Break It To Make It: How Fracturing Sculpts Tissues and Organs
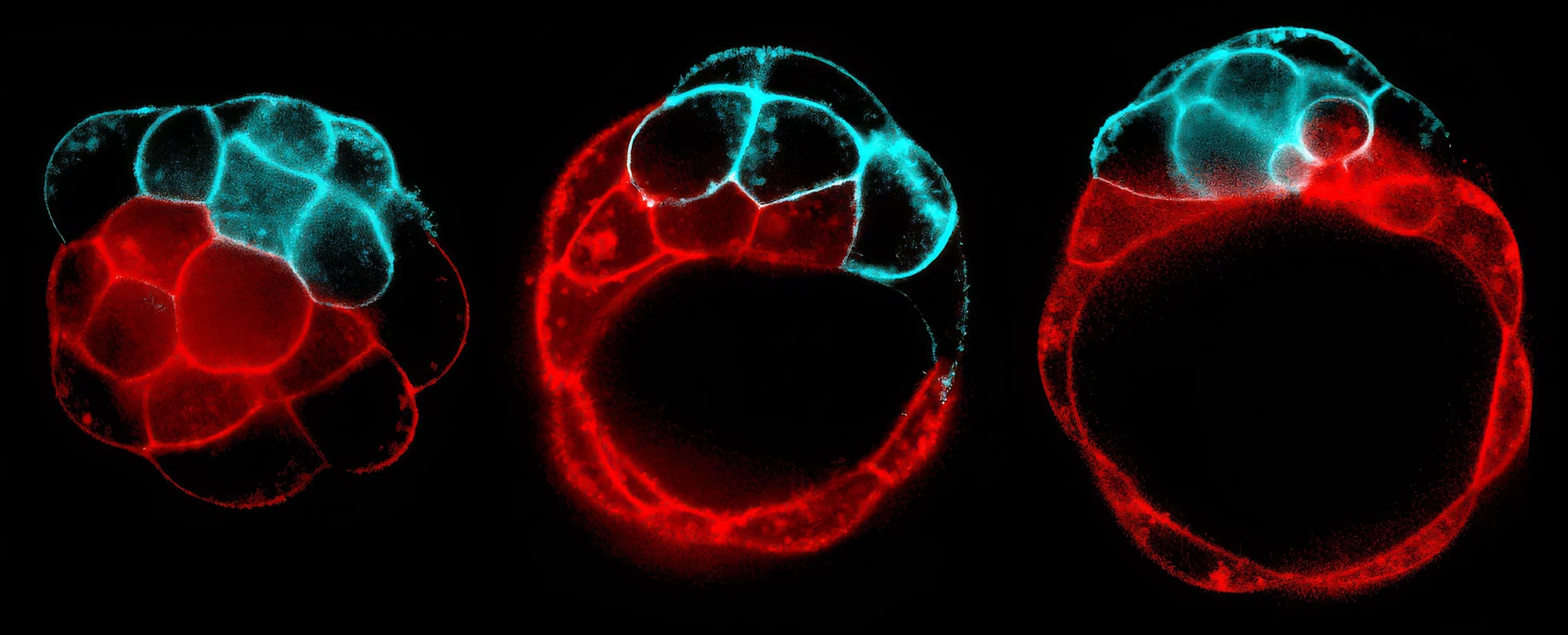
A fracturing process, shown here in stages, creates the cavity of a blastocyst, inside which an embryonic mouse will grow. The fluid inside follows the path of least resistance, preferentially breaking contacts between weaker (red) over tenser (blue) cells.
Jean-Léon Maître
There’s a moment, just before the tight mass of cells that is a developing mouse embryo implants itself in the womb, that it all comes apart.
Hundreds of tiny fluid-filled bubbles expand between each of the orb’s few dozen cells. The bubbles grow and press outward on cell membranes — and then, in a moment of fracture, pry them apart. Thin protein strands tether the cells together as the dissociated embryo floats. Over the course of a few hours, the smaller bubbles empty into larger ones, until the fluid coalesces into a single cavity. With this defining feature, the zygote becomes a blastocyst, ready to embed itself in the lining of the uterus. And inside this hollow ball of cells, reshaped by fracture, a fetus will grow.
“It’s fracturing, but not in a way like you might imagine,” said Hervé Turlier, a physicist at the Collège de France in Paris and a member of the team that characterized this process in mouse embryos. Typically, fractures are fault lines that propagate haphazardly under stress and spread through inert materials, such as ice, rock, or concrete. But the fractures that Turlier’s colleagues observed in mouse embryos display different characteristics. They emerged via a tightly controlled mechanical process, governed by differences in physical tension and cells’ bonds to one another. The fractures were also only temporary: Within hours of splitting, the cells sealed back together again. And these fractures were constructive, sculpting new shapes from developing tissues, in an evolutionary approach that scientists are uncovering across the animal kingdom.
Shaping tissues requires forces — that much has been obvious to the physicists who have peered down microscopes for almost as long as biologists have. From life’s first moments, multiplying cells are squished, stretched, and tugged to form tissues that bend and twist into organs that expand and contract. “All that is obviously involving a lot of mechanics,” said Michel Milinkovitch, a theoretical biologist at the University of Geneva in Switzerland who has observed mechanical fracturing during the formation of African elephant skin.

The biophysicists Hervé Turlier (top) and Jean-Léon Maître (bottom) teamed up to investigate whether a mechanical fracture could be a key step in constructing an embryo.
Patrick Imbert, Collège de France; Mathieu Baumer
When physicists approach biology, their first instinct is usually to take a theory about mechanics “and just apply it” to a biological system, said Marino Arroyo, a theoretical physicist at the Polytechnic University of Catalonia in Spain. But living cells and tissues are far stranger and more dynamic than the lifeless materials physicists typically study. They are filled with fluid, continually rebuild themselves, and actively respond to a complex mix of chemical signals and mechanical forces. “You have to really adapt these theories,” Arroyo said, “because living tissues are very different in many ways.”
Over the past decades, thanks to new precision instruments and advanced techniques that can image cells at high resolution, measure forces, and manipulate tissues, there has been a resurgence in mechanobiology, the study of physical forces in cells and tissues. However, the discovery of how some growing tissues deliberately fracture themselves to make new functional shapes has been a recent surprise. Using custom-built tools and computer models that incorporate decades-old theories from materials science, teams of biologists and physicists are deciphering the mechanisms at play and demonstrating that forces that seem destructive are essential to how some organs form.
A review paper, published in Development in February, compiles examples of constructive fractures in developing tissues of organisms across the tree of life. Although the underlying mechanisms may differ from tissue to tissue, and from organism to organism, the apparent ubiquity of these processes suggests that fracturing is a useful, if counterintuitive, way to sculpt tissues and their vital features. It’s also revealing new machinations of evolution.
“In biology, breaking isn’t always a failure,” said review co-author Rashmi Priya, a developmental biologist at the Francis Crick Institute in London who has watched cracks form in developing zebra fish hearts. “It’s often a necessary step in building something new.”
Tissue Fracking
Years before Turlier and his collaborators discovered how fluid cracks apart a days-old mouse embryo, researchers had a hunch that forces from the flow of pressurized fluids, also known as hydraulics, might be one way living tissues reshape themselves.
In 2015, to model fractures in epithelial tissues, scientists in Barcelona grew a single layer of cells on top of a fluid-filled gel. To their surprise, when they stretched the cell layer by pulling on the gel, it didn’t break apart; that happened only when they released the tension and let the gel relax. The team, led by Arroyo and the physicist Xavier Trepat at the Institute for Bioengineering of Catalonia, realized that the fluid-filled hydrogel behaved like a squeezed sponge: As it contracted, it expelled its water, which pushed between the cells with so much force that the cell layer ruptured.
Since cells of living tissues are brimming with water, just like hydrogels, the physicists “speculated that this hydraulic fracture could play a role in biology,” Arroyo recalled. But they didn’t pursue the idea further.
In 2019, Turlier and his collaborator Jean-Léon Maître, a cell and developmental biophysicist at the Curie Institute in Paris, observed a similar fracturing process in living tissue — mouse zygotes growing in a dish. The researchers imaged them once a minute, much faster than scientists ever had before, starting at the stage just before the zygotes would normally adhere to the womb wall. As Turlier watched, the fluid-filled bubbles ballooned between cells and then shrunk.
He immediately saw similarities to a process known as Ostwald ripening, in which many small particles, bubbles, or droplets join into fewer larger ones. This spontaneous process is the main reason why bubble baths lose their foam over time: Driven by pressure differences, air diffuses from smaller bubbles into larger ones. Similarly, in the mouse embryo, small bubbles between cells give way to large bubbles as the fluid they contain flows along pressure gradients, until only one large cavity, the blastocoel, remains.
“This is something I’ve seen over and over in soft matter physics,” Turlier said. “It took me two minutes to realize the analogy.” It took Maître’s team another year to show experimentally how the process reshapes the ball of cells that is a mouse zygote into a hollow sphere, the blastocyst.
Mark Belan/Quanta Magazine
The duo knew from their previous studies that certain cells in the mouse embryo were tenser and sturdier than others due to differences in their internal scaffolds, which underpin the cell membrane and keep it taut. Taking new measurements, the researchers saw that as fluid gushed between cells, creating indentations in their cell membranes, bubbles mostly bulged into weaker cells.
To test whether those differences affected the formation of the blastocoel, Maître’s lab mixed tenser cells with weaker ones to form chimera mouse zygotes. Then they watched the zygotes become blastocysts. This is a crucial stage of embryonic development, as the final, off-center position of the blastocoel sets the axis of symmetry for the mouse’s back and belly. No matter what, the cavity always formed adjacent to the weaker cells; in other words, the water flowed along the path of least resistance. “The fluid goes … where [cells] deform faster,” Turlier said. Their simulations produced the same result.
Genes might program the initial differences in cells’ tension, but then physics quickly takes over. The mechanics unfold “too fast for the genome to play a role,” Maître said. As fluid bubbles up between cells, it compresses any weaker cells in the zygote and fractures their connections with neighboring cells. Governed by physical forces, the growing embryo seems to have “no other choice” than to put its blastocoel to one side, Turlier said. “It’s extremely robust.”
Breaking so many contacts between cells may seem ruinous for a zygote floating in the womb. But because many small cracks appear between every cell, a single, catastrophic fracture doesn’t rip the embryo open, Arroyo said. The mechanism revealed in days-old mouse embryos “showed that mammals like us are built at [this] moment by fracturing cell-cell junctions,” he said.
Turlier and Maître’s studies of mouse embryos established the idea that cracks can shape organisms. A few years later, close observation of the developing heart would “greatly expand the idea of fracture as a morphogenetic mechanism,” Arroyo said — one that can build pliable and durable tissues and organs.
Heart Breaker
Living tissues can withstand immense forces, but no organ is quite as lively or forceful as the heart. It’s the first organ to form in vertebrate development, and it gets straight to work: beating before it’s fully formed, while it’s still a straight, deflated tube. The zebra fish heart beats about 150 times per minute — or 2.5 times per second — and expands to nearly twice its size and then contracts with each rhythmic pulse. “Just imagine from the viewpoint of an engineer trying to generate a structure that is undergoing such mechanical deformations. I mean, it’s crazy,” said Alejandro Torres-Sánchez, a theoretical computational physicist at the European Molecular Biology Laboratory in Barcelona.
Its force depends on trabeculae, muscular strands that line the heart’s inner walls to help it pump. Without this active mesh, the heart would not beat, and blood would not flow. During her postdoc, Priya, the Crick Institute biologist, discovered that trabeculae are created by mechanical forces that expel cells from the heart wall. But she wasn’t satisfied with existing explanations for why trabeculae originate in the outer curvature, a bulge that forms as the tubular heart twists into shape.
The outer curvature endures great strain: Located directly opposite the valve through which blood enters the heart, it gets hit with incoming blood. Researchers presumed that certain genes were more active in this part of the heart, and that this activity explained the formation of the muscular network of trabeculae. But when Priya and her student Christopher Chan looked, “none of the enzymes were there in the right time [or the] right place,” Priya said. Their absence left the researchers with one alternative to genetic instructions: physics.
Priya and Chan imaged zebra fish hearts as they formed, capturing up to 100 frames per second at various intervals to closely follow any changes in their shape. Just six hours after the heart started beating, they noticed prominent gaps in the outer curvature’s cardiac jelly, a solid network of proteins that props up heart tissue. These gaps spread like fractures, and one day after they appeared, trabeculae strands started to knit their way across them. That timing suggested that these gaps might have something to do with how trabeculae form.
They asked the computational scientist Daniel Santos-Oliván on Torres-Sánchez’s team to develop simulations of beating hearts, which revealed that the gaps in the cardiac jelly were indeed fractures. The model showed that as the heart pulses and takes shape, strain concentrates in the outer curvature, stretching and contracting the jelly scaffold so much that it thins, weakens, and eventually breaks. Sensing those fractures, heart muscle cells at the outer curvature then peel away from the heart wall and fall into the jelly’s newly formed cracks, where they seed the trabeculae. Without the simulations, “we could never have thought that [this process] is controlled by geometry,” Priya said.

Studies by Christopher Chan (left) and Rashmi Priya (right) showed how controlled fracture helps to build a heart that can withstand forces.
Courtesy of Rashmi Priya
To test that hypothesis, the researchers sped up the zebra fishes’ heart rate; more fractures formed in the cardiac jelly. When they slowed the hearts, fewer cracks appeared. This confirmed that the fracturing was a physical process dependent on the incredible strain of heart contractions. To test it another way, the team engineered hearts to grow straight and tubular; when they did so, the fractures changed their direction, too. That behavior is “characteristic of a fracture,” Torres-Sánchez said, and “made us sure that these were [fractures] we were looking at.”
Priya’s team has since seen these fractures in the hearts of chicken embryos. Based on these preliminary findings, “it is tempting to speculate that human hearts might be shaped by similar structural processes,” she said. In any case, their zebra fish work, which is undergoing peer review, shows that a fundamental feature of an essential organ is shaped by mechanical forces ahead of genetics.
The recent revelation that fracturing can be a constructive tool during development doesn’t mean the phenomenon is particularly common. Still, it is widespread in animals: Ruptures, tears, and cracks are known to shape zebra fish nostrils, hydra mouths, fruit fly legs, and whole flatworms. And yet because fracturing seems so destructive, almost antithetical to growth, it has only recently been recognized as a way of shaping tissues, summed up in the recent review paper co-authored by Priya, Torres-Sánchez, Santos-Oliván, and Chan.
Maître suspects that more examples of fracturing will be found now that researchers know to look for it. In recent years, researchers have discovered that, aside from fracturing, living tissues also crumple, buckle, wrinkle, and fold via physical mechanisms that produce incredible diversity with just a few slight tweaks in tissue properties and architecture. “This is really far-reaching,” Milinkovitch said. Mechanics makes evolution “much easier to understand.”