In the Gut’s ‘Second Brain,’ Key Agents of Health Emerge
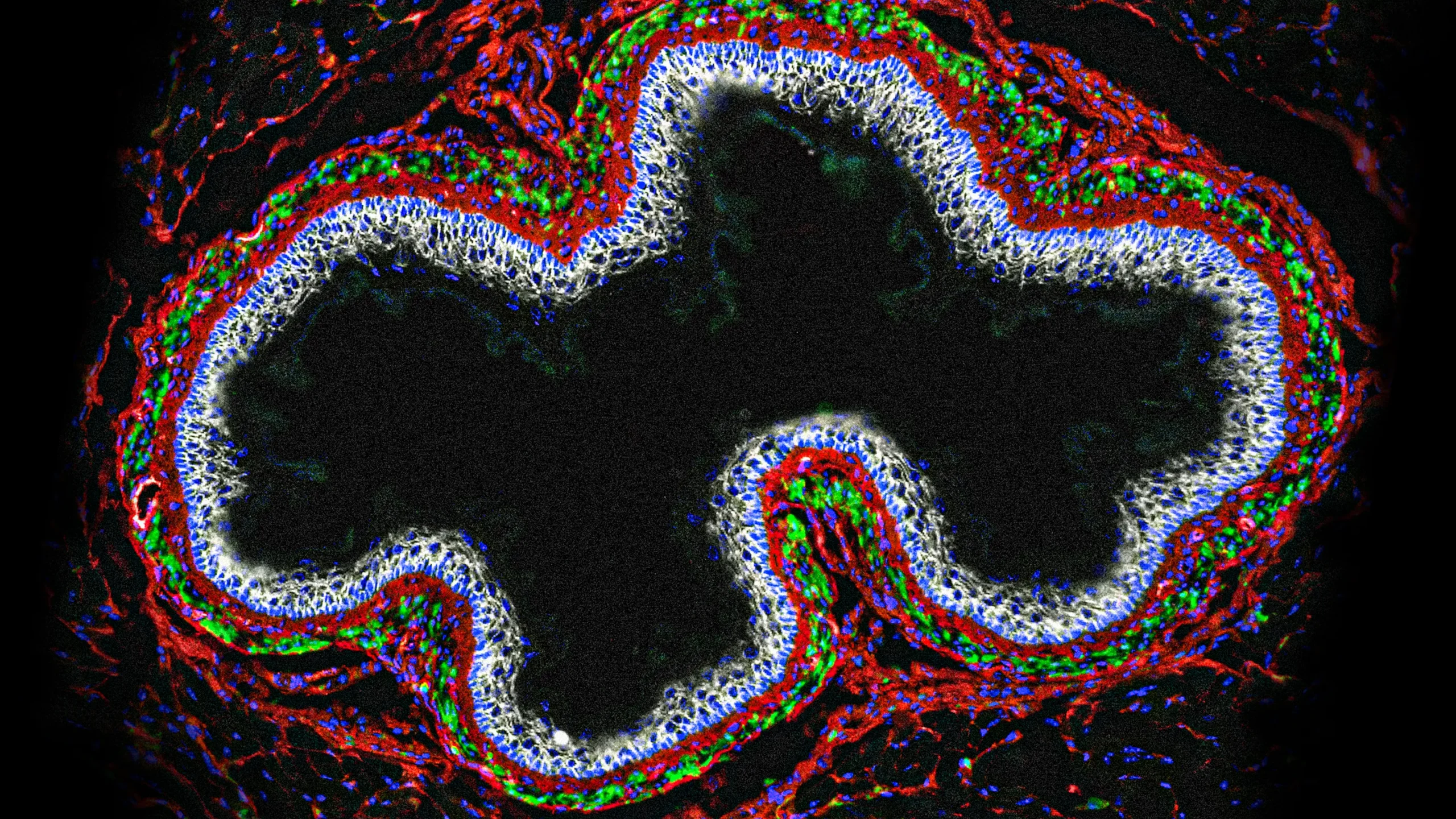
Your gut has a mind of its own. A “second brain” of nervous system cells, including glia (stained green in this cross section of a mouse esophagus), coordinates the movement and digestion of food as it moves through your body.
Marissa Scavuzzo
Introduction
From the moment you swallow a bite of food to the moment it exits your body, the gut is toiling to process this strange outside material. It has to break chunks down into small bits. It must distinguish healthy nutrients from toxins or pathogens and absorb only what is beneficial. And it does all this while moving the partially processed food one way through different factories of digestion — mouth, esophagus, stomach, through the intestines and out.
“Digestion is required for survival,” said Marissa Scavuzzo, a postdoctoral researcher at Case Western Reserve University in Ohio. “We do it every day, but also, if you really think about it, it sounds very foreign and alien.”
Breaking down food requires coordination across dozens of cell types and many tissues — from muscle cells and immune cells to blood and lymphatic vessels. Heading this effort is the gut’s very own network of nerve cells, known as the enteric nervous system, which weaves through the intestinal walls from the esophagus down to the rectum. This network can function nearly independently from the brain; indeed, its complexity has earned it the nickname “the second brain.” And just like the brain, it’s made up of two kinds of nervous system cells: neurons and glia.
Glia, once thought to be mere glue that fills the space between neurons, were largely ignored in the brain for much of the 20th century. Clearly, neurons were the cells that made things happen: Through electrical and chemical signaling, they materialize our thoughts, feelings and actions. But in the last few decades, glia have shed their identity as passive servants. Neuroscientists have increasingly discovered that glia play physiological roles in the brain and nervous system that once seemed reserved for neurons.
A similar glial reckoning is now happening in the gut. A number of studies have pointed to the varied active roles that enteric glia play in digestion, nutrient absorption, blood flow and immune responses. Others reveal the diversity of glial cells that exist in the gut, and how each type may fine-tune the system in previously unknown ways. One recent study, not yet peer-reviewed, has identified a new subset of glial cells that senses food as it moves through the digestive tract, signaling to the gut tissue to contract and move it along its way.
Enteric glia “seem to be sitting at the interface of a lot of different tissue types and biological processes,” said Seyedeh Faranak Fattahi, an assistant professor of cellular molecular pharmacology at the University of California, San Francisco. They’re “connecting a lot of dots between different physiological roles.”
They’re now being linked to specific gastrointestinal disorders and pain symptoms. Understanding the different roles they play in the gut could be critical for developing treatments, Scavuzzo said. “Hopefully, this is like the beginning of the glial-cell renaissance in the gut.”
Glia Do Everything
Scientists have known about enteric glia for over a century, but until recently no one had tools for studying them. Researchers could examine neurons by picking up the action potentials they fire. But compared to neurons, glial cells are electrophysiologically “boring,” said Brian Gulbransen, an associate professor of neuroscience at Michigan State University. Aside from a few reports that pointed to their roles in maintaining healthy gut tissue, they remained under-studied and underappreciated.

To pursue her research documenting the variety of enteric glia, Marissa Scavuzzo of Case Western Reserve University had to develop new methods to work in the gut’s harsh environment.
Jesse Zhan
That changed over the last decade or so. New tools that allow scientists to manipulate gene activity in glia or visualize them in different ways have “dramatically changed the way we look at the enteric nervous system,” said Keith Sharkey, a professor of physiology and pharmacology at the University of Calgary. For example, calcium imaging, a method Gulbransen developed while he was a postdoctoral researcher in Sharkey’s lab, allowed them to analyze glial activity by tracking calcium levels within cells.
Thanks to some of these newer technologies, scientists now know that enteric glia are among the first responders to injury or inflammation in gut tissue. They help maintain the gut’s barrier to keep toxins out. They mediate the contractions of the gut that allow food to flow through the digestive tract. Glia regulate stem cells in the gut’s outer layer, and are critical for tissue regeneration. They chat with the microbiome, neurons and immune-system cells, managing and coordinating their functions.
“We think that they do everything,” Gulbransen said. “The more that people find out about them, it’s less surprising that they do these diverse roles.”
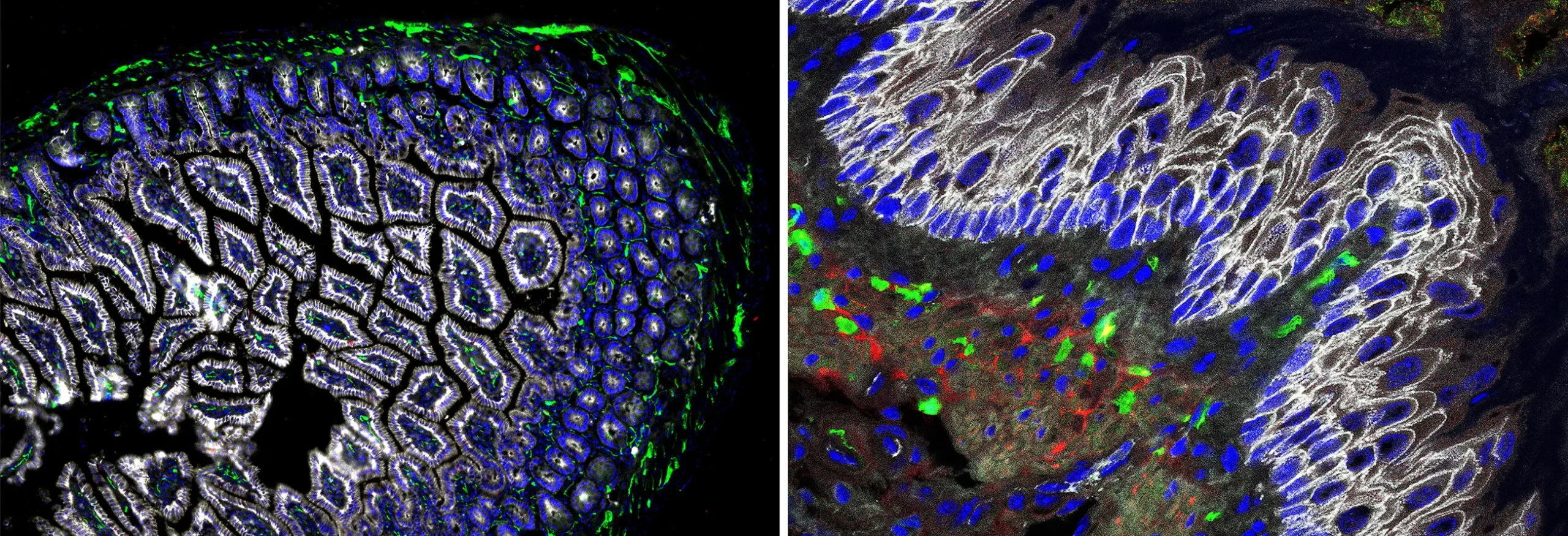
Cellular staining reveals the diversity of cells in the digestive tract. Protective epithelial tissue (stained white) forms the distinct shapes of the wall of the small intestine (left) and mouse esophagus (right). Glial cells (stained red and green) innervate muscle tissue and help coordinate the movement of food through the gut.
Maria Scavuzzo
They can also move between roles. They’ve been shown to change their identities, shifting from one glial cell type to another, in lab dishes — a useful ability in the ever-changing gut environment. They’re “so dynamic, endowed with the functional capacity to do so many different things, sitting in this incredibly fluctuating and complex environment,” Scavuzzo said.
Even as excitement builds about glia in the enteric nervous system, scientists like Scavuzzo have fairly basic questions still to work out — such as how many types of enteric glia even exist.
A Force to Reckon With
Scavuzzo became fascinated with digestion in childhood when she witnessed her mother’s medical troubles due to a congenitally shortened esophagus. Watching her mother go through gastrointestinal complications compelled Scavuzzo to study the gut in adulthood to find treatments for patients like her mom. “I grew up knowing and understanding this stuff is important,” she said. “The more we know, we can intervene better.”
In 2019, when Scavuzzo started her postdoctoral research at Case Western under Paul Tesar, a world expert in glial biology, she knew she wanted to unravel the diversity of enteric glia. As the only scientist in Tesar’s lab examining the gut and not the brain, she often joked with her colleagues that she was studying the more complex organ.
The first year, she struggled massively in trying to map out the individual cells in the gut, which proved to be a harsh research environment. The very start of the small intestine, the duodenum, where she focused her studies, was especially tough. The bile and digestive juices of the duodenum degraded RNA, the genetic material that held clues to the cells’ identities, making it nearly impossible to extract. Over the next few years, however, she developed new methods to work on the delicate system.
Those methods allowed her to get the “first glimpse into the diversity of these glial cells” across all tissues of the duodenum, Scavuzzo said. In June, in a paper published on the biorxiv.org preprint server that has not yet been peer-reviewed, she reported her team’s discovery of six subtypes of glial cells, including one that they named “hub cells.”
Hub cells express genes for a mechanosensory channel called PIEZO2 — a membrane protein that can sense force and is typically found in tissues that respond to physical touch. Other researchers recently found PIEZO2 present in some gut neurons; the channel allows neurons to sense food in the intestines and move it along. Scavuzzo hypothesized that glial hub cells can also sense force and instruct other gut cells to contract. She found evidence that these hub cells existed not only in the duodenum, but also in the ileum and colon, which suggests they’re likely regulating motility throughout the digestive tract.
She deleted PIEZO2 from enteric glia hub cells in mice, which she thought would make the cells lose the ability to sense force. She was right: Gut motility slowed, and food contents built up in the stomach. But the effect was subtle, which reflects the fact that other cells are also playing a role in physically moving partially digested food through the intestine, Scavuzzo said.
It’s possible that each involved cell type could regulate a different type of contraction, she suggested — “or they could just be additional mechanisms that organisms evolved to make sure we could keep digesting our foods to stay alive.” There are likely many fail-safes in digestion because it’s such an important process, she added.

Brian Gulbransen of Michigan State University has published new work showing how glia can contribute to gut pain, suggesting possible new treatments for gut disorders.
MSU College of Natural Science
The experiment offered clear evidence that, in addition to other cells, “glial cells can also sense physical forces” through this mechanosensory channel, said Vassilis Pachnis, the head of the nervous system development and homeostasis laboratory at the Francis Crick Institute. Then, having sensed the change in force, they can shift the activity of neural circuits to trigger muscular contractions. “It’s a wonderful piece of work,” he said.
Hub cells are only one of many glial subtypes that play functional roles in the gut. Scavuzzo’s new six subtypes, added to those characterized in previous research, together reveal 14 known subgroups of glia across the duodenum, ileum and colon. More are likely to be discovered in coming years, each with new potential to better explain how digestion works and enable researchers to develop treatments for a variety of gastrointestinal disorders.
A Pain in the Gut
Gastrointestinal diseases often come with a dose of pain, in addition to disruptive digestive issues. Eating the wrong food, or too much of the right one, can cause a stomachache. Those gut feelings are driven by enteric nerve cells, including glia. Because glia are now known to control the activity of immune cells, they are suspected to play a role in many gastrointestinal disorders and diseases, making them good potential targets for treatments.
Several years ago, Pachnis and his group found that glia are among the first cell types to respond to injury or inflammation in the mouse gut, and that tampering with enteric glial cells can also create an inflammatory response. In the gut glia seem to perform roles similar to those of true immune cells, Pachnis said, and so their dysfunction can lead to chronic autoimmune disorders and inflammatory bowel diseases, such as ulcerative colitis and Crohn’s disease. “Glial cells definitely play a role in the initiation, the pathogenesis and the progression of the various diseases of the gut,” he said.
Glia are likely involved because of their central role in communicating between the microbiome, immune cells and other gut cells. Healthy glia strengthen the intestines’ epithelial barrier, a layer of cells that keeps out toxins and pathogens and absorbs nutrients. But in patients with Crohn’s disease, glial cells don’t function properly, resulting in a weaker barrier and inappropriate immune response.
“Different subtypes of glia can be functioning differently or dysfunctioning in a wide range of diseases and disorders where motility is impacted,” Scavuzzo said. They have also been linked to neural inflammation, hypersensitivity in the organs and even neuron death.
For instance, Gulbransen and his team recently discovered that glia contribute to gut pain by secreting molecules that sensitize neurons. This is likely an adaptive response intended to draw the gut’s attention to damaging substances to dispose of them, Gulbransen said, which as a side effect causes pain.
The findings, published today in Science Signaling, suggest that targeting glia could help alleviate some of the pain created by inflammatory disorders of the gut.
Glia themselves can also become stressed by genetic problems, exposure to metabolites from the microbiome, bad diet or other factors. Fattahi has observed that, no matter the cause, stressed enteric glia influence the entire tissue, and sometimes even damage neighboring neurons or recruit immune cells, causing additional inflammation and pain.
These new studies in enteric glia will go a long way toward explaining many gastrointestinal disorders that researchers have struggled to understand and treat, Sharkey said. “I’m really excited to see how these cells have evolved to become central figures in enteric neurobiology over the years.”
It’s becoming ever clearer that the neuron doesn’t act alone in the enteric system, he added. “It’s got these beautiful partners in glia that really allow it to do its thing in the most efficient and effective way.”
Correction: November 27, 2023
The article was updated in two places to clarify that bile is not acidic.





