Cells That ‘Taste’ Danger Set Off Immune Responses
Cells with taste receptors sometimes develop inside the lungs of animals infected with influenza. By “tasting” the presence of certain pathogens, these cells may act as sentinels for the immune system.
RenderBurger for Quanta Magazine
Introduction
When the immunologist De’Broski Herbert at the University of Pennsylvania looked deep inside the lungs of mice infected with influenza, he thought he was seeing things. He had found a strange-looking cell with a distinctive thatch of projections like dreadlocks atop a pear-shaped body, and it was studded with taste receptors. He recalled that it looked just like a tuft cell — a cell type most often associated with the lining of the intestines.
But what would a cell covered with taste receptors be doing in the lungs? And why did it only appear there in response to a severe bout of influenza?
Herbert wasn’t alone in his puzzlement over this mysterious and little-studied group of cells that keep turning up in unexpected places, from the thymus (a small gland in the chest where pathogen-fighting T cells mature) to the pancreas. Scientists are only just beginning to understand them, but it is gradually becoming clear that tuft cells are an important hub for the body’s defenses precisely because they can communicate with the immune system and other sets of tissues, and because their taste receptors allow them to identify threats that are still invisible to other immune cells.
Researchers around the world are tracing the ancient evolutionary roots that olfactory and taste receptors (collectively called chemosensory receptors or nutrient receptors) share with the immune system. A flurry of work in recent years shows that their paths cross far more often than anyone anticipated, and that this chemosensory-immunological network plays a role not just in infection, but in cancer and at least a handful of other diseases.
This system, says Richard Locksley, an immunologist at the University of California, San Francisco, helps direct a systematic response to potential dangers throughout the body. Research focusing on the interactions of the tuft cell could offer a glimpse of how organ systems work together. He describes the prospects of what could come from the studies of these receptors and cells as “exciting,” but cautions that “we’re still in the very early days” of figuring it out.
Not Merely Taste and Smell Receptors
One of life’s fundamental challenges is to find food that’s good to eat and avoid food that isn’t. Outside of our modern world of prepackaged food on grocery store shelves, it’s a perilous task. Taking advantage of a new type of food could mean the difference between starvation and survival, or it could mean an early death from accidental self-poisoning. Chemosensory receptors help us make this distinction. They’re so essential that even single-celled bacteria such as Escherichia coli carry a type of this receptor.
Despite the near universality of these receptors and their centrality to survival, scientists didn’t discover the big family of genes that encode for olfactory receptors until 1991, with the ones for taste receptors following in 2000. (The olfactory receptor discovery brought the researchers Richard Axel and Linda Buck a Nobel Prize in 2004.) Olfactory receptors and taste receptors for bitter, sweet and umami (savory) are all part of a large family of proteins called G protein-coupled receptors (GPCRs) that are embedded in cell membranes. Although the precise details vary from receptor to receptor, when a GPCR binds to the proper molecule, it sets off a signaling cascade within the cell. For taste and olfactory receptors in the mouth and nose, this cascade causes neurons to fire and enables us to recognize everything from the rich sweetness of a chocolate chip cookie to the nose-wrinkling stench of a passing skunk.
The discoveries of these receptors were momentous, groundbreaking advances, says Jennifer Pluznick, a physiologist at Johns Hopkins University. But in her view, labeling them as olfactory and taste receptors rather than as chemosensory receptors entrenched the idea that they function specifically and exclusively in smell and taste. If scientists found signs of these receptors in cells outside the nose and mouth, it was easy to write them off as mistakes or anomalies. She herself was shocked to find an olfactory receptor called Olfr78 in kidney cells, a finding that she reported in 2009.
“I think I even famously said something to my postdoc adviser, like, ‘I don’t even know that I can trust this data, you know?’” Pluznick recalled. “Olfactory receptors in the kidney? Come on.”
This wasn’t the first time these receptors had shown up in unexpected tissues. For example, in 2005, the University of Liverpool biochemist Soraya Shirazi-Beechey showed in a paper published in Biochemical Society Transactions that taste receptors could be found in the small intestine as well as the mouth. Their presence was surprising, but it made a certain sense that the intestine might use a taste receptor to monitor the food it was digesting.
But then in 2010, the laboratory of Stephen Liggett, who was then at the University of Maryland School of Medicine, reported that smooth muscle in the airways of the lungs expresses receptors for bitter taste. Moreover, they showed that these receptors were involved in a dilation response of the airways that helped to clear out obstructions.
Receptors for sweetness also turned up on the cells lining the airways. In 2012, a research group led by Herbert’s colleague Noam Cohen at the University of Pennsylvania found that the sugars coating the respiratory pathogen Pseudomonas aeruginosa activated those receptors and caused the cells to beat their hairlike cilia more rapidly, a process that can sweep away invading bacteria and prevent infections.
Meanwhile, Pluznick and her colleagues had continued to study the role of the Olfr78 receptor in the kidneys. They demonstrated in 2013 that it responded to molecules secreted by intestinal microorganisms, and that signals from that response helped to direct the kidney’s secretion of the hormone renin, which regulates blood pressure. “Other labs finding similar things in other tissues was both very encouraging and very exciting,” Pluznick said.
These studies and a torrent of others from labs around the world drove home the message that these seemingly misplaced olfactory and taste receptors serve important and often vital functions. And a theme common to many of those functions was that the chemosensory receptors often seemed to be alerting tissues to the presence and condition of microbes in the body. In hindsight, that application for the receptors made a lot of sense. For example, as Herbert notes, being able to “taste” and “smell” minute traces of pathogens gives the body more chances to respond to infections before microbes overwhelm the host’s defenses.
A Job for Tuft Cells
In researchers’ assays for chemosensory receptors in tissues throughout the body, a cell type that kept popping up was a relatively rare, largely unstudied one called a tuft cell. Tuft cells had been known to science since the mid-1950s, when microscopy studies found them in the lining of practically every organ in the body, including the gut, the lungs, the nasal passages, the pancreas and the gallbladder. The passage of a half-century, however, hadn’t led to any greater understanding of what tuft cells do. The further discovery of taste receptors on many tuft cells only deepened the mystery: Given their locations in the body, they certainly weren’t contributing to our sense of taste.
As a postdoc at Harvard University in the lab of Wendy Garrett in 2011, Michael Howitt became fascinated with tuft cells, especially those found in the intestines. “They were these really intriguing, weird cells that didn’t really have a clear function in terms of the normal physiology,” said Howitt, who is now an immunologist at Stanford University. He set out to learn the enigmatic cells’ function, and he eventually got his answer — through an unexpected discovery involving the mouse microbiome.
Because some studies had hinted at a link between taste receptors and immune function, Howitt wondered whether the receptor-studded tuft cells in the intestines might respond to the microbiome population of bacteria living in the gut. To find out, he turned to a strain of mice that other Harvard researchers had bred to lack a wide variety of bacterial pathogens.
But surprisingly, when he inspected a small sample of intestinal tissue from the mice, Howitt found that they had 18 times the number of tuft cells previously reported. When he looked more closely, he found that the mice carried more protozoa in their guts than expected — specifically, a common single-celled parasite called Tritrichomonas muris.
Howitt realized that T. muris wasn’t an accidental infection but rather a normal part of the microbiome in mice — something that neither he nor Garrett had thought much about. “We weren’t looking for protozoa,” Howitt said. “We were focused on bacteria.”
To confirm the relationship between the presence of the protozoa and the elevated numbers of tuft cells, Howitt ordered another set of similarly pathogen-free mice from a different breeding facility and fed them some of the protozoan-rich intestinal contents of the Harvard mice. The number of tuft cells in the new mice soared as the parasites colonized their intestines, too.
Lucy Reading-Ikkanda/Quanta Magazine
The numbers of tuft cells also climbed when Howitt infected mice with parasitic worms. But the increase didn’t happen in mice with defects in the biochemical pathways underpinning their taste receptors, including those on the tuft cells.
Howitt’s findings were significant because they pointed to a possible role for tuft cells in the body’s defenses — one that would fill a conspicuous hole in immunologists’ understanding. Scientists understood quite a bit about how the immune system detects bacteria and viruses in tissues. But they knew far less about how the body recognizes invasive worms, parasitic protozoa and allergens, all of which trigger so-called type 2 immune responses. Howitt and Garett’s work suggested that tuft cells might act as sentinels, using their abundant chemosensory receptors to sniff out the presence of these intruders. If something seems wrong, the tuft cells could send signals to the immune system and other tissues to help coordinate a response.
At the same time that Howitt was working, Locksley and his postdoc Jakob von Moltke (who now runs his own lab at the University of Washington) were homing in on that finding from another direction by studying some of the chemical signals (cytokines) involved in allergies. Locksley had discovered a group of cells called group 2 innate lymphoid cells (ILC2s) that secrete these cytokines. ILC2s, he found, release cytokines after receiving a signal from a chemical called IL-25. Locksley and von Moltke used a fluorescent tag to mark intestinal cells that produced IL-25. The only cells that gave off a red glow in their experiments were tuft cells. Locksley had barely even heard of them.
“Even textbooks of [gastrointestinal] medicine had no idea what these cells did,” he said.
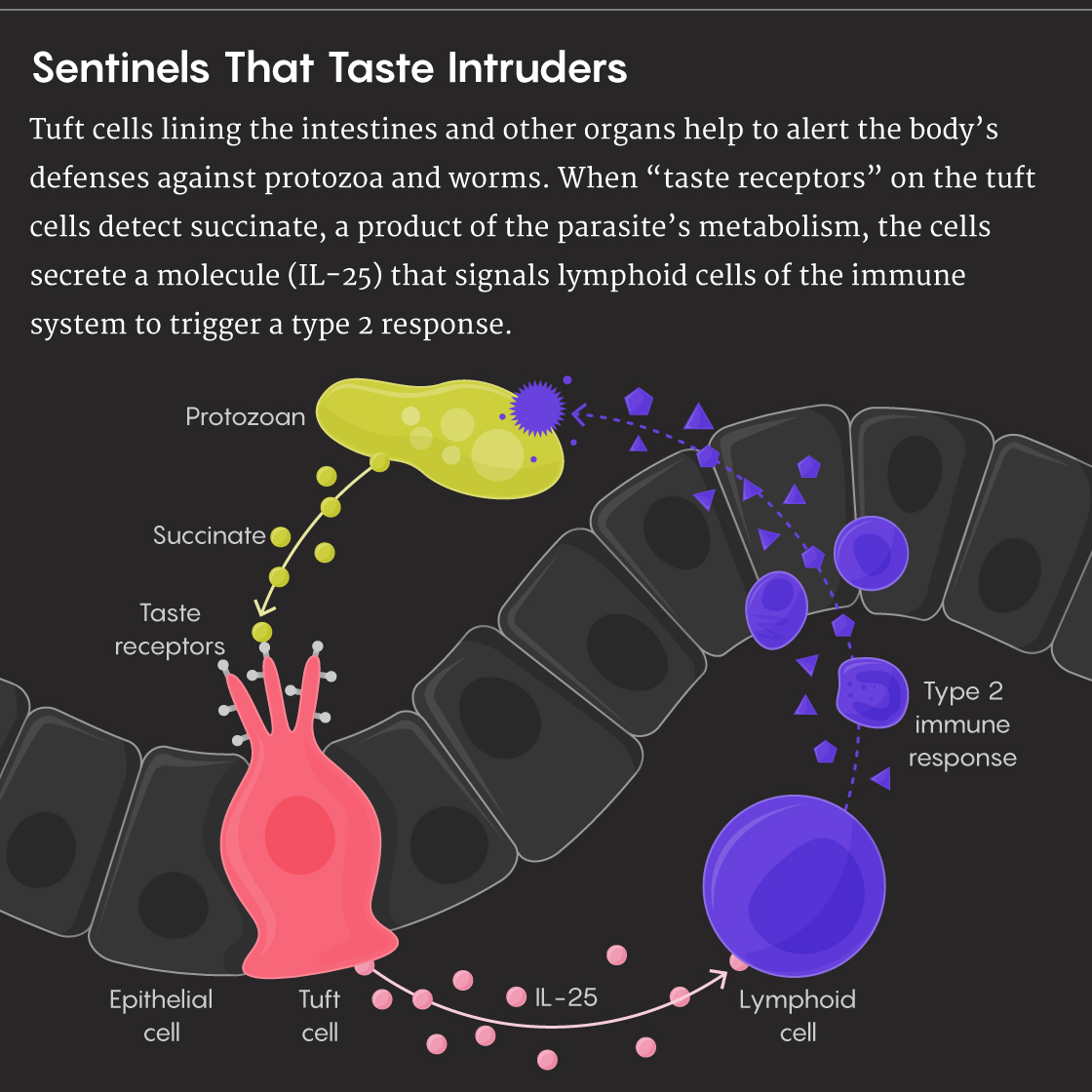
The Howitt-Garrett and Locksley-von Moltke papers were prominently featured in Science and Nature, respectively. Together with a third paper in Nature by Philippe Jay of the Institute for Functional Genomics at the National Center for Scientific Research in France and his colleagues, these studies provided the first explanation for what tuft cells do: They recognize parasites by means of a small molecule called succinate, an end product of parasite metabolism. Once succinate binds to a tuft cell, it triggers the release of IL-25, which alerts the immune system to the problem. As part of the defensive cascade, the IL-25 also helps to initiate the production of mucus by nearby goblet cells and triggers muscle contractions to remove the parasites from the gut.
For the first time, biologists had found at least one explanation for what tuft cells do. Before this, “people just kind of ignored them or didn’t even realize that they were there,” said Megan Baldridge, a molecular microbiologist at Washington University in St. Louis.
As groundbreaking as this trio of studies was, the work focused on intestinal cells. No one knew at first whether the tuft cells appearing elsewhere throughout the body play the same anti-parasitic role. Answers soon began to roll in, and it became clear that tuft cells respond to more than succinate and do more than help repel the body’s invaders. In the thymus (a small globular outpost of the immune system nestled behind the breastbone), tuft cells help teach the immune system’s maturing T cells the difference between self proteins and non-self proteins. Kathleen DelGiorno, now a staff scientist at the Salk Institute for Biological Studies, helped to show that tuft cells can help protect against pancreatic cancer by detecting cellular injury. And in Cohen’s studies of chronic nasal and sinus infection, he discovered that recognition of bacterial pathogens such as Pseudomonas aeruginosa by receptors for bitterness on tuft cells causes neighboring cells to pump out microbe-killing chemicals.
As a lung biologist and a colleague of Herbert’s at the University of Pennsylvania, Andrew Vaughan followed these tuft-cell discoveries with interest. In many cases, tuft cells appeared to be intimately involved with the part of the immune response known as inflammation. Vaughan was studying how tissue deep in the lungs repairs itself after inflammation caused by the flu virus. After reading about some of the new findings, Vaughan began to wonder whether tuft cells might be involved in the lungs’ recovery from influenza. He and Herbert infected mice with the influenza virus and searched the lungs of those with severe symptoms for signs of tuft cells.
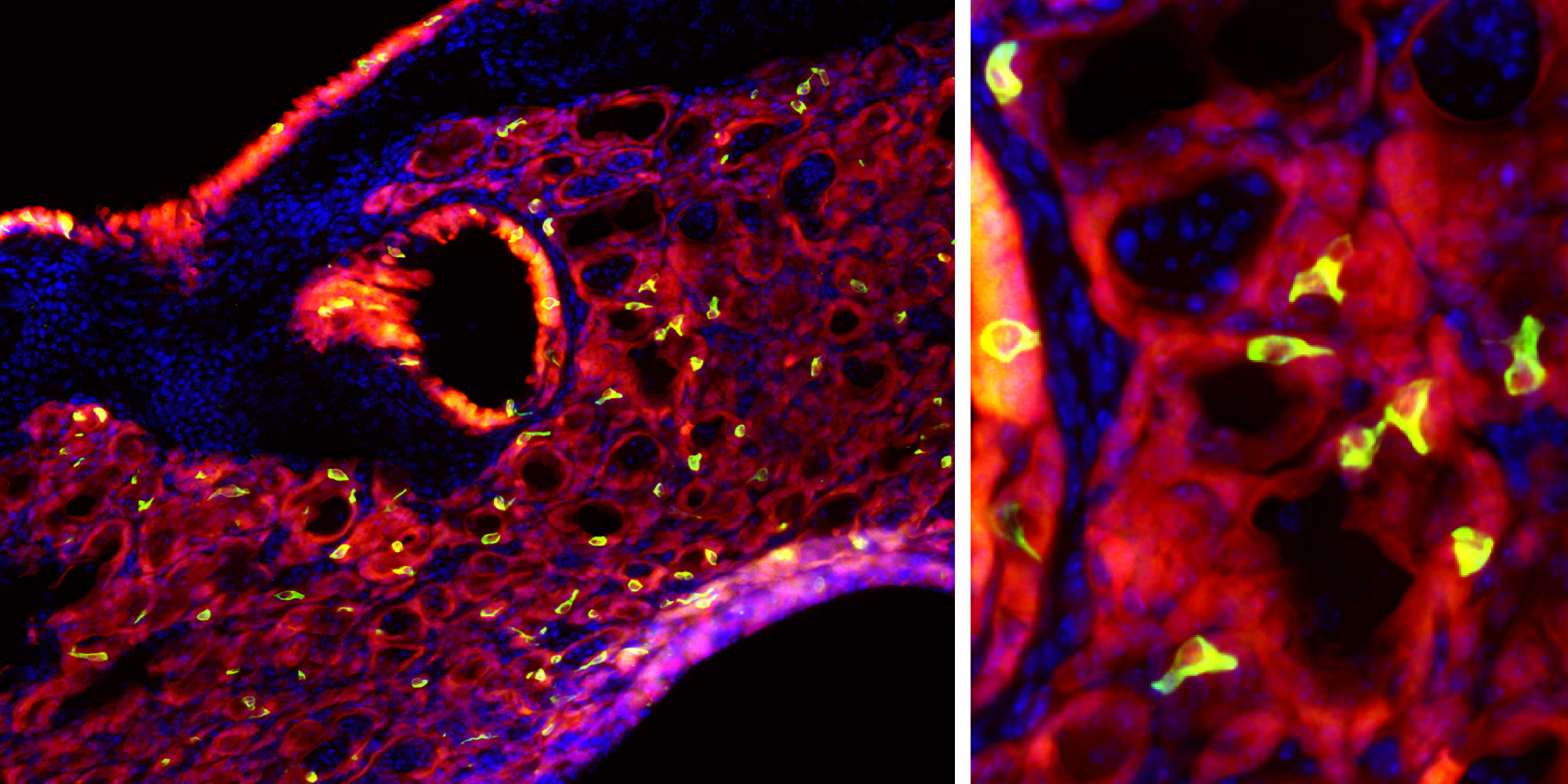
In a micrograph of lung tissue sampled 25 days after an influenza infection, newly emerging tuft cells are stained yellow/green (at left; a closeup appears at right). Tuft cells don’t normally appear in these airways, but they seem to emerge as part of the body’s response against the infection.
Courtesy of Andrew Vaughan
“Sure enough, they were all over the place,” Vaughan said. But the tuft cells only appeared after influenza infection, which made Vaughan believe that he and Herbert were “basically seeing a cell type where [it’s] not supposed to be.” Although he’s unsure exactly why this proliferation of tuft cells happens after the flu, Vaughan speculates that it might be an aspect of the body’s attempt to repair damage from the virus as part of the broader type 2 immune response.
The researchers don’t yet know what the tuft cells are doing in the lungs or what they are sensing, but Herbert believes that their ability to continually “taste” the environment for different compounds provides a key opportunity for the body to respond to even minute threats.
The tuft cell, Herbert said, is constantly sensing the metabolic products present in microenvironments within the body. “Once some of those metabolic products go out of whack … bam! Tuft cells can recognize it and make a response if something is wrong.”
Newly discovered connections between tuft cells and the immune and nervous systems provide further evidence that chemosensory receptors are multipurpose tools like Swiss Army knives, with evolved functions beyond taste and smell. It isn’t clear which function evolved first, though, or whether they all evolved in tandem, Howitt says. Just because scientists became aware of “taste” receptors on the tongue first, “that doesn’t mean that’s the order in which it evolved.”
In fact, a preliminary study in rats hints that the receptors’ immune functions may have evolved first. Two groups of immune cells known as monocytes and macrophages use formyl peptide receptors on their membranes to detect chemical cues from pathogens, and a group of Swiss scientists showed that rats use these same receptors to detect pheromone odors. Those facts suggest that at some point in history, the ancestors of rats made scent receptors out of the immunological molecules. The evolutionary history of other groups of olfactory and taste receptors has yet to be deciphered.
Whatever their history, scientists now say that a major role of these receptors is to monitor the molecules in our body, tasting and smelling them for any sign that they might be from a pathogen. Then, with help from tuft cells and other parts of the immune system, the body can fight off the invaders before they’ve gotten a foothold. But Vaughan cautioned that the sudden emergence of tuft cells in tissues like the lungs, where they are not always present, might also cause its own pathologies.
“You may not always want to have the ability to [defensively] overreact,” he said. That could be part of what goes wrong in conditions like allergies and asthma: There could be dangers “if you have too many of these cells and they’re too poised to respond to the external environment.”
Correction added Nov. 18, 2019: The caption below the photograph of Herbert originally made an unconfirmed statement that he was the first to observe the tuft cells in the infected lungs of the mice.
This article was reprinted on Wired.com.




