In Sexy Worms, Inheritance Beyond Genes Can Help Evolution
Introduction
Playing the mating game is risky. Organisms must cope with the existential risk that swiping right on the wrong choice could doom future generations to a lifetime of bad genes. They also have to contend with more immediate burdens and risks: Participants need to gather resources for courting and summon energy to pursue a potential partner. Animals engaged in amorous activities also make easy targets for predators.
Small wonder, then, that when times are good, the roundworm Caenorhabditis elegans doesn’t bother with the process. As a mostly hermaphroditic species (with a few males thrown in for variety), a C. elegans worm usually self-fertilizes its eggs until its sperm stash is depleted late in life; only then does it produce a pheromone to attract males and stay in the reproductive game. But when environmental conditions become stressful, the worms become sexually attractive much sooner. For them, sex is the equivalent of a Hail Mary pass — a desperate gamble that if their offspring are more genetically diverse, some will fare better under the new, rougher conditions.
Scientists thought this stress-induced shift was purely fleeting. But recently when scientists at Tel Aviv University raised C. elegans in too-warm conditions for more than 10 generations, they discovered that the worms continued to be sexually attractive for several more generations after they were moved to cooler surroundings. It’s an observation that highlights how inheritance does not always reduce to a simple accounting of the genes in organisms, and it may point to a mechanism that works in tandem with traditional natural selection in shaping the evolution of some organisms.
As the new paper in Developmental Cell shows, the cause of this trait wasn’t a genetic change to the worm’s DNA but rather an inherited “epigenetic” change that influenced how the DNA was used. The researchers — senior author Oded Rechavi, a biologist at Tel Aviv University, first author Itai Toker (now a postdoctoral fellow at Columbia University) and their colleagues — identified a small RNA molecule that can be passed between generations to signal for production of the pheromone. In effect, this heritable RNA molecule improves the odds that the worms will evolve in stressful times.
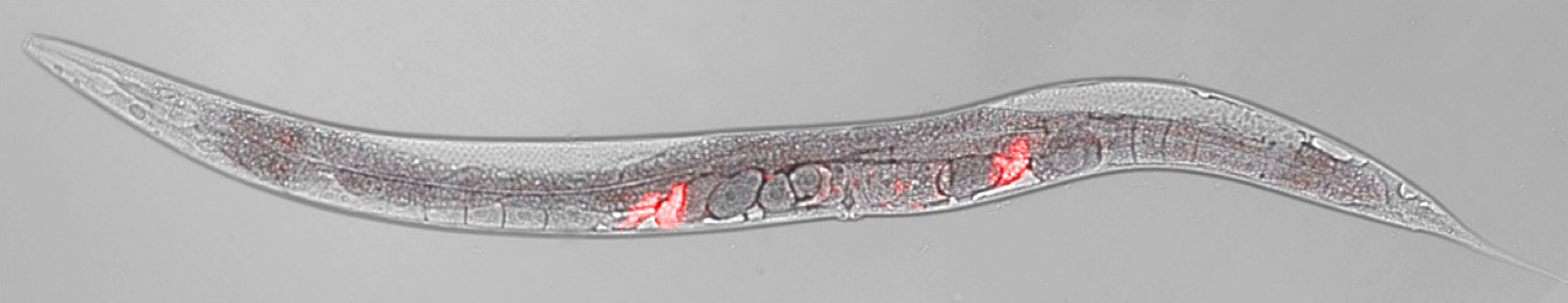
This hermaphroditic worm has just mated with a male; the received sperm are tagged with red. C. elegans worms mostly self-fertilize, but stressful conditions can make them turn sexually attractive.
Itai Toker
“Epigenetics has this double function: They’re inheritance systems, but they’re also response systems. You respond to the environment not by waiting for a mutation but by changing the way you express your genes,” said Eva Jablonka, a geneticist and philosopher of science at Tel Aviv University, who was not involved in the study.
The predominant view of evolution over the past century has been that advantageous new traits improving the fitness of populations and species need to originate as DNA mutations. But to Tai Montgomery, an RNA biologist at Colorado State University, the new study provides valuable clues about the potential for short-term epigenetic inheritance to influence longer-term evolution, too.
“It’s a tantalizing study that points to the possibility for the epigenetic inheritance of information actually leading to the evolution via changes in the DNA sequence within a population,” he said.
Breaking the Mouse-Tail Dogma
The concept of epigenetics was first explored in the 1880s, a particularly bad time to be a mouse in Freiburg, Germany.
In his attempts to separate heritable traits from nonheritable ones, the biologist August Weismann amputated the tails of hundreds of mice and then let them breed to see if the mice would transmit their new tailless status to their offspring. After going through five generations of mice and a sizable stack of pink tails, Weismann concluded that no amount of tail amputation would lead to tail-free progeny. He wrote in an 1888 book of essays that these “acquired characteristics” were not inherited because they did not impact sperm and egg. To subsequent biologists, the Weismann barrier meant that traits that arose because of the environment could not be passed on.
Cracks in that dogma began appearing in the 1940s when the British biologist Conrad Waddington experimented on fruit flies. Scientists knew that subjecting fly pupae to a pulse of heat made them mature into adults that lacked a characteristic transverse (or “cross”) vein in their wings. But when Waddington repeated the heat treatment in the offspring of the “crossveinless” fruit flies, he noticed that they eventually began to lack the cross vein even without a temperature shock; unlike Weismann’s mice, they were inheriting the acquired characteristic.
Waddington hadn’t changed the fruit flies’ DNA. Instead, he had altered something else, “the interactions of genes with their environment which bring the phenotype into being,” as he put it. He called this epigenetics because it operated on top of, or over, genetics.
If DNA sequences are the words in the book of life, then epigenetics is the punctuation, marking the difference between “Let’s eat, Grandma!” and “Let’s eat Grandma!” By telling the cell’s protein translation machinery where to work and how active to be, epigenetic signals can work either in addition to existing genetic signals or as the manifestation of these genetic commands. This layer of regulation provides an interface between the genome (which rarely changes) and the environment (which is always in flux), explained Eric Greer, an epigeneticist at Harvard Medical School.
“Epigenetics exists at the interface between the environment and the genetics,” he said.
Not until the 1970s would scientists begin to discover how epigenetics works; in fact, they are still uncovering the multitude of mechanisms cells use to regulate gene activity. Classic epigenetic regulation — the kind Waddington discovered — involves chemical tags attached to DNA and modifications of the histone proteins that change how readily a gene can be translated into protein. The growth in molecular biology tools, however, has more recently allowed researchers to identify other epigenetic mechanisms that depend on small RNA molecules in invertebrates like C. elegans.
These snippets of single-stranded nucleic acids have a built-in penchant for altering gene activity. Many of these RNA molecules are ephemeral, lasting only minutes or hours. Others are more persistent.
For example, the biologists Andrew Fire (now at Stanford University), Craig Mello (of the University of Massachusetts Chan Medical School) and their colleagues showed in 1998 that injecting small pieces of “interfering RNA” (RNAi) could prevent the translation of messenger RNA, and that these effects could be inherited in fertilized eggs for several generations. For their discovery of this phenomenon, which they dubbed gene silencing, Fire and Mello received the Nobel Prize in Physiology or Medicine in 2006.
In the years since, researchers have found evidence in invertebrates for the RNA-based epigenetic inheritance of pathogen avoidance, response to olfactory cues, longevity and more. Precisely how the gene silencing is inherited is still under study, but somehow either RNAi molecules or the instructions for making them pass from parents to offspring.
“Only in the last few years have we started being able to really provide some evidence that small RNAs themselves can be the vessel that is actually transferring information from one generation to the next,” said Lamia Wahba, a postdoctoral fellow in the Fire lab at Stanford.
From an evolutionary standpoint, it makes sense for epigenetic effects to be heritable, said Greer. With mere days between generations, the offspring of C. elegans are likely to encounter environmental conditions like those experienced by their parents. Preparing future generations for heat, famine or other stressors could help them survive harsh conditions.
But could this type of RNA-based epigenetic inheritance alter an organism’s long-term evolutionary trajectory? Or, since the trait wasn’t permanently wired into the DNA, would it be too transitory to have a lasting effect on the population? “This was an open question that we wanted to tackle,” Toker said.
The mating habits of C. elegans offered a way to find out. For more than a decade, it’s been known that in C. elegans, when the going gets tough, the tough get mating. Rechavi and Toker realized that if a small RNA was causing the acquired trait of attractiveness, then they could measure its evolutionary significance through its impact on the genetic variation in the population. Genetic variation is the raw material for natural selection, after all.
How Worms Got Hot
While a sojourn at 25 degrees Celsius might be an ideal holiday for humans, it’s borderline hot to C. elegans. If the scientists cranked up the temperature for just a few generations, not much happened. The worms continued to produce the male-attracting pheromone only when they ran short of their own sperm. But when Toker and Rechavi extended the heat for upward of 10 generations, they found that the worms began secreting the pheromone much earlier in life, long before their own sperm stash had dwindled. Intriguingly, this pattern continued for three generations after the worms were returned to cooler (20 C) conditions.
Toker and Rechavi first needed to confirm that small RNAs were responsible for the inherited mating effect. This proved to be true: When the researchers prevented short interfering RNAs from being transmitted between generations, the offspring didn’t express the pheromone to attract mates prematurely.
Merrill Sherman/Quanta Magazine
Then Toker and Rechavi checked their prediction that the transgenerational epigenetic change could affect the genetic diversity of the worm population. They found that even though worms with inherited attractiveness made 15% fewer sperm on average, they mated more often. As a result, their genes rapidly spread through the population, both in multigenerational mating competition experiments and in mathematical simulations. In this way, the epigenetic effect did seem primed to increase the “evolvability” of the worms, improving the chances that any useful new genetic traits they picked up would quickly take root in the population.
Nevertheless, Coleen Murphy of Princeton University, who also studies transgenerational epigenetic inheritance in worms, isn’t yet convinced that the observed effect is relevant to the real world. For 10 generations (roughly a month), Rechavi and Toker had to keep the worms in a narrow temperature range between 25 C and 27 C: hot enough to see the precocious mating effect become heritable but not so hot that it triggered the worms to enter what’s called a “dauer” state of stasis instead of reproducing.
“I don’t know how to interpret that significance,” she said.
Toker agrees that designing laboratory experiments to reflect real-world phenomena more accurately is a big issue for the field, and one that scientists have yet to resolve. Showing that this epigenetic inheritance can happen in the lab is only the first step toward understanding its overall significance, he said.
Another major question that biologists are beginning to face is whether multigenerational epigenetic inheritance happens in vertebrates. Although scientists have found tantalizing clues that this might be happening in mammals, and even in humans, many scientists remain unconvinced. The evidence for such inheritance, they argue, is circumstantial at best. They also point out that the offspring of animals with long life spans are less likely to experience the same environmental conditions as their parents, making epigenetic inheritance less beneficial. In January 2022, researchers at McGill University and the University of Chicago failed to replicate previous findings that showed mice could epigenetically inherit “memories” of previous infections.
Still, the recent Developmental Cell paper confirms to Montgomery that even a seemingly temporary shift in epigenetic inheritance over several generations can shape the evolution of some forms of life.
When an organism adapts to a changing environment through genetic mutations, it is committing to that new way of life, he explained. If the environment changes back, the organism will find itself adapted to conditions that no longer exist. Epigenetic changes might offer a more gradual interim solution — a way to cope with a changing environment and even to participate in natural selection before a more permanent genetic change is locked into the DNA.
“This transgenerational epigenetic inheritance could allow you to temporarily adapt to your environment,” Montgomery said. “You could have this period of time to sample the environment … and decide at what point do you commit to this being the new reality.”
To Wahba, this work is another step in understanding the ever-broadening intricacies of evolution and natural selection.
“It’s a little more complex than Darwin’s original ideas,” she said. “There’s a huge part of heritability that can’t be explained by genetics.”
Editor’s note: Coleen Murphy is an investigator with the Simons Collaboration on Plasticity and the Aging Brain, sponsored by the Simons Foundation, which also funds this editorially independent magazine.



