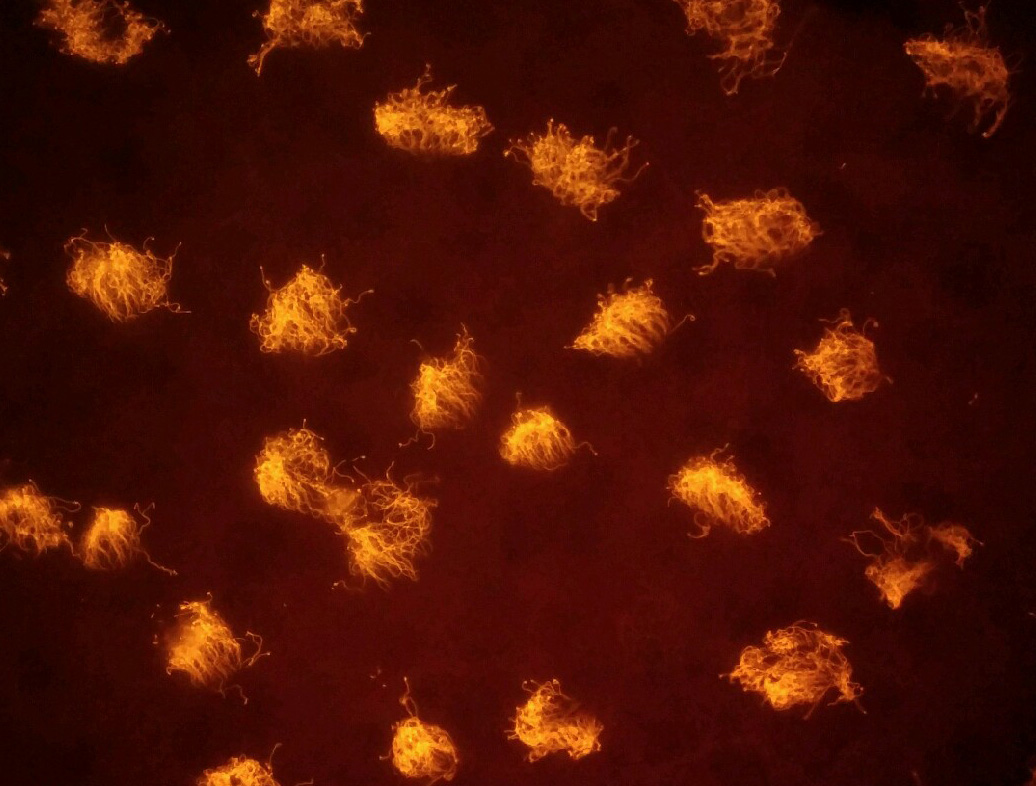
This high magnification image of xenobots reveals the cilia that grow on their surface and enable them to swim through coordinated “rowing.” The cilia of frog epithelial cells normally serve a very different purpose.
Douglas Blackiston
But the frog skin cell clusters quickly began to use their cilia for a different purpose: to swim around by beating in coordinated waves. A midline formed on the cluster, “and the cells on one side row to the left and those on the other side row to the right, and this thing takes off. It starts zooming around,” Levin said
How does the xenobot decide where to draw the midline? And what even “tells” it that doing this would be useful? That’s not yet clear.
But these entities don’t just move; they seem responsive to their environment. “They’ll sometimes go straight, sometimes in circles,” Levin said. “If there’s a particle in the water, they’ll circle it. They will do mazes — they can take corners without bumping into anything.”
He added, “I’m quite certain they do a lot of things we don’t even recognize yet.”
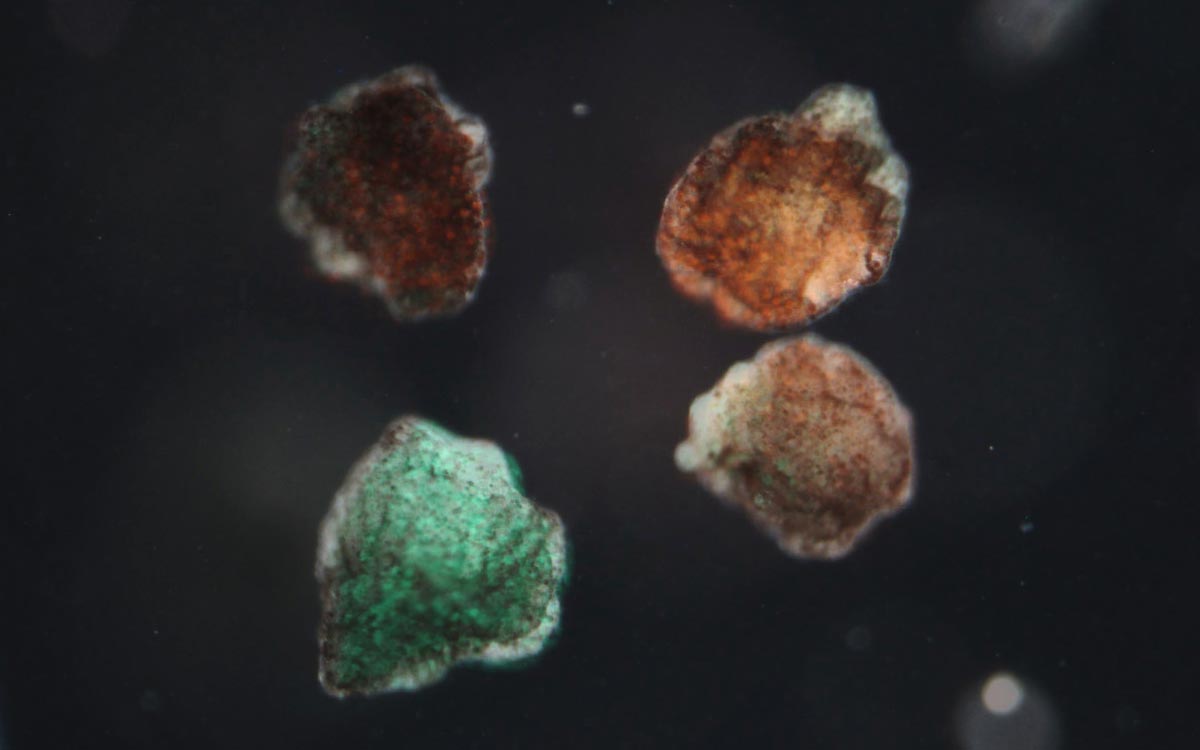
Four of the xenobots that spontaneously formed from snippets of embryonic frog tissue in the newly published experiments.
Douglas Blackiston
Jablonka thinks that most animal developmental biologists won’t be surprised by the outcome of experiments like this — but will kick themselves for not having looked for it. “They would probably say, ‘Yes, of course! Why did we not do this simple experiment before?’” she said. Solé suspects others might have accidently stumbled on similar observations, but “thought it was a mistake, or simply impossible.”
Or it might have just been overlooked — because most developmental research only aims to reveal how whole organisms or parts of them grow under normal or mildly manipulated conditions, Jablonka said. But Levin’s work has a new goal, she says: “Constructing an autonomous creature that has nothing to do with the specific form of the [original] organism.”
Xenobots normally live for about a week, subsisting on the nutrients passed down from the fertilized egg they came from. But in rare cases, by “feeding” them with the right nutrients, Levin’s team has been able to keep xenobots active for more than 90 days. The longer-lived ones don’t stay the same but begin to change, as though they are on a new developmental path — destination unknown. None of their incarnations look anything like a frog as it grows from an embryo to a tadpole.
Channels of Communication
Media reports of the earlier handmade xenobots both reveled in and worried about the idea of miniature robots made from living matter. Might they breed and develop minds of their own? In truth, neither possibility was remotely likely: The cells could survive in a nutrient medium, but they couldn’t replicate into new xenobots. And they didn’t have any nerve cells that might act like a mind.
But even though xenobots have no nervous system, that doesn’t mean the cells can’t communicate with one another. One cell might release a chemical that sticks to surface proteins on another cell, triggering a biochemical process within the recipient. This type of cell signaling happens constantly during embryonic development, and it’s one way that neighboring cells control one another’s fate — the type of tissue each cell ultimately becomes. Adhesive proteins enable cells to attach to one another and to sense mechanical forces and deformations. In developing embryos, mechanical cues like this may also guide to become the right tissue type.
Levin thinks that cells also commonly communicate electrically — that this isn’t just a property of nerve cells, although they may have specialized to make good use of it. In a xenobot, “there’s a network of calcium signaling,” Levin said — an exchange of calcium ions like that seen between neurons. “These skin cells are using the same electrical properties that you would find in the neural network of a brain.”
For example, if three xenobots are set spaced apart in a row, and one of them is activated by being pinched, it will emit a pulse of calcium that, within seconds, shows up in the other two — “a chemical signal that goes through the water saying that someone just got attacked,” Levin said.
He thinks that intercellular communications create a sort of code that imprints a form, and that cells can sometimes decide how to arrange themselves more or less independently of their genes. In other words, the genes provide the hardware, in the form of enzymes and regulatory circuits for controlling their production. But the genetic input doesn’t in itself specify the collective behavior of cell communities.
Instead, Levin thinks that it programs cells with an ensemble of tendencies that produce a repertoire of behaviors. Under the normal conditions of embryogenesis, those behaviors follow a certain path toward forming the organisms we know. But give the cells a very different set of circumstances, and other behaviors and new emergent shapes will appear.
“What the genome provides for the cells is some mechanism that allows them to undertake goal-directed activities,” Levin said — in effect, a drive to adapt and survive.
Innate Drives to Survive
One such goal that Levin and his colleagues think they have seen is known as infotaxis, a push for cells to maximize the amount of information they get from their neighbors. Cells may also seek to minimize “surprise,” the chance of encountering something unexpected. The best way to do that, Levin says, is to surround yourself with copies of yourself. Some other goals are based on pure mechanics and geometry, such as minimizing the surface area of a cluster.
The genomic programs for the pursuit of these goals, he says, are very ancient. Indeed, a reversion to something like ancestral behavior from before cells figured out how to work together may emerge in cancers — where cells adopt a potentially lethal mode of organizing themselves that sets proliferation ahead of cooperation.
If that’s right, then the variety of body shapes and functions in natural organisms is not so much the result of specific developmental programs written into their genomes, but of tweaks to the strengths and tendencies of these single-cell behaviors, which may come from both the genome and the environment.
Jablonka guesses that the behaviors on display in the xenobots are probably “something like the most basic self-organization of a multicellular animal-cell aggregate.” That is, they are what happens when both the constraints on form and the resources and opportunities provided by the environment are minimal. “It tells you something about the physics of biological, developing multicellular systems,” she said: “how sticky animal cells interact.” For that reason, she thinks the work might hold clues to the emergence of multicellularity in evolutionary history.
Solé agrees with that. “One of our dreams in the study of synthetic complexity is to be able to move beyond the actual repertoire of life forms that we can see around us, and to explore alternatives,” he said. The fossil traces of simple animals that began to evolve before the Cambrian era, more than about 540 million years ago, give only the vaguest hints of how multicellularity arose through the interactions of single-celled organisms.
That cells might be programmed to collectively “compute” their own ways solutions to growth and form, rather than for their genome to prescribe them, makes sense in evolutionary terms, because it means that the collective goals of the cells in a tissue remain resilient to disturbance. There’s no need to hard-wire a contingency plan into the genome for every injury or challenge the tissue might face, because the cells will spontaneously revert to the right course. “What you have is organs and tissues that have very specific large-scale goals, and if you try to deviate them off of that, they will come back,” Levin said.
This robustness against disruption seems to be borne out by the fact that the xenobots can regenerate from damage. “Once they’ve developed this new body, they have some ability to maintain it,” Levin said. In one experiment, a xenobot was cut almost in two, its ragged halves opened up like a hinge. Left to itself, the hinge shut again and the two fragments rebuilt the original shape. Such a movement requires substantial force applied at the hinge joint — a situation skin cells would not normally encounter, but which they can apparently adapt to.
Navigating Without a Map
Whether the xenobots really are on a new and distinct developmental path remains unclear at this point. Christoph Adami, a microbiologist at Michigan State University, suggests that the xenobots’ development of cilia, for example, might not reflect some novel “decision” but rather just an automatic response to the mechanical forces acting on the cell clusters. He thinks that more work, perhaps by tracking changes in gene expression, will be required to establish what’s happening.
But Levin said that the idea of cells collectively deciding on and remembering goals is supported by experiments that he and his colleagues conducted previously on Xenopus tadpoles. To become a frog, a tadpole has to rearrange its face; the genome was thought to hard-wire a set of cell movements for every facial feature. “I had doubts about this story,” Levin said, “so we made what we call Picasso tadpoles. By manipulating the electrical signals, we made tadpoles where everything was in the wrong place. It was totally messed up, like Mr. Potato Head.”
And yet from this abstract rearrangement of tadpole features, normal frogs emerged. “During metamorphosis, the organs take unusual paths that they don’t normally take, until they settle in the right place for a normal frog face,” Levin said. It’s as if the developing organism has a target design, a global plan, that it can achieve from any starting configuration. This is far different from the view that cells are “following orders” each step of the way. “There’s some way the system is storing a large-scale map of what it’s supposed to build,” Levin said. That map is not in the genome, however, but in a kind of collective memory of the cells themselves.
If, however, you totally reconfigure the cells, it seems you can change the map. The next step is to work out what the rules are that create the new map — so that we can control it and build what we want. “We know very little about the plasticity of developmental programs,” Adami said. “Our thinking has been shaped by a few well-studied organisms and genes, like worms, flies and sea urchins. But there is likely an iceberg of ancient potential pathways under every tip.”
Fundamentally, Levin says, no one yet know what factors specifically induce cells to multiply and spread in a flat layer, gather into a dense mass, make an organlike structure … or grow into a mobile “bot.” The challenge now is to discover the rules and to learn how to apply them for desired outcomes. “We need to learn how cells themselves encode whatever pattern they’re supposed to build, and then to rewrite that target morphology,” he said.
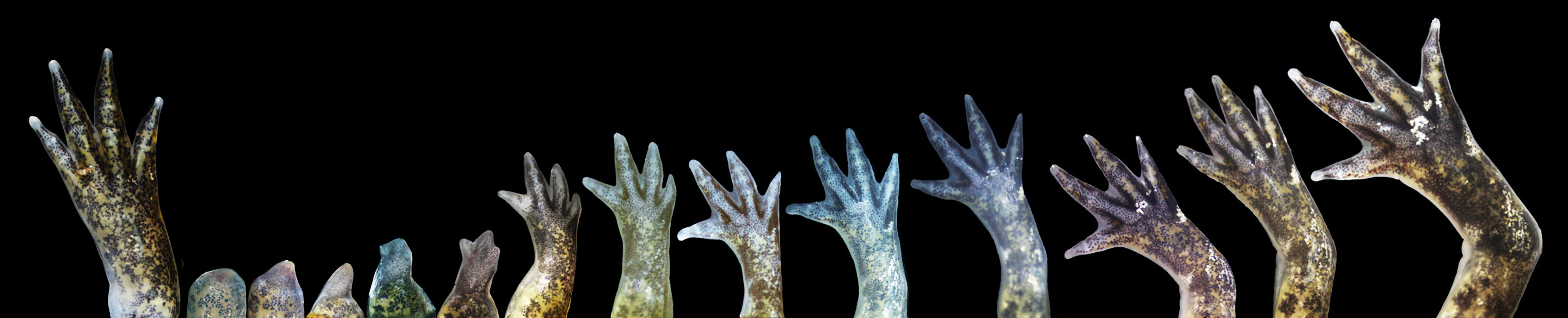
Axolotl salamanders can regenerate an amputated leg (shown in a time sequence from left to right). Levin and other researchers hope that further studies of what directs the development of form in embryos will someday make it possible to regenerate human limbs as well.
James Monaghan laboratory/Northeastern University
He thinks the outcomes might include the possibility of regenerating tissue and limbs — a trick that some amphibians, such as axolotls, are adept at but which we can’t do. “To me, this is the answer to the problem in regenerative medicine that we’re going to hit very soon,” he said. We’re very good at switching genes and manipulating molecules in cells, but we don’t know how to turn those dials to make fingers, eyes or limbs. “It’s entirely not obvious how you get changes to 3D anatomy by manipulating that lowest genetic level,” Levin said. “We need to learn how cells themselves encode whatever pattern they’re supposed to build, and then to rewrite that target morphology and let the cells do their thing.”
The potential for cells to find their way to body plans was dramatically illustrated recently with a report that when some sea slugs become heavily infected with parasites, their head separates from the body through self-induced decapitation and then regrows an entire new body within a few weeks. It’s tempting to see this as just an extreme case of regeneration, but that perspective leaves some profound questions hanging.
“First, where does the information for the anatomy it’s trying to regenerate come from?” Levin asked. “It’s easy to say ‘genome,’ but we now know from our xenobots that there is extreme plasticity, and cells are actually willing and able to build very different bodies.”
The second question, he says, is how regeneration knows when to stop. “How do cells know when the ‘correct’ final shape has been produced, and they can stop remodeling and growing?” he asked. The answer is critical for understanding the unruliness of cancer cells, he thinks.
Levin’s group is now studying whether adult human cells (which lack the versatility of embryonic cells) display a similar ability to assemble into “bots” if given the chance. Preliminary findings suggest that they do, the researchers said.
Organisms, Living Machines or Both?
In their paper, Levin and his colleagues discuss the potential of xenobots as “living machines” that could be used as microscopic probes or deployed in swarms to perform collective operations such as cleaning up watery environments. Adami, however, remains to be convinced that the Tufts team understands enough to begin to do this. “They have not shown that you can design these things, that you can program them, that they are doing anything that is not ‘normal’ once you release the mechanical constraints,” he said.
Levin is undeterred, however, and thinks that the ramifications of xenobots for fundamental science may ultimately go far beyond their biomedical or bioengineering applications, to any collective system that exhibits an emergent design not specifically encoded in its parts.
“I think this is bigger than even biology,” Levin said. “We need a science of where larger-scale goals come from. We’re going to be surrounded by the internet of things, by swarm robotics, and even by corporations and companies. We don’t know where their goals come from, we’re not good at predicting them and we’re certainly not good at programming them.”
Solé shares that wider vision. “This work is remarkable in particular for how much it reveals about the generative potential of self-organization,” he said. He feels it might broaden our view of how nature creates its endless forms: “One thing we also know well is that nature constantly tinkers with biological matter and that different functions or solutions can be achieved by different combinations of pieces.” Maybe an animal, even a human one, is not an entity written in stone — or rather, in DNA — but is just one possible outcome of cells making decisions.
Are xenobots “organisms,” though? Absolutely, Levin says — provided we adopt the right meaning of the word. A collection of cells that has clear boundaries and well-defined, collective, goal-directed activity can be considered a “self.” When xenobots encounter each other and temporarily stick, they don’t merge; they maintain and respect their selfhood. They “have natural boundaries that demarcate them from the rest of the world and allow them to have coherent functional behaviors,” Levin said. “That’s at the core of what it means to be an organism.”
“They are organisms,” Jablonka agreed. It’s true that xenobots presumably can’t reproduce — but then, neither can a mule. Moreover, “a xenobot may be induced to fragment and form two small ones,” she said, “and maybe some cells will divide and differentiate into motile and nonmotile ones.” If that’s so, xenobots could even undergo a kind of evolution. In which case, who knows what they might become?