Immune Cell Assassins Reveal Their Nurturing Side

Macrophages and other cells of the immune system are known as destroyers of bacteria and other pathogens. But research is finding that they also have important jobs in healing and molding the body.
Maylee for Quanta Magazine
Introduction
After a heart attack, patients are increasingly often offered the option of stem cell therapy, in which stem cells from their bone marrow are injected into the heart to help it heal. Skeptics, however, point out that solid evidence of the therapy’s benefits is lacking: It’s worked modestly in some animal studies, but its effectiveness is uncertain, and scientists have only been able to guess at how it helps if it does.
Last November, a team of cardiologists set out to provide some clarity on this controversial treatment. Instead, their work found evidence that some immune system cells play a nurturing, healing role that is far removed from their familiar calling as bloodthirsty protectors of the body.
Scientists and doctors from Cincinnati Children’s Hospital led by Jeffery Molkentin first injected stem cells into mice whose hearts had been temporarily deprived of oxygen to mimic a heart attack. Their hearts showed some transient inflammation from the injections, but the mice healed mildly better than those that received a placebo. Still, it was possible that the inflammation, rather than the stem cells, contributed to the improvement: “Any good immunologist will tell you, you need an inflammatory response to get healing,” said Molkentin.
To find out, the team injected a second set of heart-damaged mice with zymosan, a chemical that induces inflammation, instead of stem cells — and they saw the same improvement in heart function. Finally, they injected only bits and pieces of dead cells, which would do nothing more than prompt the immune system to infiltrate the tissue and clean up the debris. Even this, they found, improved heart function.
The protective benefit of the treatment didn’t come from a regenerative effect of the stem cells, the researchers realized. It came instead from the inflammatory immune response, which seemed to set up what Molkentin calls “a second wave of healing.”
Molkentin’s study is the latest in an avalanche of papers over the last decade showing that certain immune cells moonlight in roles unrelated to fighting disease. The phenomenon is not limited to the heart. These immune cells, many of which reside permanently in specific tissues, have been identified as participants in a range of biological activities, including heartbeat regulation, the stabilization of pregnancy, and even brain development.
More Than Elite Assassins
Immune system nomenclature is rife with cell labels suitable for fierce, battle-ready warriors. The Greek name “macrophage,” for example, translates as “big eater” and evokes images of ravenous, rotund cells gobbling up the bits of debris floating around them. “Natural killer” (NK) lymphocytes sound like elite assassins sailing through the bloodstream, breezily picking off disease-causing cells.
“When the names were given to immune cells, it was always in the context of what they did to protect us,” said Muzlifah Haniffa, an immunologist and single-cell genomics researcher at Newcastle University. Haniffa recently published an atlas of the developing blood production system in the human fetus as part of the Human Cell Atlas project, which aims to develop a comprehensive map that catalogs every type of cell in the human body. She believes that the names we gave immune cells may have pigeonholed them and prevented us from understanding their full range of abilities.
Consider the macrophage. It is a type of phagocyte — literally an “eating cell” — described and named in 1882 by the Russian biologist Elie Metchnikoff. Metchnikoff had a hunch that starfish could rid themselves of foreign bodies, so as an experiment, he inserted a rose thorn into a transparent starfish larva. Peering through the primitive microscope on his desk, he watched as hungry phagocytic cells lumbered toward the thorn, surrounded it and gobbled it up.
Metchnikoff didn’t immediately assume that these phagocytes are specialized for disease fighting and pathogen removal alone. Trained as an evolutionary biologist, he knew that simple organisms generally had relatively unspecialized cells. He therefore hypothesized that phagocytes performed a variety of other basic biological jobs that molded and maintained the healthy function of normal tissues for the animal.
But immunology was a highly contentious field in those days, and Metchnikoff, who was considered fiery and difficult, wasn’t taken seriously at first by his peers. Many of them believed instead that antibodies and other substances in the blood plasma, not cells, were the primary agents of immunity. It was years before subsequent research substantiated Metchnikoff’s cellular immunology concepts and he was recognized for his work on the macrophage, which quickly became one of the immune system’s star players. (In 1908 he and Paul Ehrlich shared a Nobel Prize for their separate studies of the basis of immunity.)
The next century brought some of the greatest advances in medicine and immunology, and amid the exuberant fanfare for these discoveries, the nonimmunological roles that Metchnikoff had suggested for the macrophage faded into the background. But now, because of advances in immunology and single-cell RNA sequencing, these roles are coming back into focus.
The Heart of the Matter
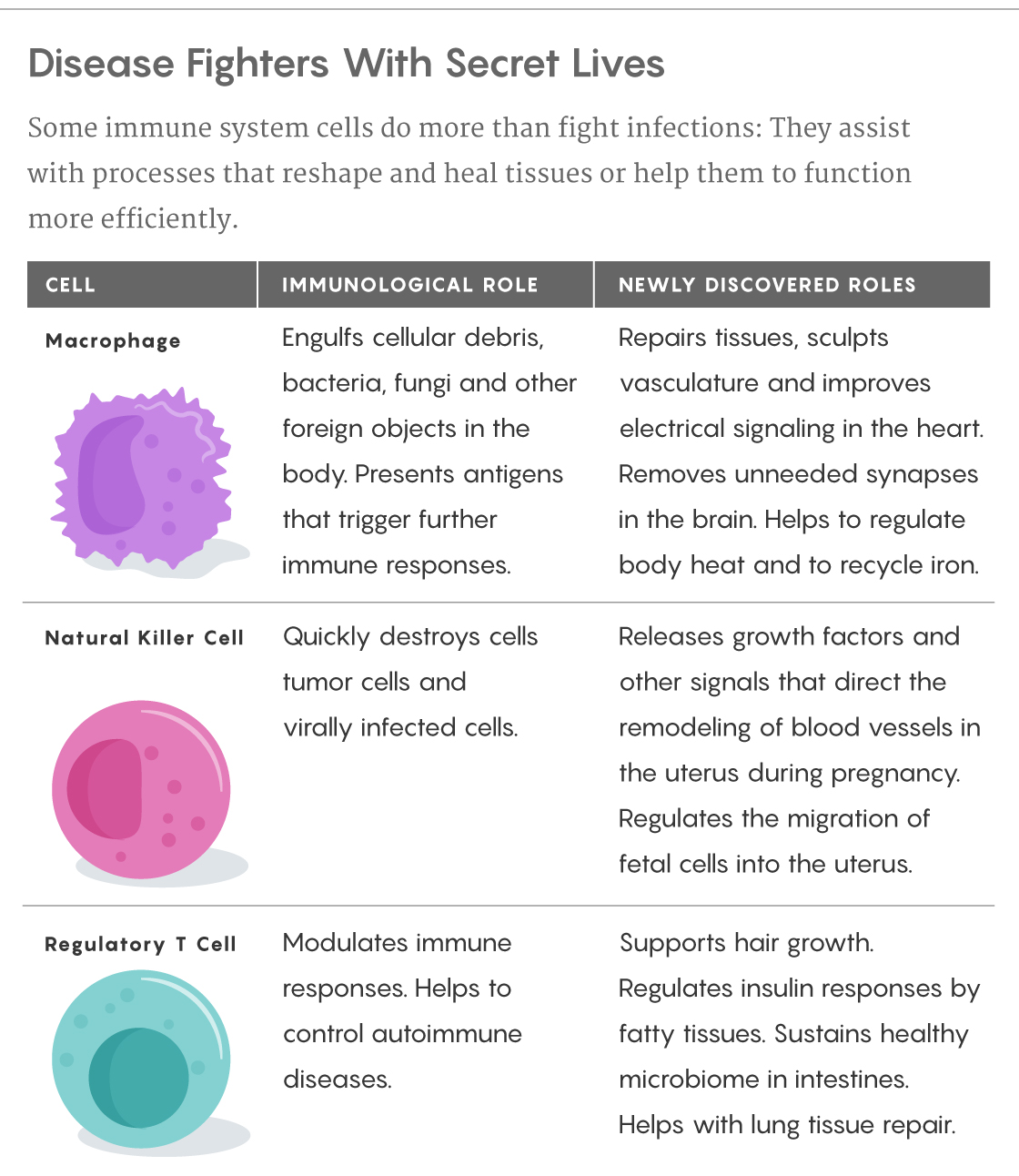
Metchnikoff would probably have found Molkentin’s heart study validating. The cells that delivered the healing boost to the hearts of the mice are a subset of what are called tissue-resident macrophages.
Unlike the macrophages that circulate in the blood and look for pathogens, these cells migrate into the heart during embryonic development and remain there for the rest of their lives. Over the past decade or so, evidence has accumulated that they perform a variety of tasks, such as aiding in the maturation of coronary vasculature and maintaining a proper heartbeat.
“They’re doing activities that are not normally associated with immunology, such as helping tissues reshape and change in response to stresses, or repair and regenerate, or even conduct electricity,” said Kory Lavine, an assistant professor of medicine at the Washington University School of Medicine in St. Louis.
Lavine gained some of the first insights into the origins of tissue-resident macrophages in the heart while studying sex-mismatched heart transplants — cases in which the heart of a female donor went to a male recipient or vice versa. In biopsied tissues, he was able to see that the macrophages in the heart were from the original donor, which meant that they stayed in the heart for the lifetime of the organ.
Lucy Reading-Ikkanda/Quanta Magazine
In 2014, Lavine published a paper showing that when an embryonic heart is damaged, these cells can repair and regenerate the tissue. Then in 2016, his group presented evidence that CCR macrophages in the heart actively sculpt the mature layout of the organ’s blood vessels. During embryonic development, blood vessels are laid down in the heart before the blood begins circulating. Once blood flow starts, however, vessels are weeded out so that only the best routes are maintained. Lavine found that CCR macrophages are integral to this process.
A few years ago, research was published showing that macrophages are abundant throughout an electrically conductive region of the heart known as the atrioventricular (AV) node, which connects the heart chambers called the atria and ventricles in mice and humans. The macrophages in the AV are elongated, with projections that extend their reach. When scientists bred mice that lacked these cells, they found significant delays in the conduction of electrical signals through the AV node. When they chemically blocked the activity of these macrophages, they witnessed “AV block,” an impairment of the electrical signal traveling from the atria to the ventricles.
How the macrophages aid conduction is still unclear, but it appears that their presence primes the heart cells’ firing signals to travel more quickly. The finding has scientists now looking into whether abnormalities in heart macrophages can lead to arrhythmias in humans.
The heart is not unique. In fact, most tissues and organs in the body have their own cache of tissue-resident macrophages. They have been found to carry out key functions, as if they were a part of the organ in which they reside. In the brain, for example, they remove axons and aid in the pruning of synapses during development. Those in adipose tissue help to regulate body heat. Macrophages have even been found to aid in the recycling of iron in the spleen and liver.
From Killers to Builders
If macrophages are the greatest multitaskers in the immune system, then natural killer cells are the most poorly named. NK cells were first identified for their ability to destroy tumor cells on contact by blasting them with chemicals that induce apoptosis or cellular suicide. They are powerful players in the immune system’s defensive arsenal.
But almost from the time of their discovery, scientists have noted that subpopulations of these NK cells reside full time in the liver, skin, kidney and uterus. And unlike their deadly cousins, these cells don’t kill.
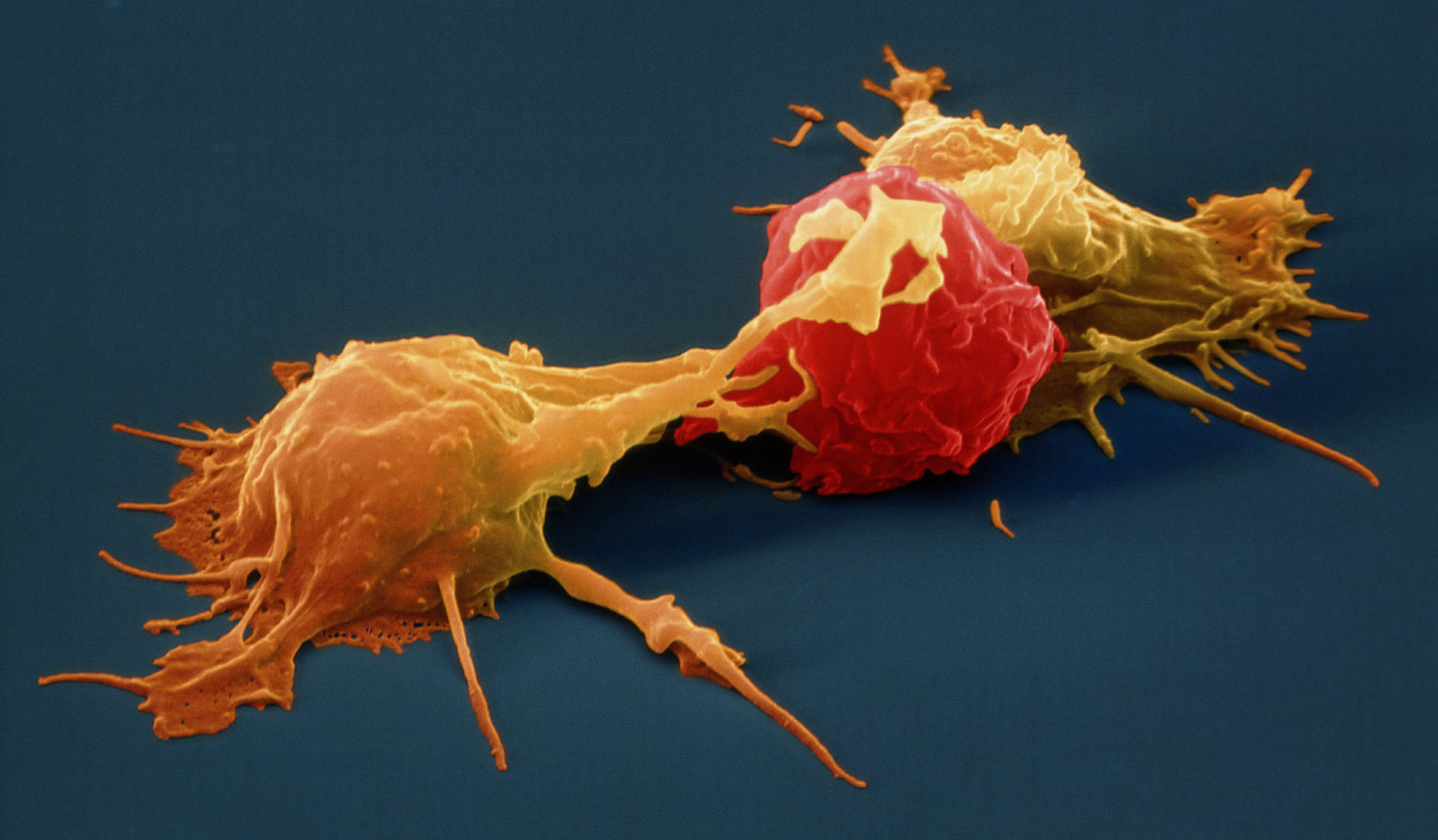
Two natural killer (NK) cells (yellow) attack a cancer cell (red) in this artificially colored micrograph. NK cells took their name from how avidly they attacked abnormal and infected cells, but some of them also help to ensure the blood supply to the uterus during pregnancy.
Eye of Science/Science Source
In the uterus, NK cells make up 70% of the white blood cells during the first half of pregnancy. Early experiments in mice showed that when these uterine NK cells were isolated and pitted against mouse lymphoma cells — a natural adversary — they lacked the cancer-fighting powers of their immune system brethren. This revelation prompted scientists to ask what, exactly, the NK cells were doing there.
Early work by the pioneering scientist Anne Croy of Queen’s University pointed to an answer. The Canadian scientist, who was trained as a veterinarian, studied pregnancy and the immune system in mice. She noticed that these cells tended to congregate at the very edge of the maternal-fetal interface in pregnant mice, where the placenta meets up with the lining of the uterus. That fact led her to hypothesize that NK cells were involved in the remodeling of blood vessels in the uterus.
During pregnancy, fetal cells “remodel” maternal arteries in the uterus so that they will no longer respond to the mother’s fight-or-flight signals. Imagine a pregnant mouse being chased by a cat (or a pregnant woman fleeing a tiger): The rush of adrenaline she experiences makes the blood vessels to her organs shrink and shunts blood to her muscles to aid her escape. But those changes could be detrimental and possibly lethal for a fetus in her uterus by depriving it of blood and the oxygen and nutrients it delivers. Evolution has invented this remodeling mechanism as protection against that physiological response to a crisis.
In a set of landmark experiments, Croy showed that uterine NK cells control the vascular changes that happen during pregnancy by means of substances they secrete from their granules. In NK cells that circulate throughout the body, similar granules are normally filled with an assassin’s cocktail of toxins, but in the uterine NK cells, they carry growth factors and chemokines (messenger molecules) that attract other cells. Once released, these molecules attract endothelial cells and trophoblasts, which are fetal cells from the placenta that can also remodel the blood vessels in the uterus.
“Instead of being killers, they are really builders,” Francesco Colucci, an immunologist at the University of Cambridge, said of the NK cells. Colucci published research showing that uterine NK cells regulate the extent to which fetal cells can invade the uterus, and he is now using RNA sequencing techniques to characterize different types of uterine NK cells (as described in a paper he published on this in Nature Communications in late January).
“Natural killer cells are actually playing a role in supporting healthy pregnancies, but it has got nothing to do with killing,” said Haniffa. In 2018, Haniffa and colleagues published a map of the maternal-fetal interface with single-cell resolution that revealed the gene activity of these NK cells, further elucidating their dexterity.
Mapping Out a Theory
Natural killers and macrophages are some of the best-characterized examples of multitasking immune cells, but there are many more. Regulatory T cells, or Tregs, a subset of the T lymphocytes, modulate the immune response. But they have been shown to be involved in other processes as diverse as hair growth in the skin and insulin regulation in fat tissue. Innate lymphoid cells — lymphocytes that do not express the same antigen receptors as B and T cells — are involved in metabolism and even the healthy function of the nervous system. Also, combinations of these cells have been observed in crosstalk with stem cells, helping to maintain the regeneration of tissues that constantly turn over like the skin and the intestinal lining.
“For a long time, people thought of the immune system as basically what’s in your blood,” Haniffa said. “Then they realized that your immune system doesn’t just exist in your blood, it exists in every tissue.” Moreover, the immune system cells embedded in tissues and even among your microbiota are in communication. The cells in the brain called microglia have traditionally not been recognized as part of the immune system, but they consume cellular debris like macrophages. They have also been shown to respond to signals from gut microbiota. “We should view the immune system as a bit like a matrix that exists in the entire body,” Haniffa said.
Aviv Regev, a computational biologist at the Broad Institute who helped launch the Human Cell Atlas, echoes these thoughts. You can think of immune cells as one of the major sensing systems in the body along with the nervous system, she said: “We often thought of [immune cells] in narrower functional terms, but we increasingly realize that their roles are broader.”
Haniffa wants to explore the immune system’s role in embryonic development. Last October, she and her colleagues published a study in Nature that detailed the gene activity in individual cells from the developing blood system and immune system of a human embryo. They profiled more than 200,000 cells from the embryonic yolk sac, liver, skin and kidneys at various points between the seventh and 17th weeks of development. The work was a milestone because it was the first effort to map the development of the human immune and blood systems, including red and white blood cells, with single-cell resolution.
According to Shruti Naik, a stem cell biologist and single-cell researcher at New York University, “this paper has huge implications” for our “understanding of not just human gestation but inborn errors in immunity and developmental disorders.”
Haniffa found that a trove of immune cells was present very early in human development, which she thinks could signify that the cells have an important part to play in the development of tissues. She points out that mast cells, which are traditionally involved in allergic reactions, show up in the yolk sac during the first trimester. Why would they be there when allergy is not typically an issue for embryos? But mast cells have been also implicated in blood vessel development in cancer, so Haniffa wonders whether they might have something to do with healthy blood vessel formation too.
Regev notes that more research is still needed to elucidate the functions of mast cells at various stages of development. But to her, “the possibility that the cells that arise early have more diverse functions in developing tissue is a very compelling hypothesis.”
Correction: February 12, 2020
An earlier version of this story misstated the year in which Metchnikoff identified phagocytes; it was 1882, not 1845.



