How a Human Smell Receptor Works Is Finally Revealed
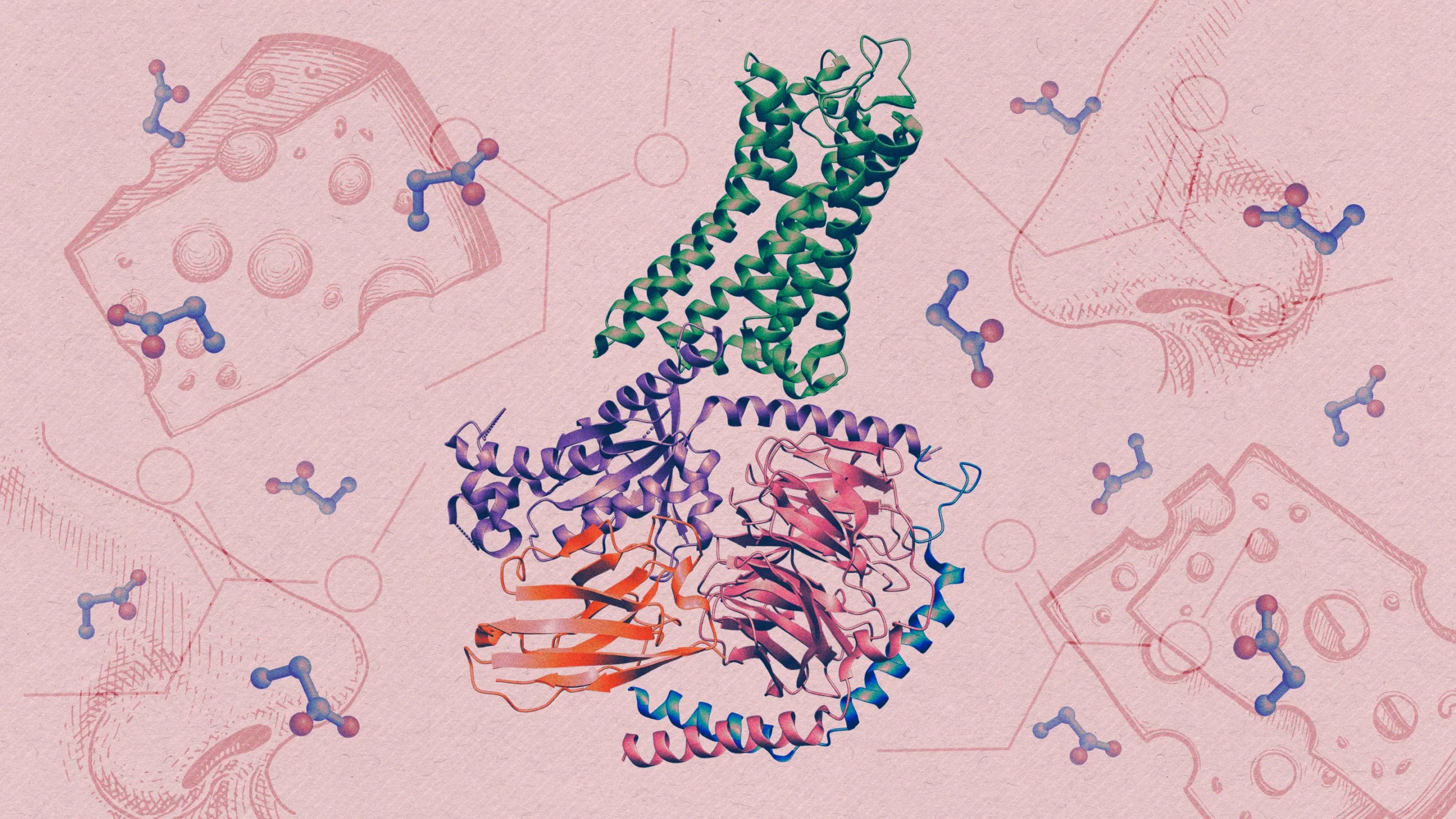
Researcher recently identified how a type of olfactory receptor protein (at center) in the human nose detects airborne molecules of propionate, a component of the smell of Swiss cheese.
Kristina Armitage/Quanta Magazine; Sources: NIH/NIDCD; ArtBalitskiy/iStock; Alhontess/iStock
Introduction
For the first time, researchers have determined how a human olfactory receptor captures an airborne scent molecule, the pivotal chemical event that triggers our sense of smell.
Whether it evokes roses or vanilla, cigarettes or gasoline, every scent starts with free-floating odor molecules that latch onto receptors in the nose. Multitudes of such unions produce the perception of the smells we love, loathe or tolerate. Researchers therefore want to know in granular detail how smell sensors detect and respond to odor molecules. Yet human smell receptors have resisted attempts to visualize how they work in detail — until now.
In a recent paper published in Nature, a team of researchers delineated the elusive three-dimensional structure of one of these receptors in the act of holding its quarry, a compound that contributes to the aroma of Swiss cheese and body odor.
“People have been puzzled about the actual structure of olfactory receptors for decades,” said Michael Schmuker, who uses chemical informatics to study olfaction at the University of Hertfordshire in England. Schmuker was not involved in the study, which he describes as “a real breakthrough.”
He and others who study our sense of smell say that the reported structure represents a step toward better understanding how the nose and brain jointly wring from airborne chemicals the sensations that warn of rotten food, evoke childhood memories, help us find mates and serve other crucial functions.
The complexity of the chemistry that the nose detects has made olfaction particularly difficult to explain. Researchers think that human noses possess about 400 types of olfactory receptors, which are tasked with detecting a vastly larger number of odoriferous “volatiles,” molecules that vaporize readily, from the three-atom, rotten-egg-smelling hydrogen sulfide to the much larger, musky-scented muscone. (One recent estimate put the number of possible odor-bearing compounds at 40 billion or more.)
“In my mind, one of the most amazing things about olfaction is our ability to detect and discriminate such a wide array of volatiles,” said Hiroaki Matsunami, an olfaction researcher at Duke University and an author of the new study.
Caught in the Act
Perched on the surface of neurons in the nose, olfactory receptors change shape when they snag odor molecules. This reconfiguration prompts the neurons to send signals to the odor-processing parts of the brain. Researchers have long sought to see in detail how the interaction between receptor and odor molecule plays out.
A study published in 2021 gave them a glimpse of that process in insects: A group at Rockefeller University determined the structure of an olfactory receptor in the jumping bristletail, as well as the basis for the receptor’s ability to recognize molecules with divergent chemistry. However, that discovery didn’t tell researchers much about human olfaction because insect olfactory receptors work fundamentally differently from ours.

One of the leaders of the new research is Hiroaki Matsunami, a neuroscientist and molecular geneticist at Duke University who studies mechanisms underlying the senses of smell and taste.
Les Todd/LKT Photography Inc.
Human olfactory receptors belong to an enormous family of proteins known as G-protein-coupled receptors (GPCRs). Situated within cell membranes, these proteins contribute to a vast array of physiological processes by detecting all kinds of stimuli, from light to hormones.
Over the past two decades, researchers have determined detailed structures for an ever-expanding number of GPCRs — but not for the olfactory receptors among them. To get enough receptors for these studies, researchers must produce them in cultured cells. However, olfactory receptors generally refuse to mature properly when grown outside olfactory neurons, their natural habitat.
To overcome this problem, Matsunami and Claire de March, who was a research associate in Matsunami’s lab, began exploring the possibility of genetically altering olfactory receptors to make them more stable and easier to grow in other cells. They joined forces with Aashish Manglik, a biochemist at the University of California, San Francisco, and Christian Billesbølle, a senior scientist in Manglik’s lab.
Although this effort was progressing, the team decided to give the extraction of a natural receptor one more shot. “It’ll probably fail just like everybody else has,” Manglik recalled thinking. “[But] we should try it anyway.”
They improved their odds by picking an odor receptor, OR51E2, that is also found outside the nose — in the gut, the kidney, the prostate and other organs. Through Billesbølle’s meticulous efforts, they managed to obtain enough OR51E2 to study. They then exposed the receptor to an odor molecule that they knew it detected: propionate, a short fatty acid produced by fermentation.
To generate detailed images of the receptor and propionate locked together, the interaction that triggers a sensory neuron to fire, they used cryo-electron microscopy, an advanced imaging technique that captures snapshots of proteins that have been rapidly frozen.
The team found that within the structure of the interlocked molecules, the OR51E2 had trapped propionate within a small pocket. When they enlarged the pocket, the receptor lost much of its sensitivity to propionate and to another small molecule that normally activates it. The tweaked receptor preferred larger odor molecules, which confirmed that the size and chemistry of the binding pocket tunes the receptor to detect only a narrow set of molecules.
The structural analysis also uncovered a small, flexible loop atop the receptor, which locks down like a lid over the pocket once an odor molecule binds inside it. The discovery suggests that this highly variable looping piece may contribute to our ability to detect diverse chemistry, according to Manglik.
The Underlying Logic of Scent
OR51E2 may still have other secrets to share. Although the study focused on the pocket that holds propionate, the receptor may possess other binding sites for other odors, or for chemical signals it might encounter in tissues outside the nose, the researchers say.
Also, the microscopy images revealed only a static structure, but these receptors are in fact dynamic, said Nagarajan Vaidehi, a computational chemist at the Beckman Research Institute of the City of Hope who also worked on the study. Her group used computer simulations to visualize how OR51E2 probably moves when it’s not frozen.
For de March, who has moved to France’s National Center for Scientific Research, the map of OR51E2 turned years of speculation into reality. She noted that she has been studying theoretical models of odorant receptors throughout her career: The new findings were “the first time I had the answers to everything I was wondering when I was working on these theoretical models,” she said.
Other human olfactory receptors, especially those closely related to OR51E2, likely function similarly, Matsunami said. He and other researchers see the identification of the functional structure as a step toward understanding the underlying logic that guides the operation of our sense of smell.
But they have a long way to go. Scientists have at best an inkling of which molecules activate only about a quarter of the human olfactory receptors.
Still, with more structures like that of OR51E2, it may be possible to open the biological black box of olfaction, said Joel Mainland, an olfactory neuroscientist at the Monell Chemical Senses Center who was not involved in the new research. With more insights into how the neural coding for olfaction works, “the hope is that now we’ll be able to make confident models about what odors will bind to given receptors,” he said.
The question of how receptors selectively respond to airborne chemicals is only a piece of the bigger puzzle of smell, however. To fully understand the sense, researchers also need to figure out how the brain translates the incoming information about receptor activity into a perception, said Matt Wachowiak, an olfactory neuroscientist at the University of Utah who was not involved in the study.
In the real world, almost everything we smell contains a mix of many chemicals, at varying concentrations. “Somehow we recognize that pattern, generally very quickly, and in different situations,” he said. “The real challenge is figuring out: How does the brain do that?”



