Inside the Din, Cells Fight Noise With Noise
For cellphone whisperers, families that live near highways, airports or trains, music-loving commuters, and even individual cells, noise is an inescapable fact of life. In everyday human experience, the nuisance is often manageable. Soundproof barriers and noise-canceling headphones help scrub the cacophony from the sweet sounds of music or silence. But for the cells that make up all living things, noise — meaning random variability in the outside environment, including fluctuating food sources, pathogens and deadly toxins, or random processes within the cells themselves — can be a matter of life and death.
In recent years, scientists discovered that the inside of the cell is a surprisingly noisy place, overturning a long-held assumption that its inner workings are regular and predictable. Molecules within the cell move around and interact randomly, meaning that any subsequent biochemical reactions, such as the production of RNA and protein — needed for nearly all cell activity — also have some element of randomness. How can a cell carry out its jobs — eating, dividing, differentiating — if the machinery that drives those mechanisms is a molecular commotion? The development of an embryo, for example, looks like a well-programmed process, orchestrated by predictable waves of gene activity to produce a specific pattern of tissues. How could it possibly emerge amid this pervasive noise?
“I had no clue how incredibly widespread and pervasive noise is within the cell,” said Michael Elowitz, a physicist turned biologist at the California Institute of Technology. “Now I see noise as a lens through which to look at all kinds of cellular behaviors.”
Most early efforts to study noise focused on how cells deal with this intrinsic randomness, which has side effects that range from annoying to lethal. But as the field evolved, more scientists began to explore how cells can actually use their internal noise to cope with the external noise of an unpredictable environment. Those random bursts of molecular activity can drive all manner of cellular decisions, even the identity the cell chooses to take. Now scientists increasingly ask whether noise is mainly a dangerous enemy that cells must guard against, or if it is sometimes an ally, with potentially beneficial traits that evolution can act upon. It may be that both cases are true. “Noise can be both an obstacle for some types of cellular behaviors as well as a useful feature for others,” Elowitz said.
In microbes, for example, noise-based approaches may help them hedge their bets. Just as investors diversify their portfolio to protect against an unpredictable market, a population of bacteria use noise to vary their methods for dealing with a fickle world.
HIV
The HIV virus may also employ noise as a survival mechanism. A certain percentage of the pathogen becomes silent or “latent” when it infects immune cells. Antiviral drugs can kill the active virus but not the latent versions. But the latent virus can later become active, making the infection almost impossible to eradicate. (That’s why people with HIV have to stay on the drugs for life.)
Weinberger and his collaborators found that noisy gene expression is a major factor controlling the virus’s switch from active to latent, which allows it to hedge its bets. Indeed, “it’s among the noisiest gene expression systems ever reported,” Weinberger said. One of the strategies for dealing with latent virus has been to try to wake up all the dormant copies and then wipe them out with antivirals, an approach dubbed “shock and kill.” But last year, a group from Johns Hopkins University showed that the wake-up process is also random. That means some amount of virus will always remain dormant, creating a major impediment to finding a cure. “This paper really casts a shadow over that dream,” Weinberger said.
Weinberger has proposed that the virus evolved this strategy to enhance its chance of survival, but that’s still a controversial idea. HIV, after all, was born before drugs were developed to treat it. “I think most HIV virologists would say it’s an accident,” he said. To settle the question, Weinberger needs to show that a latent version of the virus is fitter than a version lacking that capacity, something his team is now working hard to do.
A number of studies have shown that random fluctuations in microbial genes trigger some microbes to hunker down into a dormant, drug-resistant state. This phenomenon, known as bacterial persistence, is one reason some infections can be difficult to eradicate. Within a diverse bacterial population, fast-growing microbes are usually better off. But when hit with an antibiotic, the dormant microbes are the ones that survive. “It’s like when the president gives a State of the Union address, and they send one member of the cabinet to an undisclosed location,” said James Collins, a bioengineer at Boston University.
As new examples of beneficial noise come to light, scientists are working to better understand the implications for cell behavior, evolution and human health. “What are the functional consequences of noise?” said Leor Weinberger, a biophysicist at the University of California, San Francisco.
Pinpointing the effects of noise will be important in developing treatments for infections, and perhaps even cancer. “Antibiotic persistence is a great example, where noise has an immediate impact on infectious disease,” Elowitz said. (Persistence differs from antibiotic resistance, which occurs when mutations in microbial DNA, rather than a dormant state, enable them to tolerate drugs.)
Understanding how noise works in the cell could also help scientists explain a puzzling occurrence in genetics whereby some potentially potent mutations don’t affect all the individuals carrying them. Research suggests that random fluctuations in gene activity could explain some instances of the phenomenon, known as partial penetrance, which likely plays a role in some human diseases.
Noise has traditionally been difficult to study in biology, because most biological techniques, such as measurement of gene activity, average the output of many cells, erasing individual variability. But the development of new, high-resolution imaging technology able to capture single cells has finally enabled scientists to reliably chart noise at this level, and to study how it influences behavior. “We’re in the midst of a revolution in biology, the transition from analyzing cell averages to looking at the properties of individual cells,” Elowitz said. “That’s changing our view of lots of things.”
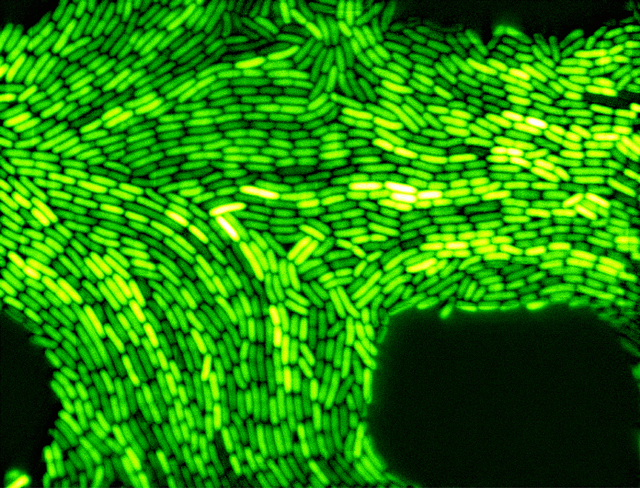
A turning point came in 2002, when Elowitz and his collaborators mapped random gene activity in single cells for the first time. Elowitz’s team had been trying to engineer glowing bacteria that would blink on and off like a Christmas light. The effort worked, but the flashing was erratic rather than regular — even identical cells quickly became hopelessly out of sync, he said. Realizing that biologists had no idea how noisy cells are, Elowitz set out to learn more.
The researchers started with two copies of a gene, which were identical except for the fluorescent color they produced, red or green. If gene activity is controlled in a regular, deterministic manner, then the genes should produce equal amounts of red and green light, which would combine to make a cell glow yellow. If it’s a noisy, random process, however, the two genes’ activity would differ, with some cells producing lots of green and others lots of red, or any number of other variations. The resulting effect, published in 2002 on the cover of Science, is a festive combination of all three colors. “This is what noise looks like,” Elowitz said. “You can see the cell is a non-deterministic machine.”
To figure out whether noise might influence important cellular decisions, Elowitz and his collaborators turned to bacteria, which have an occasional habit of picking up foreign DNA in the environment and incorporating it into their genomes. It’s a high-risk activity with a potentially high payoff. The foreign DNA might contain viral genes that can kill the cell, or it might harbor genetic modules that render the cell resistant to poisons. The researchers theorize that when hit with an environmental catastrophe, such as antibiotics, those cells with extra bits of DNA are more likely to have the tools to survive. Once again, the bacteria appear to be hedging their bets.
Only a small subpopulation of cells are capable of picking up DNA, a state known as “competence,” at any given time, even when the cells are genetically identical and grown under the same conditions. Elowitz and Gurol Suel, then a postdoctoral researcher in Elowitz’s lab, discovered that the decision to become competent is governed by random gene activity, which in turn triggers a positive feedback loop. Elowitz likens it to jiggling the handle on a toilet. Lightly jiggling the handle will sometimes initiate a flush by chance. “In the cell, noise is constantly ‘jiggling the handle’ that controls cell fate decisions, and this causes some cells — randomly — to switch fates,” he said. The study, published in Nature, was one of the first to show that noise could serve a biological function.
In 2009, Suel, by then heading his own lab at the University of Texas, and Elowitz pushed forward again, showing that this noise could influence the cells’ survival. They engineered a less noisy version of the genetic circuit that triggers the competent state and compared both strains. Under a broad range of conditions, bacteria with the noisier circuit are more likely to survive.
“At the time, there seemed to be a prevalent view of noise as a kind of nuisance, as a deficiency of life, at least in comparison to human-engineered systems,” Elowitz said. “I was coming from physics, where people devise clever ways to reduce noise and even more clever ways to utilize noise to make measurements. And so I think that physics background led me to wonder whether cells, like physicists, might have also come up with ways to use noise too.”
Suel, now at the University of California, San Diego, and his collaborators are now trying to figure out how noise influences a community as a whole. “Are cells aware of each other’s noise? If a cell’s neighbor is very noisy, should that cell also become noisy?” he asked. “The cell to cell relationship, and the influence among cells, is very poorly understood.”
The consequences of noise aren’t limited to microbes — noise may also play an important role in development of more complex organisms, including humans. Embryos employ a number of mechanisms to progress from a mass of undifferentiated cells to differentiated tissues. Some evidence suggests that random expression of different genes might play a role. Elowitz’s team is now examining the role stochastic gene expression plays in stem cells, undifferentiated cells that give rise to the different tissues in our bodies. These cells spontaneously switch states, but scientists don’t yet know what triggers that decision. The researchers believe noise could play a role, so they combine single-molecule analysis with time-lapse movies of individual cells to track random activity in many different genes as the cells switch states.
Chance also plays an important role in the development of both our noses and our eyes. The light sensitive cells in our eyes randomly select whether to detect red or green light. Though scientists haven’t figured out how, cells in our nose each randomly choose to produce just one of about a thousand different odor-detecting receptors. The result is an array of cells capable of detecting thousands of smells. “The cell takes advantage of noise to generate diversity without relying on a complex system,” said Claude Desplan, a biologist at New York University.
However, Desplan doesn’t think noise generally drives development. “I think stochastic choice is only needed when you need to increase diversity beyond what you can do in a deterministic manner,” he said. For example, Desplan’s team has studied two different species of fly; in one, the distribution of light sensitive molecules in the eye is random, while in the other it is deterministic (the light sensitive molecules are arranged in regular rows), suggesting that nature can employ both approaches.
Desplan’s contradictory flies illustrate one of the major debates in the field. How important is noise? In contrast to using noise, nature has evolved powerful ways to control it, such as robust circuits that still work reliably in the presence of noise. Noise can be both beneficial and detrimental, but which quality predominates? Desplan favors the latter. “In most cases, noise is a bad thing,” he said. “But sometimes we can take a bad thing and make it a good thing.”
According to Ido Golding, a physicist at Baylor College of Medicine, the idea that randomness might be important in microbes’ development and evolution has been oversold. “I think there have been some examples, but there is danger of over-interpreting them,” he said. “I would guess that 99 percent of the time, the cell is fighting unwanted fluctuations or finding ways to live with those fluctuations.” Golding believes that some of the effects other scientists attribute to noise are actually due to deterministic factors they aren’t yet able to measure. Though it’s clear that there is some level of biochemical noise, “blaming noise every time you see differences among cells is a very dangerous thing,” he said.
In a review published in Science in December, Golding and Alvaro Sanchez, a biologist at Harvard University who studies how yeast might use noise, put aside their differences to explore a question at its very heart. Is noisy gene expression a trait that can be selected for, just like height or eye color, or an inherent property of the gene? “We could either be grumpy and reject each others papers all the time, or write a review to hash out the differences,” Golding said.
If noise is a trait encoded in the sequence of the gene, evolution may be able to fine-tune it, just as the evolutionary process might make a predator faster or a fish a better swimmer. Experiments in yeast suggest that this is the case. “You can have genes with lots of noise or little noise,” said Sanchez. Scientists can tinker with pieces of DNA that control gene activity and change not just how much RNA is produced, but how noisily it’s produced. This question hasn’t been studied as extensively in bacteria. However, some results point in the other direction. Golding’s team has shown that in E. coli, noise is tied to a gene’s activity level, suggesting it is an inherent property of the genome or related cellular machinery.
As the two scientists worked together to review the literature, they began to see each other’s point of view. “The conclusion of our paper is that it’s still an open question, but I used to be much more stubborn,” Golding said. “Now I fully appreciate there is conflicting evidence, which means some of us are wrong.”
This article was reprinted on ScientificAmerican.com.



