The Hidden Mathematical Dance Inside Plant Cells
Introduction
Living on light is a dangerous game. Not only do the sun’s rays carry ultraviolet waves that can snap DNA strands and degrade molecules, but they also vary wildly in intensity. Plants must endure and thrive through soft morning light and blazing summer afternoons, through shade one moment and full sun the next. Their solar calories come in a trickle — or a deluge.
“Think of a cloud obscuring the sun, and suddenly the cloud passes and the sun ray hits a leaf,” said Nico Schramma, a biophysicist at Amsterdam University Medical Center. “Something has to change because the intensity might change a hundredfold.”
Plants aren’t passive. They respond accordingly. They can reorient themselves by rotating their leaves and stems to seek sunbeams or shade, but this mechanism works on a scale of minutes or hours. For finer responses, their cells must mobilize as well. Within every plant cell are chloroplasts, disc-shaped organelles that turn sunlight into sugars. And while plants have to remain mostly stationary, chloroplasts do not.
“Chloroplasts move,” Schramma said. He likened their behavior to that of a flock of sheep seeking shade on a bright day: Intense light similarly shepherds chloroplasts into shaded patches along the cell wall.
“Light is the best friend and worst enemy of chloroplasts,” said Mazi Jalaal, a biophysicist at the University of Amsterdam who supervised Schramma’s doctoral work. “They need it for photosynthesis. But the moment the light intensity goes too high, they have to run away from it.”
Recently, Schramma and Jalaal have obsessed over a mystery of chloroplast physics. How does each organelle balance the plant’s appetite for light with its distaste for too much? And how, in turn, is this expressed as patterns within a cell? In fall 2025 in the Proceedings of the National Academy of Sciences, they reported that the chloroplasts in Elodea, a common aquarium waterweed that they use as a model plant, self-organize into a sort of mathematical optimum. They pack their cell’s surface densely enough to absorb ample light while populating the cell sparsely enough to meander and hide efficiently when they need to.

The biophysicist Nico Schramma recently solved a math problem tucked into a plant cell: Do sunlight-harvesting organelles pack optimally?
Courtesy of Nico Schramma
“The beautiful thing we see here is what a great designer evolution is,” said Dakota McCoy, an evolutionary biologist studying photosynthesis at the University of Chicago. “When you see something fit your simulation really, really well, like this paper does … is it a coincidence, or is it that it evolved to be that way?”
Natural Math
Jalaal’s path to this field sounds like a punch line. His mother taught high school biology. His father, high school physics. The family debated which direction the teenager would go in his career. Voilà, biophysics.
“[Our lab’s] questions always are biology questions,” Jalaal said. “Often you immediately find out that you must solve a physics problem to solve those biological questions.”
Plants provoke many such questions. In 2021, as he and Schramma ping-ponged ideas for a new project, they learned about Elodea densa. Elodea is rather unextraordinary: a staple in home aquariums and middle school science classes. In its favor, the waterweed has simple leaves, which are easier to inspect under a microscope than those of land plants. For the researchers, this offered a shortcut to new questions. “There are so many physics problems that these organisms have to solve during the course of evolution,” Jalaal said.

In his lab at the University of Amsterdam, Mazi Jalaal uncovers fluid dynamics in living organisms, including plants and algae. “There are so many physics problems that these organisms have to solve during the course of evolution,” he said.
Courtesy of Mazi Jalaal
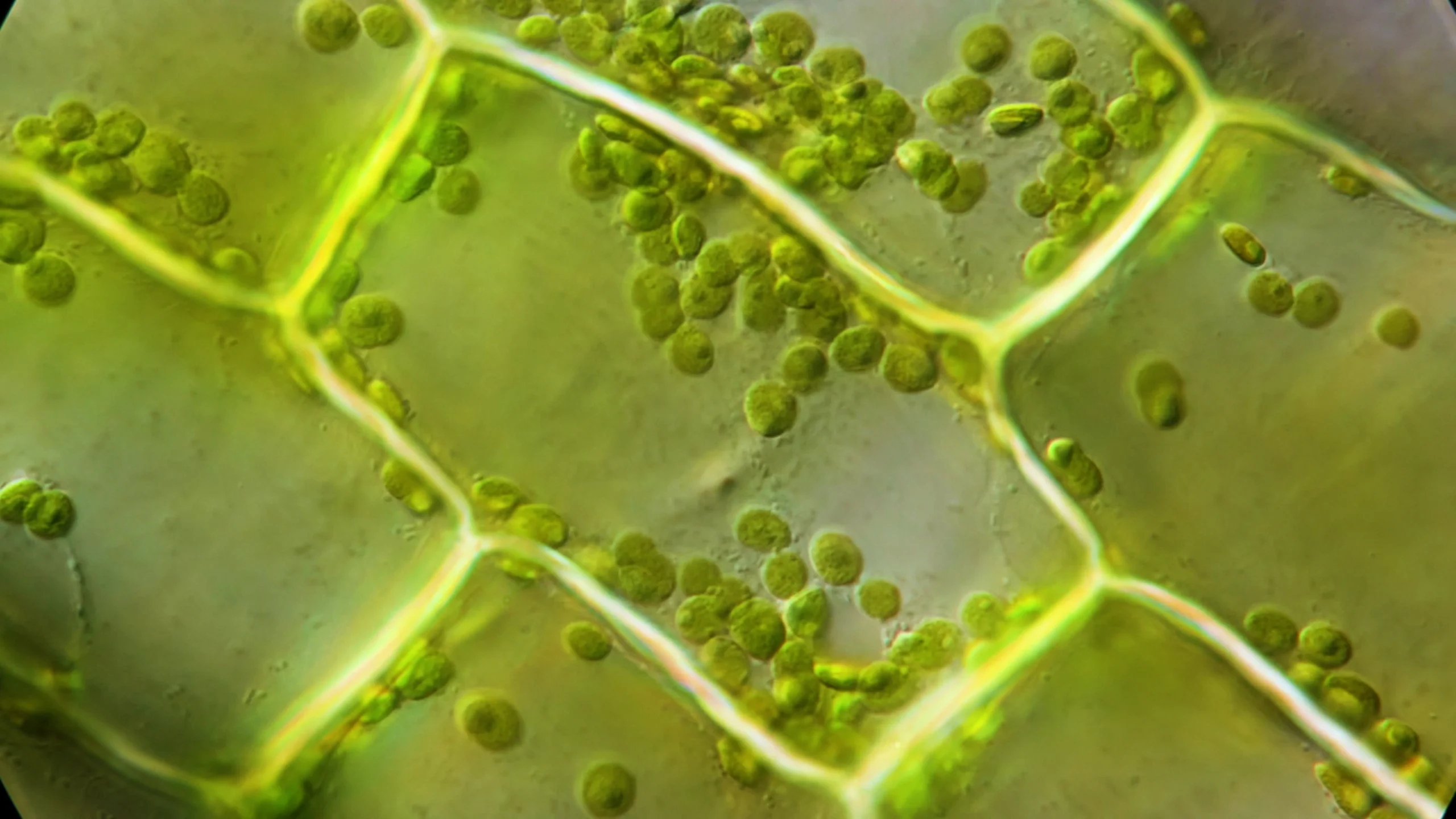
After watching some YouTube videos, they ordered the plant online. Schramma pinched off some centimeter-long leaves. Under the microscope they resembled a brick wall of rectangular cells. Chloroplasts visibly freckled each cell from end to end. He soon made the observation — familiar to plant biologists — that chloroplasts grow in a medley of sizes and fan out to collect light or huddle against cellular side walls to shield themselves from it.
“It is striking when you look at them on the microscope,” Jalaal said. He wondered how the uneven blobs manage to organize so neatly under different light conditions.
They learned that few plant biologists had studied chloroplast motion in great detail. Two trailblazers — Roger Hangarter from Indiana University and Masamitsu Wada from Tokyo Metropolitan University — were retired. Wada had begun experimenting with chloroplast motion back in the 1990s by looking into how the organelles move or anchor themselves by sprouting cytoskeletal fibers, including actin and microtubules. Hangarter had studied these mechanics, as well as the geometry of chloroplasts, in dozens of different plant species.
“Protection’s a real important thing,” Hangarter said. “If you can’t move your chloroplasts, they’re likely to get some damage, and then you’ve got to expend energy repairing them. It could even kill the plant.”
For every possible biological inquiry, Jalaal found, the physics involved was under-explored. Physicists know that small units of all sorts can self-organize into large, complicated structures from simple interactions — a phenomenon known broadly as emergence. Picture murmurations of starlings staining the sky like ink drops or flocks of sheep flowing like liquid. Self-organization intersects with mathematics in so-called packing problems, which date back to Johannes Kepler. The 17th-century astronomer, best known for his studies of planetary motion, proposed that the densest way to stack spheres is to arrange them in a pyramidal pile, the way you’d set out oranges in a grocery store (in two dimensions, this problem is more like arranging pennies on a flat tabletop). This might seem obvious, but proving it was quite challenging, especially for higher dimensions or other shapes.
Plant cells must solve their own packing problem, Jalaal realized: A handful of discs in different sizes must organize and pack themselves efficiently in both bright and dim sunlight. When he looked at Elodea cells under the microscope, he saw chloroplasts packing the cell evenly. This suggested a sort of “computation” occurring in the plant, Jalaal said. “Even before going to how chloroplasts move, [the microscope image] was a clear sign there’s so much happening here.”
Chloroplasts evolved long before plants did. Around 3 billion years ago, a bacterium evolved the chemical tools to feed on light. Then, a billion years later, another cell swallowed one without killing it and took the bacterium’s light-harvesting tools for itself. This primordial gulp, creating a condition known as endosymbiosis (also thought to be the origin story of mitochondria), is believed to have happened just once. But the hybrid cell it created was so fit that its descendants blossomed into every alga and plant species on Earth. “We think of symbiosis as something like a clown fish and an anemone, but a plant cell with its domesticated chloroplasts is also,” McCoy said.
Around the early 1800s, microscopists noticed chloroplasts ambling around plant cells; decades later, they observed the organelles reacting to light. Some 200 years after that, Schramma earned curious looks from neighboring physicists as he carried a second fish tank into his lab.

Compared to land plants, Elodea densa has simpler leaves that are easier to inspect under a microscope.
Courtesy of Nico Schramma
Packing and Moving
A plant cell is a crowded venue. Much of its inner volume is taken up by a fluid-filled blob — the central vacuole — that exerts pressure on other occupants. Caught between a vacuole and a hard place, the chloroplasts, nucleus, and other organelles are smushed against the plant cell’s rigid rectangular walls.
How might a chloroplast elbow its way through the crowd? Hangarter and Wada had debated the possible mechanics: Maybe the cytoskeleton of protein filaments ferries it about, maybe the chloroplast buzzes along randomly, maybe a mix of both. From the biophysical side, Jalaal and Schramma wondered how a plant cell can organize the movement of its chloroplasts, which its survival depends on.
Their answer: glassy behavior. A so-called glass transition occurs when a material can reversibly move between solid and liquid states without undergoing a full phase transition, the way a gelatinous glob of chicken stock melts with a little heat and solidifies again when cool. A similar transition is known to happen in some bacterial cells. In 2023, using a combination of experiments and mathematical modeling, Schramma and Jalaal reported that Elodea cells occupy a critical point at such a transition. When light conditions are constant, the cell is stable and firm, and chloroplasts remain in place. When a change in light intensity prompts activity, the dense cellular contents behave more like a liquid. To hide from light, some chloroplasts even tuck themselves behind others in a 3D cluster along the cell wall — an arrangement that’s only possible if the cell interior becomes more fluidlike first.
But that explanation left questions open. Differences in the size, shape, or density of chloroplasts in the cell could steer the system away from a glassy transition. “Why this density? Why this shape?” Schramma said. “Does it mean anything?”
Jalaal and Schramma reframed their question as a packing problem. The chloroplasts are discs that can come in various sizes and quantities in different cells. Their container, a rectangular cell, can grow to virtually any length and width — but must allow chloroplasts to pack when lights are dim and hide along its walls when light shines brightly. Given those constraints, they asked, what is the relationship between the plant cell’s size and shape and the chloroplasts’ size and number?
Working with the soft matter physicist Eric Weeks at Emory University, they borrowed an algorithm from theoretical physics. They programmed 30 to 130 discs, which varied in diameter, to scatter around a two-dimensional rectangle of fixed length and width. At first, the discs collectively covered 1% of the box. The simulation enlarged first one disc and then another by the same growth factor, and so on. The process continued for many rounds — discs grew, empty space shrank, the growth factor tightened — until the discs could no longer grow. At that point they were as densely packed as they were going to get.
After 30,000 simulations under a slew of parameters, the model predicted an optimal geometry for the cells to maximize both light absorption and avoidance. At this cellular size and shape, chloroplasts would have the space to pack optimally in a single layer to maximize light absorption — filling a cell’s exposed surface area between 70% and 80% — while still having enough room to shuffle away to the walls to avoid the light when they needed to.
But this was just theory, a perfect world imagined and solved on a laptop. To prove out the findings in real biology, Schramma stuck his arm back into the fish tank, nipped more leaves, and snapped microscope images of the cells within to measure Elodea’s actual packing fractions. The real data matched his predictions almost perfectly.
Schramma couldn’t believe it at first. “I ran a couple of times into Mazi [Jalaal]’s office like, ‘I made a mistake!’ Then, 10 minutes later: ‘Oh, actually, no!’” he recalled. “I was obsessed with checking.”
The chloroplasts were indeed packed in a mathematically optimal way. The Elodea cells were small enough for the particular assemblage of chloroplasts within to pack themselves tightly to absorb maximum light, and large enough for chloroplasts to maneuver around one another to avoid it when needed.
When Schramma considered what the result meant for still-growing cells (which change shape), he concluded that cells would maintain optimal packing only if they grew in a single direction. Turns out, that’s how Elodea cells grow.
Selection or Serendipity?
Finding a pattern in nature is one thing; proving that it is an evolutionary product is another. Either chloroplast packing evolved to optimize light absorption and avoidance as the researchers suspected, or it arose coincidentally out of adaptations to other pressures they face.
“I’m very convinced that efficient packing is itself the product of evolution,” said McCoy, who was not involved in the study. “The hand of natural selection is brutal. If you’re not efficient enough or not good enough at your job, it’s pretty easy to go extinct.”
Schramma himself is cautious about this conclusion. Ruling out a coincidence would require proof that a leaf prioritizes the versatility it gets from mobile chloroplasts of a particular size. Some such evidence appears to exist.
In a 2023 study, the experimental botanist Katarzyna Glowacka grew tobacco variants with different-size chloroplasts at the University of Nebraska. She expected an army of small chloroplasts to make leaf cells more agile photosynthesizers. It didn’t. In fact, getting tobacco to shrink its chloroplasts was surprisingly difficult. “It’s almost like the plant was resistant,” Glowacka said. “Almost like there is something stopping them from getting smaller.” Ultra-small chloroplasts could be weak collectors of light and carbon dioxide, or they may be crammed too tightly, like the contents of a large home stuffed into a studio apartment. She concluded that the natural size of tobacco chloroplasts was optimal.
To Jalaal, a firmer conclusion will require looking at these traits in other plants. The answer may differ from species to species, and among other photosynthesizers such as algae. Elodea’s solution to its chloroplast problem could be widespread — or totally unique. The only way to know for sure is to look.
“In physics, we search for universality,” Jalaal said. “In biology, we embrace non-universality and biodiversity.”