New Hybrid Species Remix Old Genes Creatively

In Africa, the group of fish known as cichlids has explosively diversified to more than 700 species in 150,000 years. Hybridization between species may help to account for how the new species arose so quickly.
Sabena Jane Blackbird / Alamy Stock Photo
Introduction
To biologists eager to understand how one species can split into two, the apple maggot fly seemed primed to spill the genetic secrets.
Sometime within the past 170 years, populations of Rhagoletis pomonella jumped from infecting the fruits of native hawthorn trees in North America to infecting apple trees introduced by European colonists, a move that formed an incipient species. The new apple-specialist insects even altered their developmental schedule by maturing earlier, when apple trees are fruiting. Finding the new genetic differences that distinguished and isolated the two types of flies should have been easy.
The researchers did find one — but the timing seemed all wrong. It wasn’t a new mutation at all; instead, the genetic variant had originated in an isolated Mexican population over 1 million years ago and then spread north after some ancient hybridization event. How could a speciation gene be orders of magnitude older than the actual speciation?
In the past, this might have been interpreted as a biological fluke. But as researchers investigate more genomes from diverging lineages, especially those that form species at a blistering pace, this pattern keeps popping up.
Many genetic variants tied to the reproductive isolation of species are older than the species themselves. These old variants are often injected into lineages through hybridization with distant relatives.
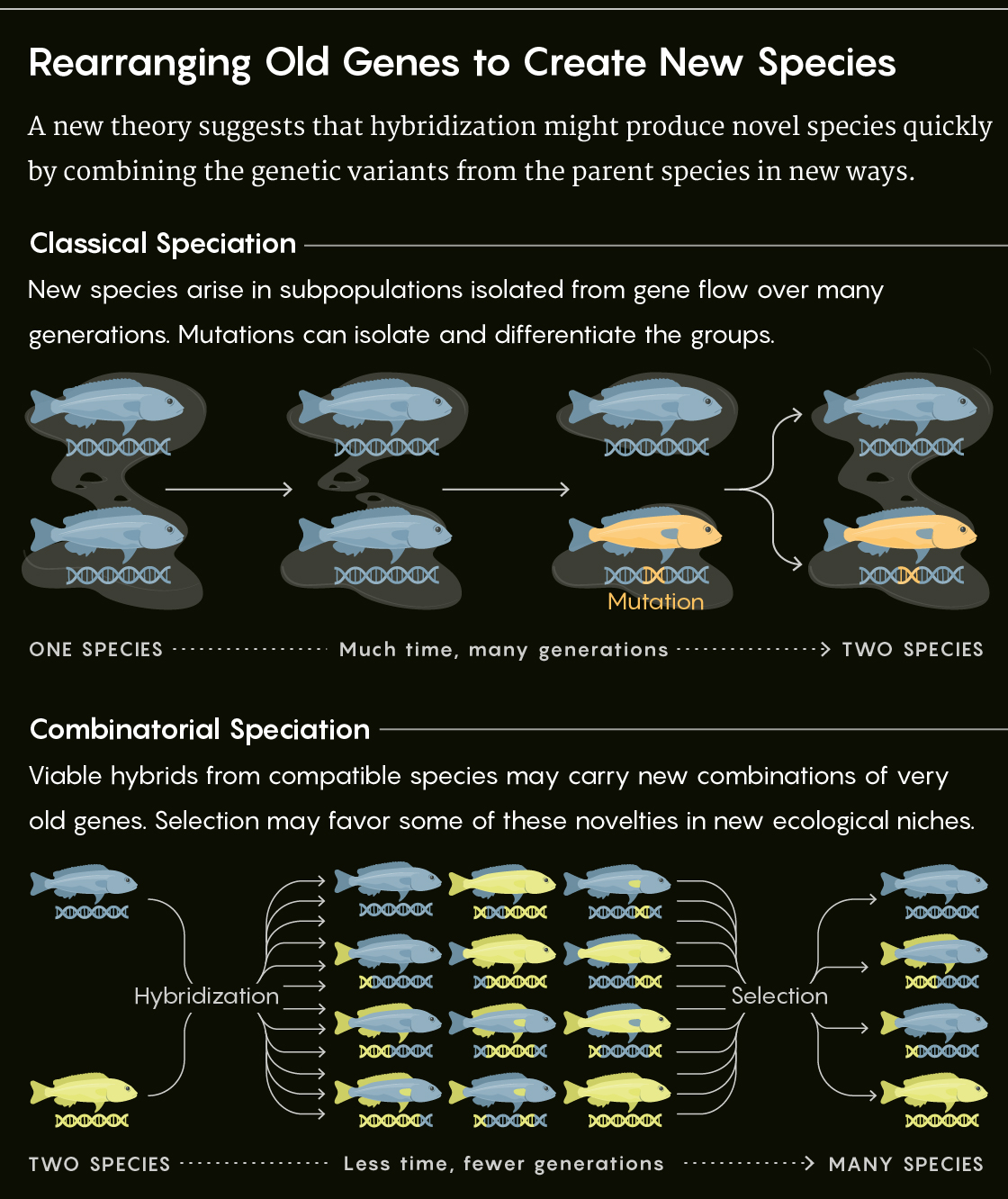
A recent review in Trends in Ecology & Evolution argues that this phenomenon reveals something fundamental about how new species form. Old variants recast in new roles may sometimes be more important role in the origin of species than new mutations are. And hybridization — long considered an evolutionary dead end — instead acts as a catalyst for combining old gene variants in new ways, fueling rapid diversification.
The evolutionary biologists David Marques and Ole Seehausen at the University of Bern and Joana Meier at the University of Cambridge call this new view of the origin of species combinatorial speciation.
“All this new genomic data so clearly shows that speciation in animals and plants often works by building linkages, including between old genetic variants,” Seehausen said. “We need to ask ourselves: Is this a very common mechanism compared to the classical view?”



From top to bottom: Ole Seehausen, head of the division of aquatic ecology at the University of Bern’s Institute of Ecology and Evolution; Joana Meier of the department of zoology at the University of Cambridge; David Marques, a postdoctoral researcher in Seehausen’s laboratory at the University of Bern.
Ole Seehausen, head of the division of aquatic ecology at the University of Bern’s Institute of Ecology and Evolution; Joana Meier of the department of zoology at the University of Cambridge; David Marques, a postdoctoral researcher in Seehausen’s laboratory at the University of Bern.
Courtesy of Ole Seehausen; Joana Meier; David Marques
Classically, the origin of new species is a slow, gradual process dependent on random mutations that build a bigger beak or a sexier song. If this novel trait helps a subpopulation exploit a new ecological niche or makes its members less likely to mate with neighboring populations, a new species may be born. As lineages remain isolated from one another, their diverging genomes accumulate differences that become increasingly incompatible. When speciation is complete, the genomes are so different that they don’t work when combined in a hybrid, producing infertile or unfit “dead ends.”
Yet that is precisely the opposite of what Seehausen and his colleagues say is happening in many rapidly diverging species. Combinatorial speciation, they argue, explains how the genetic novelty for speciation can arise so quickly and is consistent with discoveries that hybridization is more commonplace than previously thought. Other biologists urge caution, however, because we simply don’t know enough about the evolutionary consequences of hybridization across the tree of life. They argue that there are too many known examples of extensive hybridization failing to create new species for anyone to speculate too boldly about the importance of combinatorial modes of speciation.

The Fastest Speciators in the World
The three authors’ views are shaped by their work on one of nature’s most explosive species radiations — that of African cichlid fish. In just 150,000 years, well over 700 species have radiated into a technicolor panoply of shapes, sizes and ecologies. Since Lake Victoria formed 15,000 years ago, about 500 species have diversified within its shores, making it an ideal system for biologists trying to understand the early stages of speciation.
Seehausen started by studying how gene flow erodes differences between cichlid species in the lakes. Species can specialize to thrive at a particular depth, adapting to different light environments and ecological niches along the steep slopes of the rift lakes. Disturbances, like eutrophication or human activity, can literally muddy the waters. Such disruptions obscure normal barriers to mating, causing distinct species (which would know better if they could see clearly) to exchange genes and form a hybrid swarm.

Three meters below the surface of Lake Victoria, the waters are already cloudy. Such murky conditions can help to separate various depths of the lake into distinct niches for multiple species. They also raise the likelihood that species will sometimes interbreed accidentally.
Courtesy of Florian Moser, Eawag
Seehausen believes that this kind of periodic mixing has happened often in the history of this lineage. “I started thinking about what that does to those lineages, and what speciation and re-mixing and re-speciation might do to the dynamics,” he said.
In 2004 he argued that hybridization between relatively distinct but still compatible lineages could inject large amounts of genetic diversity into a gene pool. Through recombination, this ancestral diversity gets sliced and diced into new configurations.
That rapid expansion of genetic possibility gives lineages traction in filling new ecological niches. Given an opportunity, such as a newly created lake, successful new combinations of old alleles shoot off from the hybrid swarm. Not all combinations work, of course, but selection can sometimes be weaker in newly formed environments. That relaxed selection can give a hybrid cichlid that has adapted a new way to scrape algae off rocks extra time to sort out any incompatibilities between its parents’ genomes that might otherwise become a handicap. At the same time, other unique combinations of old alleles elsewhere in the genome might be incompatible with those in other offshoots of the hybrid swarm, buffering the budding lineage from gene flow.
This view of speciation is gaining evidential weight as genomic sequencing reveals the ancestries involved in more speciation events. For instance, a study by Meier and her colleagues published in Nature Communications in 2017 confirmed that the rapid radiation of 500 species of cichlids in Lake Victoria stemmed from a hybrid swarm likely to be older than the lake.
Within the lake, all species descended from a common ancestor, probably within the past 15,000 years. But when the researchers tried to place the Lake Victoria group of fishes on the larger cichlid tree, they fell between two very ancient river species — one from the upper Nile, and one from the Congo River.
According to Meier, some parts of the Lake Victoria cichlid genomes more closely resemble that of the Nile species while others are closer to the Congo one. “They’re a genetic mosaic of these two species that hybridized at the origin of Lake Victoria cichlids,” she said. Those mosaic genomes seeded the ancestral hybrid swarm with enough genetic variation from the parental lineages to fuel the fishes’ rapid spread and speciation.

Same Gene, Different Combinations
This mosaic pattern is apparent in one of the most well-studied genes in cichlids — one that helps them see. The long-wave sensitive (LWS) opsin gene helps tune the eye to different levels of ambient light and is exceptionally diverse within Lake Victoria cichlids. That variety falls into two broad categories: one for shallow, clear water and one for deeper water where red light prevails. Seehausen says that variation in this gene helped African cichlids fill the full spectrum of available niches in newly formed lakes. It also helped to reproductively isolate emerging species, because different genotypes are associated with different coloration patterns in the males, a major sexual signal to females.
Lucy Reading-Ikkanda/Quanta Magazine
The exceptional variation in LWS opsin didn’t exist in a single gene pool until the Congo lineage, which possessed the shallow-water version, hybridized with the Nile lineage, which had the deep-water version. The smashing together of these two versions of the gene, and subsequent rearrangements within them, set off an explosion of new physical diversity within the hybrid population that “appears to have facilitated adaptation to an extreme range of light conditions and visual ecologies,” the authors wrote.
Patterns like this popped up elsewhere in the genome as well. As the researchers investigated genomic regions that had spearheaded the differentiation among the Lake Victoria cichlids, they found new combinations of variants from each parental lineage sorted into the newly emerged species.

Other Kinds of Combinations
The ancient admixture event that prompted the diversification of African cichlids is just one of the ways old alleles can be recombined to help form new species. Combinatorial speciation encompasses the classic mechanism of hybrid speciation that farmers and gardeners know so well: In plants, it’s common for hybridization to immediately create a new species that is reproductively isolated from its parents.
Combinatorial speciation also describes situations in which a large, highly genetically diverse population can sprout numerous offshoots over millennia, forged from new combinations of older rare alleles. That’s what Marques and other researchers say has happened with the three-spined stickleback, a tiny armored fish that lives in both marine and freshwater habitats. Since the last ice age, stickleback have repeatedly colonized lakes and streams across the northern hemisphere. Research shows that key genes associated with freshwater colonization, such as one that controls the reduction in the fishes’ armored plating, are frequently ancient marine variants. Some of these alleles may even predate the existence of three-spined stickleback themselves.
The list of species groups with similar patterns goes on — Darwin’s finches, the apple maggot fly, capuchino seedeaters, Hawaiian silverswords. The scientists who work on these systems have long recognized the potential importance of hybridization in their radiations.
But Marques and his colleagues suggest that the accumulated genomic evidence warrants the introduction of “combinatorial speciation” as a new term to frame future research. The word “combinatorial,” Marques said, seemed to best describe the crucial “generation of new combinations from existing variation, which is really the commonality.” With language that better cuts to the essence of how the speciation is happening, he thinks, researchers will be better equipped to assess the importance of contributions from various mechanisms, including mutations, across the tree of life.
“I think it’s a helpful way of reframing a lot of varied, but related, mechanisms under a catchall term,” said Nora Mitchell, a plant biologist who studies hybridization at the University of Wisconsin, Eau Claire. She says she’d like to see introductory biology move away from describing the evolution of biodiversity as relying solely on the origination of new mutations, and toward a more accurate view that encompasses how existing variation can be co-opted.

Hybridization Is Variable
Some biologists urge caution in assuming that we know what all this hybridization is actually doing. “As a field, our understanding of the extent of hybrid ancestry in the genome has far outpaced our progress in understanding its evolutionary impact,” wrote Molly Schumer, an evolutionary biologist at Stanford University, and her colleagues in a commentary last year in Heredity.
Schumer says she’s excited by the past decade of work that has revealed how extremely common hybridization between species is and how signs of hybridization are “sort of persistent in their genome over long timescales.” But she warns that we’re still in the early stages of understanding the role of hybridization in speciation. “Was hybridization something that was really important in the speciation event, or was it just something that happened in the evolutionary history of these species?”
Her view is shaped by her work on swordtails, a diverse group of freshwater fish. Their genomes show rampant hybridization, but Schumer says the effects of that hybridization differ from those of the previous examples. For swordtails, the outcome of hybridization is generally bad: Because the two parental genotypes can’t coexist in the same genome, the crosses lead to problems, and natural selection weeds them out.
Schumer agrees that combinatorial speciation more or less captures what’s happening in the adaptive radiations outlined by Marques and colleagues. But she cautions against taking these compelling examples as evidence that the phenomenon is widespread. The frequency of hybridization among species in the wild is still uncertain and poorly understood. “We don’t really know the answer to the question about whether this is a useful framework until we have a handle on that,” she said. “But it is really useful and important to have these continued discussions circling back to our fundamental theories and asking are they holding up with new evidence.”
Schumer says it will take a lot of hard work to understand the effect hybridization has on speciation. Scientists have become adept at identifying regions in genomes that are derived from hybridization. “But what is it doing, how is it impacting an organism?” That, she says, is still less clear.
For cichlids, some of those regions, like LWS opsin, undoubtedly helped fuel their radiation. But such examples may be the exception, not the rule. Schumer says that many other lineages mixed about as much as the cichlids did but didn’t subsequently radiate.
“Hybridization is really common, and much of the time it just might be neutral or deleterious gene flow,” she said. But even if we knew how helpful or harmful it is, that wouldn’t enable us to pinpoint its importance in speciation compared with, say, the gradual accumulation of genetic incompatibilities. And while some crosses between species yield hybrids that are viable and fertile, even closely related species sometimes turn out to be highly incompatible. “There is a lot we don’t understand about the genetic interactions impacting hybrids, let alone the interplay between the genetics of hybrids and their environments,” she said.
Evolutionary biologists can find it satisfying when grand, unifying theories seem to suggest themselves from the data. But biology is messy. “These processes may just end up being quite system specific,” Schumer said.
Spot photos of cichlids by Timothy Alexander, Oliver Selz and Ole Seehausen




