Solution: ‘Evolutionary Math and Just-So Stories’
In our August Insights puzzle, we explored the ability of certain population structures to accelerate or slow down evolution. Ever since Darwin’s spectacular evolutionary discoveries in the Galápagos Islands, it has become increasingly clear that the process of evolution is accelerated on island archipelagos. The key geographic feature seems to be that the archipelago should have small islands that are far enough from each other to provide relatively long periods of isolation for small populations of a species, but near enough to allow occasional mixing of populations from different islands. Our first problem explored the mathematical consequences of such a scenario.
Problem 1
The probability (p) that an advantageous mutation will get “fixed” (spread through an entire large population of hundreds or thousands of individuals) is 1 − 1/r where r is the relative fitness conferred by the mutation. Thus, a mutation that increases fitness by 5 percent (r = 1.05) has only a 4.7 percent chance of fixation in a large population. By contrast, a mutation that confers a tenfold fitness advantage is almost certain to fix (p = 0.9), while one that has a twofold fitness advantage has an even chance of fixation (p = 0.5). However, even for the mutation that has only a small fitness advantage of 5 percent, the probability of fixation is much larger if the total population is small. Thus in a population of just nine individuals, the fixation probability of the above mutation rises to about 13 percent.
Now imagine that there is an archipelago with a large island that can support a population of thousands of individuals of a species. The archipelago also contains several tiny satellite islands or glorified rocks that can support just nine individuals each. The individuals on these islands cannot leave their rocky home under normal circumstances, but once in a hundred years, there arises a storm powerful enough to blow an individual from the satellite to the main island. If a mutation with a 5 percent fitness advantage has achieved fixation on one of the tiny islands, how long would it take for there to be an even chance that the mutation would achieve fixation on the large island? (Assume that the interval between two storms is long enough for the mutation to either fix or die out on the main island.)
As Mark Pearson rightly remarked:
This is straightforward. At each storm, the invaders come. There is a 5 percent chance that by the end of the 100 years, the invaders have “achieved fixation.” If they haven’t, there’s another storm, another 5 percent chance at achieving fixation in the next 100 years. And so on. Each storm and follow-up battle of life and death between the two differently-fit sets of individuals is independent. Each of these invasions can be considered to be a Bernoulli trial…
A Bernoulli or binomial trial is a random experiment whose end result is one of two mutually exclusive states — heads or tails, win or lose, or in this case fixation or extinction — under the condition that the probability of each state remains constant every time the experiment runs.
In this case, every 100 years the chance that the mutation will die out without getting fixed on the large island is 0.95. That is, on most such occasions, the fixation process fails. The next fixation “trial” occurs after another 100 years, again with a 0.95 probability of failure. However, because the trials are independent, the probability that both fail is 0.952, which is 0.9025, meaning that there is close to a 10 percent chance that the mutation will have successfully fixed. As we count up the centuries, we just have to increase the exponent. The fewest centuries for which the probability of failure falls below 0.5 (implying a possibility of fixation greater than 50 percent) is 14 (0.9514 is about 0.49). Hence the answer is 1,400 years as Mark Pearson correctly calculated, after Jack Wimberley pointed out the correct interpretation of the question. Timescales on the order of 1,400 years represent a mere blink of an eye in evolutionary terms, so the potential clearly exists for the unique structure of an archipelago to accelerate evolutionary change.
In the second problem, I presented three population structures based on John Rennie’s article “Mathematics Shows How to Ensure Evolution,” which described a mathematical study that used graph theory to show how certain population structures can accelerate or slow down natural selection. The mathematical approach is depicted below.
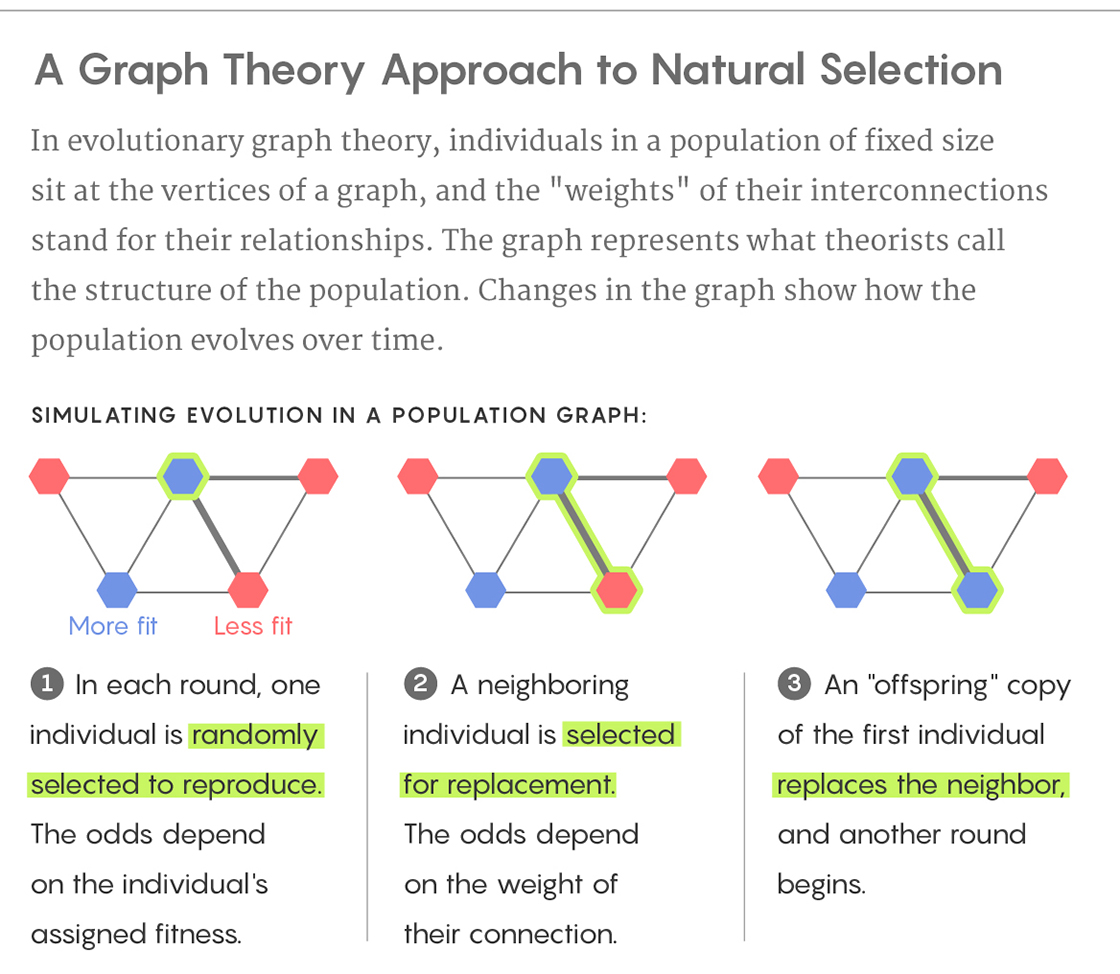
Lucy Reading-Ikkanda/Quanta Magazine
Problem 2
Below are three different population structures that could exist among nine individuals.
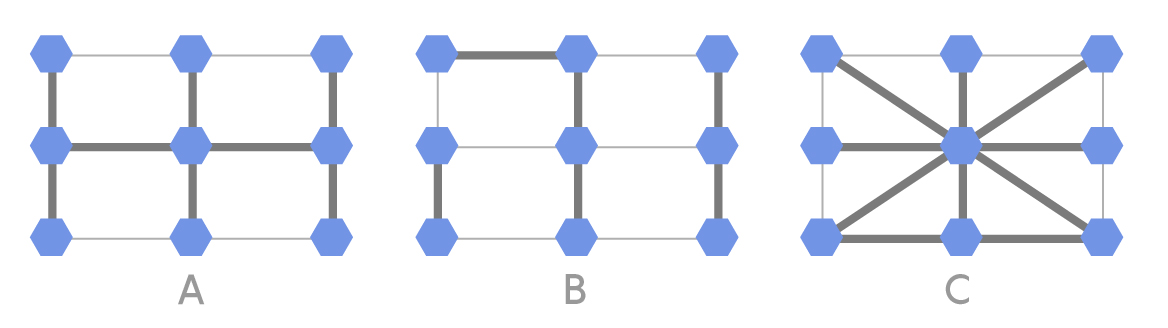
Lucy Reading-Ikkanda/Quanta Magazine
In each of the above population structures, the thicker lines represent strong connections and the thinner lines represent weak connections. One of the nine individuals is initially selected at random to acquire a mutation that gives it a fitness that is 5 percent greater than the others. In each round, one individual is randomly selected to reproduce with a probability proportional to its fitness. Next, one of its neighbors is replaced by an offspring copy of the selected individual. A thick connection indicates a high probability of the neighbor being selected, while a thin connection indicates a low probability. In this process, a normal individual can be replaced by a mutated individual or vice versa. This process continues until the mutation either disappears or all nine individuals are mutated (fixation). For each of the three population structures A, B and C, can you predict if fixation will happen more easily, normally or less easily? Why? What would happen if the thick and thin lines in the configurations were swapped?
Readers who enjoy programming can set up a simulation to see whether the intuitive expectations pan out. Assume that a neighbor with a thick connection to the selected individual has five times the probability of replacement as one with a thin connection. Recall that the fixation probability for a mutation with a 5 percent fitness advantage in a population of nine is about 13 percent. Is the value for any of the configurations significantly greater or less than that?
In setting up this problem, I attempted to mimic the successful population structures shown by the researchers in their published paper. Specifically, the authors suggested that there should be a certain subset of highly connected vertices that they called a hub. The remaining vertices were split by the hub into a number of short “branches.” Structures A and C seemed to have large hubs and small branches, so I was quite optimistic that in these structures the fixation probability of a mutation would increase. But just like Xin Yuan Li, who carried out the giant’s labor of writing the code and doing the simulation, I, too, was surprised that there was no acceleration of evolution in any of these structures at all! Most interesting to me was Li’s intuitive expectation of which structures would be successful, which was precisely the opposite of mine! According to Li:
Both A and C consist of one large component while B can be considered a collection of three small components. It makes sense then that B would have higher fixation probability than A and C. The advantageous mutation would have a greater chance of establishing on one island then propagating to the other islands. Further there are more connections in C compared to A so there are more opportunities for the mutation to be wiped out.
Perhaps the fact that two independent lines of reasoning favored completely opposite structures might explain why no net acceleration of evolution was found. In such small structures it might not be possible to have the “pure” hubs and branches that were described in the far larger and more sophisticated research model. Nevertheless, the results suggest that the kinds of structures required to accelerate or slow down evolution may be quite difficult to obtain in nature.
Such models can stimulate discussion and further research and may eventually lead to a deeper understanding of the evolutionary process. But as nonspecialists who follow cutting-edge scientific research, how much stock should we put in mathematical models of evolution that have not been validated by rigorous empirical data? Here is a partial list of caveats worth applying to all evolutionary claims. I am sure that evolutionary biologists could add many more.
- The conclusions of purely theoretical models, whether qualitative or mathematical, should be treated as plausible and interesting speculations. Evolution is a contingent, historical process. That something could possibly happen does not mean that it did happen.
- Any attempt to generalize the model beyond its restricted domain must be carefully and skeptically examined. The conclusions may be true but may apply only in circumstances so unusual as to have had no historical importance in evolution.
- Conditions under which evolution took place should be shown to be effective in the “environment of evolutionary adaptation,” not under conditions as they are now. Environmental conditions change, and the contribution to fitness by a given trait or mutation will vary over time, as Mark Pearson commented.
- Our minds are naturally drawn toward simple single-cause explanations as we discussed in a previous Insights puzzle column. We should resist the lure of Occam’s razor in subjects as complex as evolutionary biology. Rather, every theoretical model should be treated as a plausible contributor to the actual evolutionary story, which is likely to involve the sum total of many diverse and sometimes contradictory scenarios. Empirical research in evolution should try to assess the degree to which each model contributes to the picture.
- Only rigorous empirical work that shows consistent and mutually supporting results in empirical fields such as paleontology, field biology, biochemistry and genetics can establish the true course of evolution.
- Finally, conclusions about the evolution of traits should apply universally to all or almost all members of a species. This is especially true in elucidating the evolution of human traits. If the traits or their mechanism of evolution are not found across most or all ethnic populations, they may be the results of culture rather than biology. It is worthwhile to try to find exceptions or opposite practices somewhere within the world’s diversity of cultures and figure out why the hypothesized pattern or mechanism was not successful.
So what about the “grandmother hypothesis” that I mentioned in the puzzle — the idea that grandmothering helped humans and whales to live longer by gathering food for their offsprings’ children, thus freeing up their daughters to have more children more quickly? The hypothesis makes sense, is supported by some empirical findings and is certainly suggestive, but after the above discussion, it makes sense to be patient and hold out for much stronger empirical results. Applying the last two points above, for example, we should ask questions about what the anthropological and genetic findings are. Do grandmothers do this in all cultures? At what age on average do the purported benefits of having grandmothers diminish or fade in modern times, and can it be shown that it has increased over evolutionary time? What are the genetic changes that were the immediate causes of the lengthening of the lifespan? There is plenty of work to be done.
It is not easy to answer the question that I posed: “Evolutionary stories like the grandmother hypothesis are easy to construct from mathematical models, but how well do they reflect reality?” Realist commented, “About as well as physics hypotheses constructed from mathematical models do.” I beg to differ. I think single-cause models have a far greater chance of succeeding in physics than in the complicated process of biological evolution. The “soft sciences” are soft because nonlinearity and multifactorial causation run rampant in them, far more so than in the hard sciences.
Thanks to all who contributed. The Quanta T-shirt for this puzzle goes to Mark Pearson. Congratulations!



