Building Codes for Bacterial Cities

Jeff Sheridan for Quanta Magazine
Introduction
The greenish glow under the microscope might not have rivaled Times Square in its intensity, but to the microbiologist Katharine Coyte, her first glimpse of this bustling city was just as exciting. As a doctoral student at the University of Oxford, she had engineered a small plastic slab studded with holes, like the world’s tiniest slice of Swiss cheese, called a microfluidic chip. It was meant to resemble the porous environments, such as soil, in which many bacteria live, and she had seeded it with different strains of E. coli. She tagged each of the strains with a fluorescent protein to give it a distinctive color, which would allow Coyte to measure how many of each cell type were present. As a steady stream of water brought nutrients to the setup, she stood back and watched the bacteria build a microbial megalopolis known as a biofilm.
Biofilms are everywhere. Far from being rare alternatives to the lone microbe swimming in a flask or sprawled in a Petri dish, 99.9 percent of the simple cells called prokaryotes default to living in close quarters among millions of their compatriots. These biofilms of clustering bacteria can create impossible-to-eradicate infections on central lines and catheters; they foul everything from sewer lines to our teeth. The billions of bacteria that can live in a single biofilm cover themselves with a sticky combination of sugar and protein called the extracellular matrix, which effectively glues them to their surface of choice. Both the matrix and the physical shape of the biofilm protect those cells living at the center of the structure from attack by predators and antibiotics. The size of the biofilm and the interaction of cells within it give the different cells the opportunity to specialize in a particular task, such as acquiring food, subverting predators or acting as a reservoir of genetic material from which to regrow the entire structure.
“We can watch these complex communities in what is essentially their natural habitat and study their structure or metabolism. There’s a lot we can learn when we can study biofilms in their whole freaky native state,” said Melissa Garren, a marine biologist at California State University, Monterey Bay.
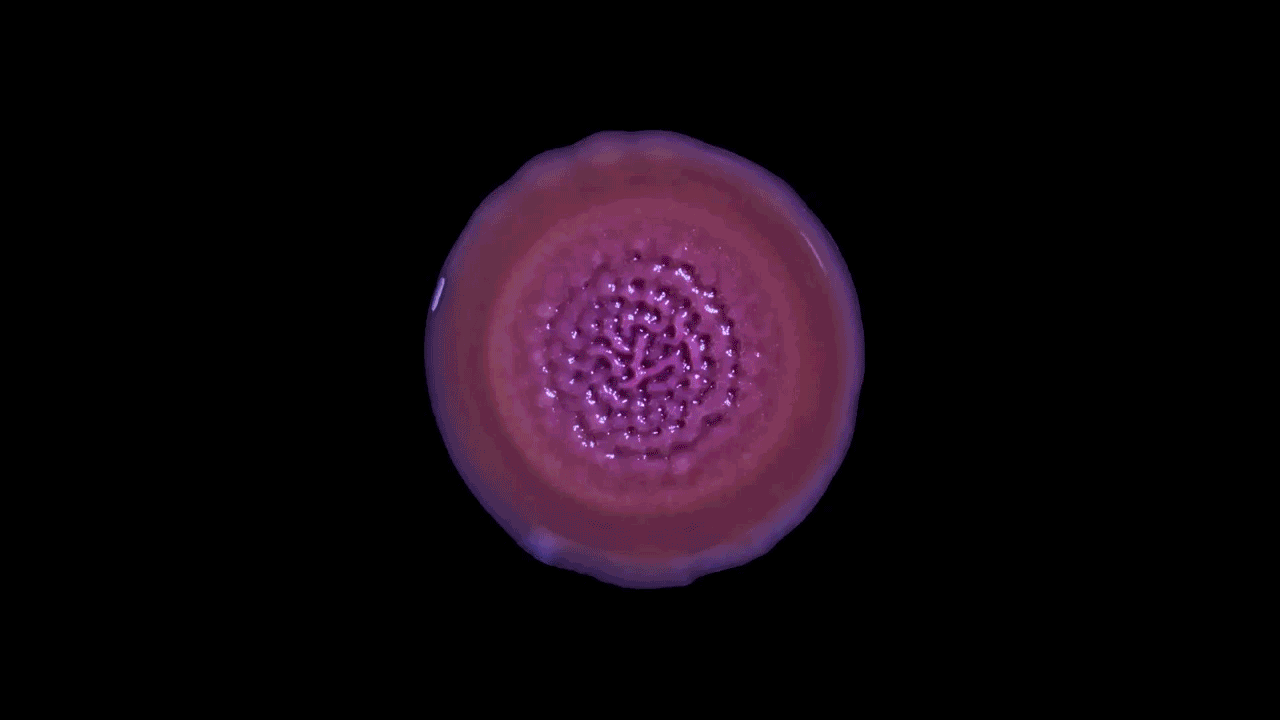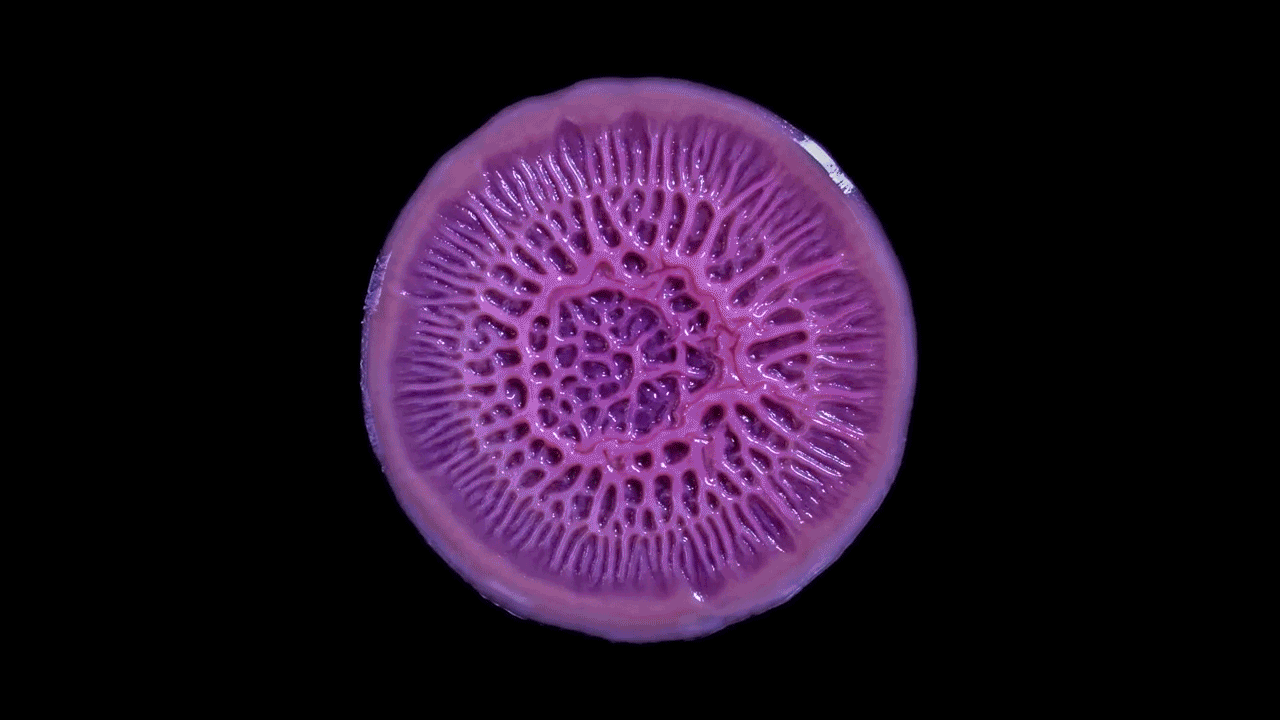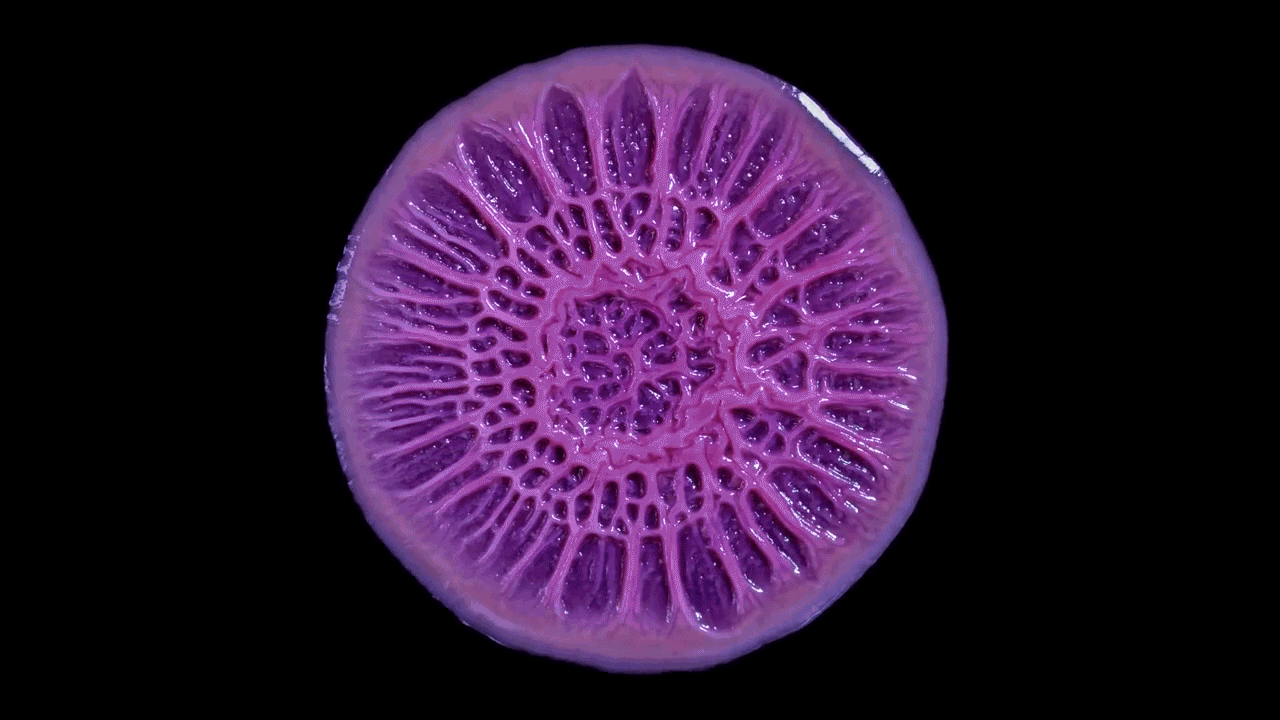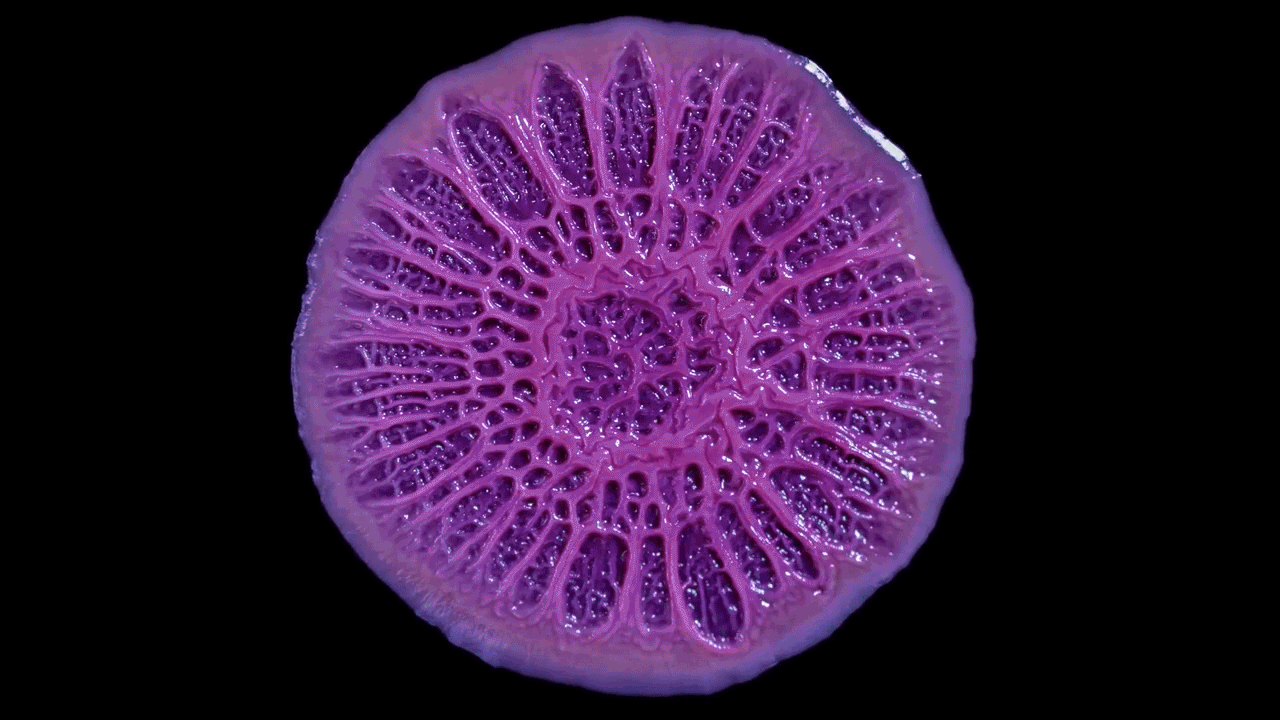
Time-lapse video shows the growth of Pseudomonas aeruginosa as a biofilm on a flat plate. Biofilms can look like simple patches of slime but their dynamic inner architecture reflects the responses of bacterial strains to competition and physical forces.
Scott Chimileski and Roberto Kolter, Harvard Medical School
As a case in point, the microfluidic chip experiments by Coyte, now a postdoc at Memorial Sloan Kettering Cancer Center in New York, and her colleagues illuminated how the flow of water and the number of bacteria present can constrain and reconfigure the growth of biofilms. Their work, published this year in the Proceedings of the National Academy of Sciences, adds to a burgeoning literature demonstrating that physical forces like hydrodynamic shear and the stickiness of the extracellular matrix shape the growth of microbes into complex collectives. These studies provide a big step on the path to decoding the rules that govern the formation and evolution of biofilms.
Those biophysical forces are like universal zoning rules for the biofilm cities: they govern how the inhabitants obtain food and building materials, how they can move and how they interact with one another. Just as urban planners use their knowledge of civil engineering principles and regulations to build better cities for people, microbiologists and bioengineers can use these rules to make objects more or less hospitable to the billions of cells that live in and around us.
On September 17, 1683, Antonie van Leeuwenhoek wrote to the Royal Society of London with news of tiny “animalcules” he had seen under the microscope. From the teeth of himself, two women and two old men who had never cleaned their teeth, Leeuwenhoek had scraped “a little white matter, which is as thick as if ’twere batter,” and looked at it with his hand-polished lenses. His letter described “an unbelievably great company of living animalcules, a-swimming more nimbly than any I had ever seen up to this time,” in what was to become one of the first descriptions of bacteria recorded. Scientists now know that the tooth plaque Leeuwenhoek described was actually a biofilm, not a simple collection of individual bacteria.
Despite the dates of Leeuwenhoek’s early discoveries, it took microbiologists until the 1970s and ’80s to begin investigating biofilms. Early pioneers like Bonnie Bassler of Princeton University identified the chemical signals that bacteria use to take a census of their colleagues in a process known as quorum sensing. Other researchers decoded the genes that synthesized the extracellular matrix and how this matrix could vary between species and even within the same individual.
Important as those scientific contributions were, the studies suffered from a fundamental limitation. What makes a biofilm different from a mass of identical bacteria are the multitudes of interactions among the component microbes. Yet those scientists were largely obliged to observe biofilms growing in Petri dishes or the equivalent. And it became increasingly clear that those simple laboratory conditions could not support the full range of biophysical interactions that shape biofilms and their function.
For example, many of the interactions among the bacteria in biofilms are intensely competitive, both within a biofilm and between them. Fast-growing organisms will snatch up nutrients before their neighbors take over their space. Other bacterial species secrete antibiotics to prevent competitors from elbowing into their territory. To prevent freeloading cells from getting the benefit of extracellular matrix produced at a cost of energy and valuable nutrient resources, some species of bacteria attach themselves to matrix molecules to “privatize” them. The microbes in a biofilm can also specialize at different tasks, depending on where in the biofilm they are located and their genetic background. Cells on the edge focus on acquiring food and warding off predators, whereas those in the core often hunker down and serve as reserves. The cells within also walk the fine line between sharing resources and competing for them. The success or failure of all these interactions depends strongly on precise details of the dynamic physical circumstances in which the biofilms are growing, which can change significantly over a scale of microns.
What results, Coyte says, is a complex cooperative that looks and acts differently from bacteria growing in pure culture. “We have a habit of thinking of bacterial evolution in Petri dishes, which means we’re missing crucial factors on selection pressures based on physics,” she said.
“If you want to understand how a biofilm grows, you need to see individual cells. The resulting architecture and physiology arise from these interactions,” Drescher said.
Even the earliest experiments on the physical forces affecting a growing biofilm challenged existing ideas. Microfluidic chips showed that the texture and shape of the surface to which the microbes attached were far more important than anyone thought, because they controlled the distribution of nutrients and the shape of the resulting biofilm. Similarly, rapidly flowing water can impede the formation of a biofilm because hydrodynamic shear forces can pry off loose cells.
But researchers have found exceptions. In 2008, Wendy Thomas, a bioengineer at the University of Washington, showed that some strains of E. coli have evolved fringelike attachment threads called fimbriae that adhere better in fast-flowing water than in a more torpid stream. Other bacteria create a different type of extracellular matrix that gives them superior sticking strength in areas with high amounts of hydrodynamic shear. The picture becomes only more complex as researchers recognize more layers of physical variables and biological responses as relevant.
This complexity turns out to be essential to understanding the real-world behaviors of many bacteria. Consider the bacterium Pseudomonas aeruginosa, which can cause life-threatening infections in immunocompromised individuals and can form resilient biofilms on medical equipment such as ventilators and catheters. Like E. coli, Pseudomonas strains differ in the type of matrix molecules they make, if they make them at all. In January 2017, Bassler, Drescher and their colleagues showed that, in simple environments, strains that produce matrix always win out over their nonproducing cousins.
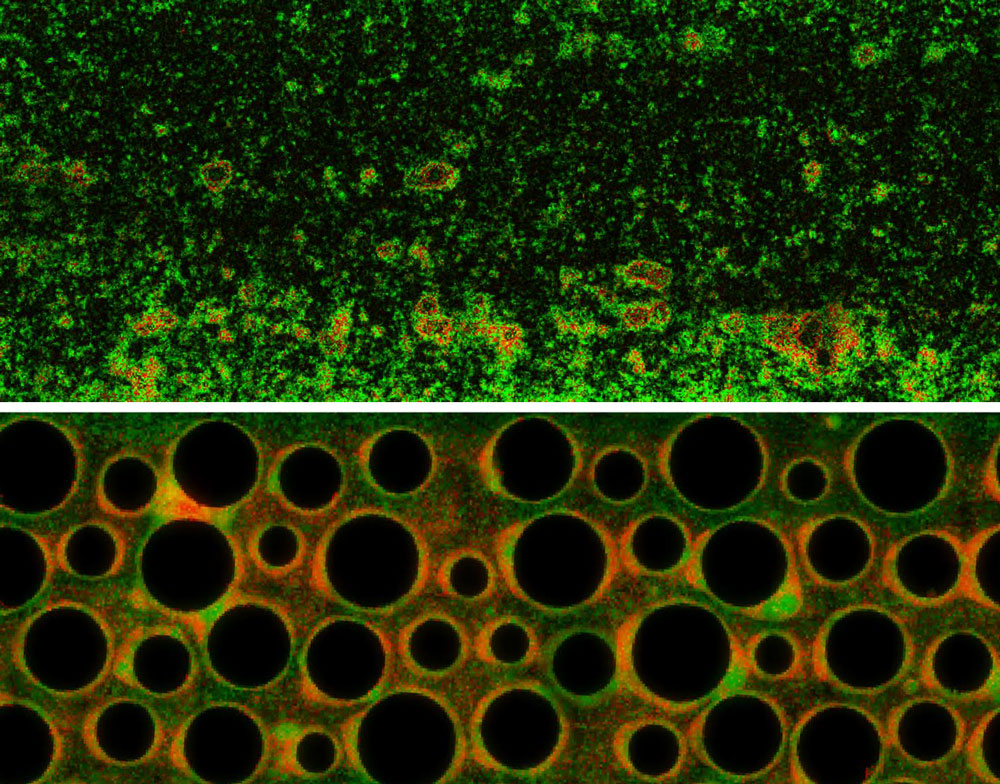
In the top panel, under conditions of uniform hydrodynamic flow, a matrix-producing wild type strain (green) of Pseudomonas aeruginosa grows better as a biofilm than a co-existing mutant strain (red) does. In the bottom panel, the insertion of obstructions (black disks) to simulate a porous environment creates much more complex flow conditions, leading to a more balanced growth of both strains.
Carey D. Nadell, Deirdre Ricaurte, Jing Yan, Knut Drescher, Bonnie L. Bassler, doi: 10.7554/eLife.21855
Then the scientists made the environment more complex, substituting simple linear structures for curved or branching ones. This serpentine network created traffic problems, a growing pain common to all developing regions. At first, the researchers found, the matrix-producing Pseudomonas out-competed their neighbors because they could stick to the surface despite the continuous flow of water. Then the problems began. The resulting biofilm of matrix producers grew so large that it cut off its own flow of water and nutrients, providing a perfect flow-free niche in which matrix-free cells could grow without competition from their matrix-producing counterparts. Water flow and surface microstructure, then, allowed different strains of Pseudomonas to dominate the resulting biofilm.
“Different strains or species of bacteria have different types of interactions in a biofilm, similar to the different vibes you can get in different neighborhoods in the same city,” according to Carey Nadell, a senior scientist in Drescher’s lab and first author on the new study describing this work, which was published this year in eLife.
In the growth of biofilms, the race goes not always to the swift. Coyte’s study in Proceedings of the National Academy of Sciences looked at how more moderate growth could result in greater long-term success in complex environments. Mathematical models Coyte devised supported that hypothesis, so to explore it further, she built the microfluidic chip that mimicked the soil habitat of many biofilms. She tagged each of two strains of E. coli — one that grew into biofilms quickly and one more slowly — with its own fluorescent colored marker to identify them under the microscope.
What she and her colleagues observed was that under conditions of low flow, the slow-growing strains prospered because the fast-growing strains tended to cut off their own access to resources. When flow speeds where high, both varieties often washed away, but the fast-growing strains were more successful at dispersing cells downstream to colonize other locations. At intermediate flow speeds, the results were more complex. As the fast-growing E. coli biofilm got thicker, it shifted the flow of water and nutrients toward its slower-growing brethren. That advantage was not necessarily permanent, however: as the slow-growers’ own thickness began to block the movement of water, the advantage could sometimes shift back to the fast-growing strain. This stabilizing back-and-forth between the strains moderated the overall growth of the biofilm, yet both strains continued to grow and disperse cells.
“Previous studies were descriptive; they weren’t quantitative enough. These studies helped to change that,” says Yang Liang, a microbiologist at Nanyang Technological University in Singapore who co-authored a review of Coyte’s and Nadell’s work in Trends in Microbiology. Even tiny shifts in surface structure or water flow can alter which microbes thrive — if they survive at all.
Antibiotics have worked wonders in fighting many types of dispersed infections, but bacteria growing as biofilms are far less vulnerable and far more resilient. Decades spent throwing ever-more powerful antibiotics at biofilms hasn’t stemmed the death tolls from them. Focusing on the biophysical rules that control these bacterial cities at the microscale might just give scientists a way to halt the urban sprawl and give more humans a fighting chance.
This article was reprinted on TheAtlantic.com.