What Physical ‘Life Force’ Turns Biology’s Wheels?
Kristina Armitage/Quanta Magazine
Introduction

You’re the earliest known life form. There’s no food around right now. It would be great to go somewhere else. But you’re stuck. Really stuck. At your size (a couple of microns), water feels like tar, or rather, it feels the way being stuck in tar will eventually feel to a human. What do you do?
[One or more billion years later.] You’ve found the perfect solution.
Literally perfect.
“You can assume the system is working optimally,” said Aravinthan Samuel, a biophysicist at Harvard University.
Evolution has created the flagellar motor, a combination propeller/brain that enables single-celled bacteria to move toward food sources. It’s an electric motor that rotates at several hundred revolutions per second — faster than the flywheel in a race car engine — to twirl a tail-like flagellum that pushes the cell along. When the flagellar motor rotates counterclockwise, it propels the cell through the water 10 or more times its own length in a second. The motor can also rotate clockwise, causing the cell to tumble about randomly. This amazing, self-assembling, signal-processing, direction-switching molecular machine is so powerful yet so spare that, billions of years later, it’s still used by bacteria in virtually every gut and puddle on Earth.
Since the discovery of the bacterial flagellar motor in the 1970s, biologists and creationists alike have marveled at its design like medieval architects staring with awe at the dome of the Pantheon built by their Roman ancestors. It’s hard to fathom the level of engineering achievable by a billion years of bacterial evolution, especially with only 20 minutes between cell generations, which allows for a truly astronomical number of mutations and trial runs. Creationists hold up the bacterial flagellar motor as a prime example of intelligent design — specifically the concept of “irreducible complexity,” a biological system so intricate, they say, that it couldn’t possibly have arisen in stages through the gradual, stepwise process of Darwinian evolution.
Over the past few decades, scientists have toiled to unravel how the flagellar motor works — namely, how it rotates and switches directions.
Now they finally have. A wave of studies since 2020 has cracked the molecular structures of the flagellar motor’s parts, including, most importantly, the small cogwheels that turn the larger cogwheel at the flagellum’s base. The final pieces of this dynamic puzzle fell into place as recently as March 2026.
“My lifelong quest is now fulfilled,” said Mike Manson, a professor emeritus of biophysics at Texas A&M University who started studying the flagellar motor in the 1970s. “I finally understand how this thing I’ve been studying for 50 years actually works. That’s about as satisfying as can be.”
The workings of the flagellar motor are ingenious indeed. But when I began interviewing these scientists about what they’ve figured out, I didn’t anticipate that the explanation of the motor would clarify all of biology for someone like me, who seeks mechanistic, physical explanations. The machine, I learned, exploits a driving force I had not known about (though biophysicists have) — the physical “life force” that powers processes in cells. This “proton motive force” doesn’t just turn the cogs of the flagellar motor; it’s the juice we all run on.
![]()
The flagellar motor was discovered by the late Howard Berg, an ingenious experimenter who spent most of his career at Harvard. Berg set out in the early 1970s to apply his training in physics to understanding how bacteria move. The problem was that, under a microscope, Escherichia coli, Salmonella, and other motile bacteria almost instantly swam out of frame. So Berg invented and built an automatic tracking microscope that could keep a bacterium in view as it moved around. “What it recorded were all the corrections that had to be made to the microscope stage in order to keep the bacterium in place, and that of course gives you a readout of what the path of the swimming bacterium was,” said Manson, who joined Berg’s project as a postdoc in 1975.
The data revealed that bacteria “run and tumble” — that is, they switch back and forth between swimming straight and rolling around chaotically. Berg theorized that bacteria change their swimming state based on the chemical gradients sensed as they swim. Their default behavior is to swim straight. If the concentration of sugars and other nutrients is increasing, the cell keeps going forward. If the concentration drops, it tumbles; reoriented in a new direction, the bacterium then resumes swimming straight. This process keeps the bacterium in the vicinity of harvestable molecules, which it absorbs through channels in its cell wall and membrane.
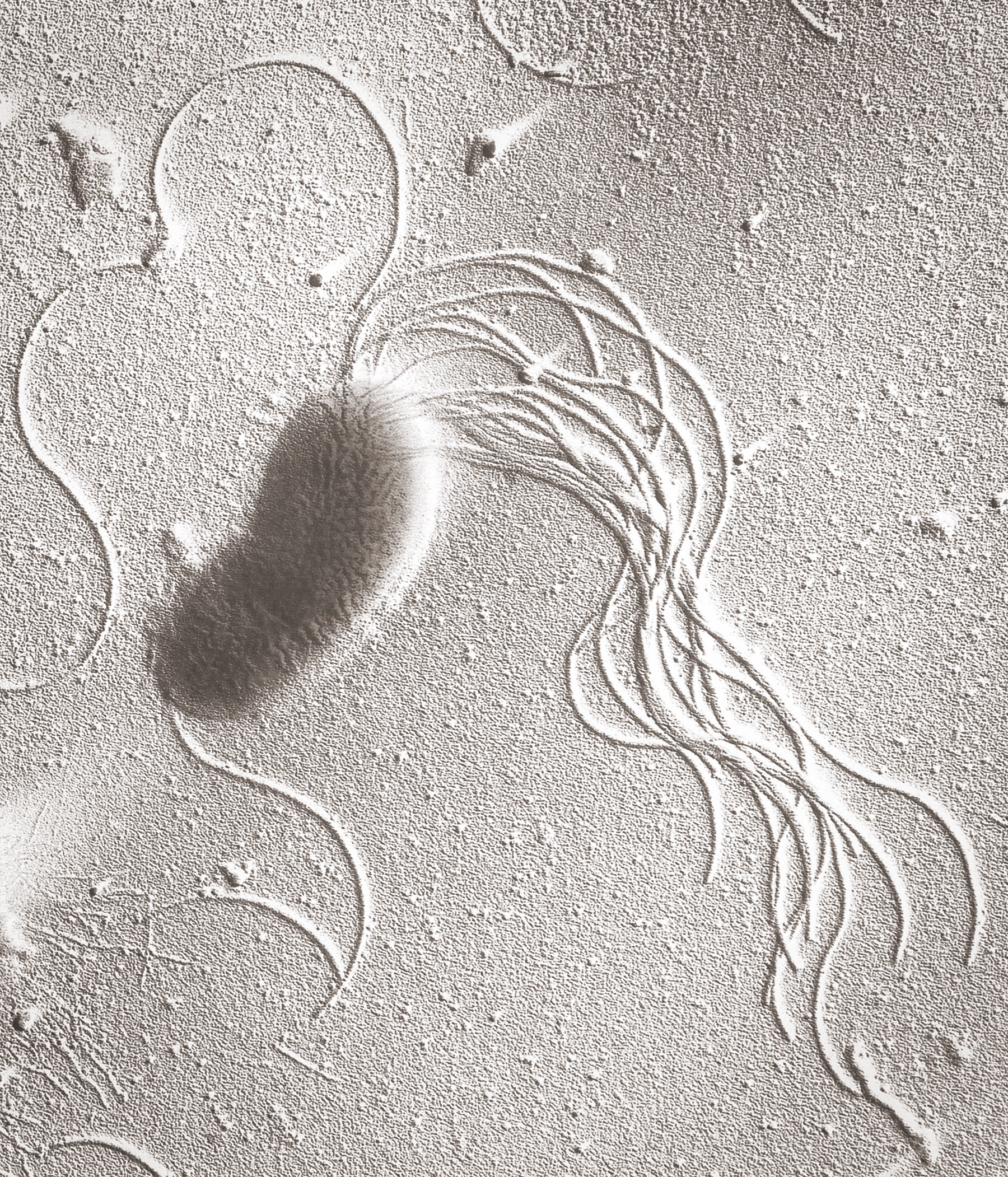
A transmission electron microscopy image reveals the cluster of flagella that a Pseudomonas fluorescens bacteria uses to move around water in soil.
Dr Tony Brain/Science Photo Library
Berg guessed that the flagellar motor was a rotor that turned the flagellum like a screw. “He did it by sticking two cells together by their flagella and seeing them spinning in opposite directions from each other,” Manson said. “From that, with no knowledge, he hypothesized that the bacterial flagellum rotates. Way ahead of his time. That was 50 years before understanding how this motor works.”
Further experiments indicated that the flagellar motor also switches direction. When its flagella — bacteria typically have several protruding from their surfaces — are all spinning counterclockwise, they form a bundle that trails behind the swimming cell like a braid in the wind, steering it straight. But as soon as one flagellar motor reverses direction and starts rotating clockwise, the bundle falls apart; the reverse-twirling filament unravels the braid and puts the cell’s flagellar motors at cross-purposes, kicking the cell around.
Before Berg’s work, “the idea of a molecular motor was bonkers — no way anything rotates,” said Samuel, Berg’s former student who now runs a Harvard lab of his own. It could wiggle, sure, but rotate? “It requires a certain geometry that people didn’t think was accessible to biology.”
Au contraire. “Biology can build wheels,” Samuel said. “Now we know.”
![]()
Improvements over the last 15 years in an imaging technique called cryo-EM (cryogenic electron microscopy) have enabled researchers to see the flagellar motor’s component parts. That has clarified how it works.
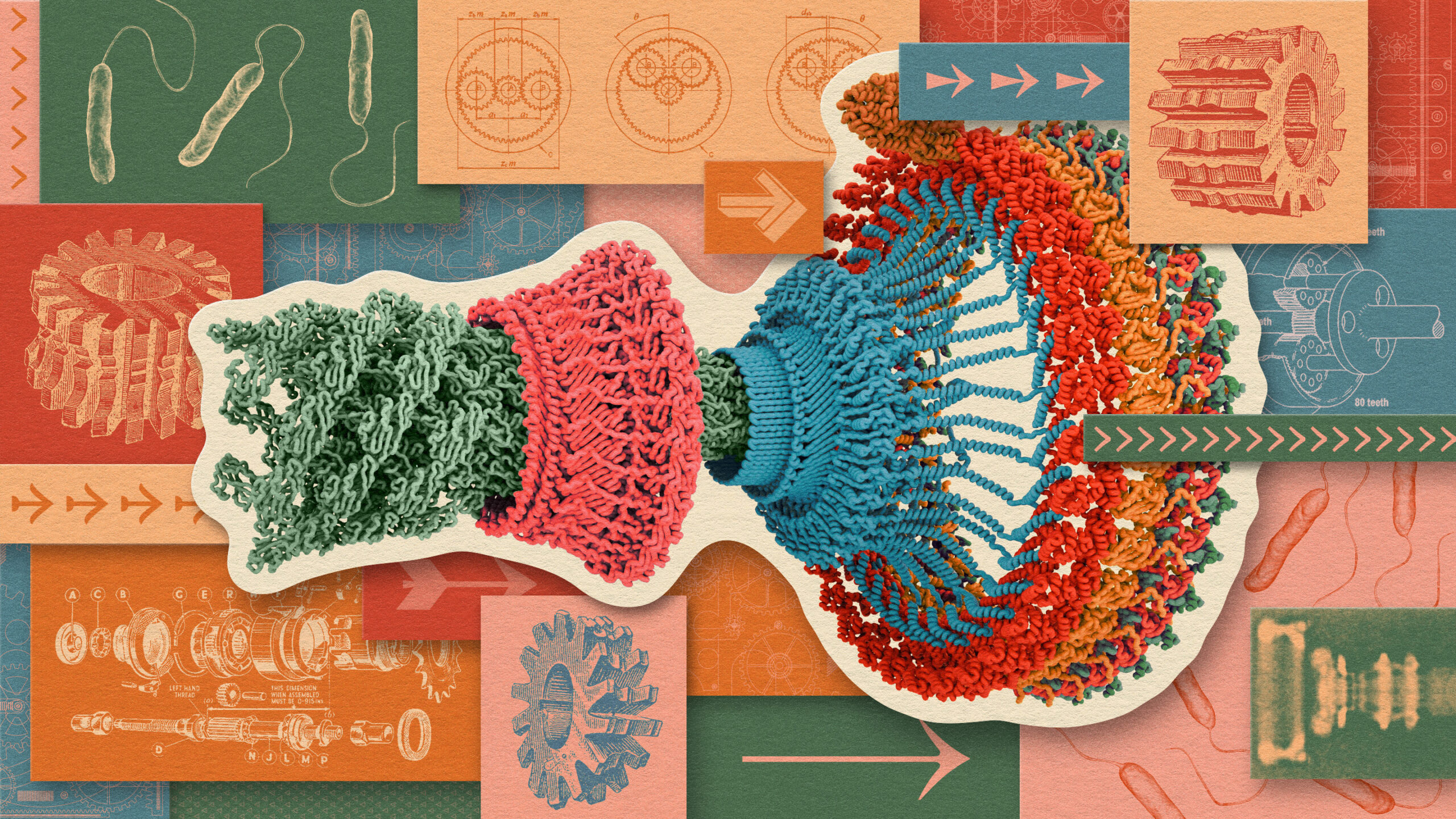
At the base of the motor is the “C ring” (or “cytoplasmic ring”), a ring of 34 identical proteins floating in the cytoplasm within the cell membrane. Scientists in the 1980s and ’90s figured out that when the C ring rotates, the flagellum does too. But why and how it rotates wasn’t obvious.
David S. Goodsell/PDB101.rcsb.ord/Modified by Quanta Magazine
The stars of the show, recent research showed, are the motor’s “stators,” smaller protein complexes that anchor themselves above and outside the C ring. The number of stators varies by bacterial species (E. coli has 10 or 12 available per flagellum), and how many lock into the C ring at a given time depends on the weight of the cell or the viscosity of the surrounding fluid.
Each stator consists of two central proteins that dangle from the cell wall and five proteins of a different kind that form a pentagonal ring around the pair. This pentagonal structure is the part that rubs up against the C ring.
The 5:2 geometry of the stators was revealed in 2020 in a pair of cryo-EM studies, one by Susan Lea and a team at the University of Oxford, and one from a group led by Nicholas Taylor of the University of Copenhagen and Marc Erhardt of Humboldt University of Berlin. The finding pointed to a hypothesis about how the whole motor works: The stators’ pentagonal rings rotate, which then turns the larger C ring, and with it the whole flagellum.
Each pentagonal ring turns like a turnstile, one-tenth of a revolution at a time. What pushes through the turnstile is a stream of protons — the same positively charged particles found in atoms. Protons flow into cells of their own accord, for reasons I’ll get to. This is the proton motive force.
The asymmetric positioning of two proteins inside a pentagonal ring allows a proton from outside the cell to weakly bond to one of them. As the proteins jostle, the proton unbinds, exerting torque on the ring as it goes. That creates an opportunity for the same process to take place with the other central protein. In this way, protons effectively pedal the engine of the flagellar motor. Every second, more than 2,000 of them pass through the pentagonal turnstiles. In December 2025, Samuel published the results of an experiment that verified this.
Protons always want to flow into cells, never out. In passing that way, they always push the pentagonal rings clockwise. Normally, this turns the C ring counterclockwise (like the opposite turning of interlocking gears), which propels the swimming cell forward. How, though, can the flagellar motor switch directions? In 2024, another pair of cryo-EM studies, from Lea, then with a team at the National Institutes of Health, and a group led by Tina Iverson at Vanderbilt University, revealed the answer.
Recall that a flagellar motor switches directions, causing the bacterium to tumble, when environmental conditions seem to be getting worse. When fewer nutritious molecules drift in, the bacterium “phosphorylates” proteins called CheY, tagging them with phosphorus atoms. Within milliseconds, phosphorylated CheY molecules diffuse around the cell, and one of them binds to one of the C-ring proteins. This small change triggers a transformation: The protein flips into a different structural configuration, which flips the next protein, and then the next. Almost instantly the whole C ring reshapes itself, like a hair clip snapping into the other of its two stable forms. Samuel’s team confirmed that the system is sensitive to a single signaling molecule in a study published in March 2026.
While the C ring is in its altered shape, the stators — the little clockwise-revolving motors — rotate against the inner edge of the C ring, rather than its outer edge. As a result, the C ring turns clockwise too. The flagellar bundle falls apart, and the cell tumbles.
Soon enough, the unstable phosphorus atom falls off the CheY protein, causing the proteins of the C ring to flip back to their original stable formation and turn counterclockwise again. The bacterium returns to forward movement, in a new direction, is search of more food.
“It’s a really elegant way of turning a unidirectional power into bidirectional rotation of the large object,” said Lea, who is now at St. Jude Children’s Research Hospital.
![]()
The proton motive force that drives the flagellar motor was proposed in 1961 by Peter Mitchell, a biochemist who worked out of his own private lab at a country estate in Cornwall, England. Though initially dismissed and even ridiculed, Mitchell went on to win the 1978 Nobel Prize in Chemistry for his idea that a current of protons constantly flows into the cell as the cell vigorously pumps them back out, and that this is the driving force behind key cellular processes.
Protons flow in because they’re diffusing from an area of high concentration (outside the cell) to an area of low concentration (inside). There are fewer than 100 free protons inside a bacterium at a time, while a similar volume of the surrounding water has tens of thousands. The cell maintains this state with machines called electron transport chains that pump out thousands of protons per second. As protons are pumped out, thousands more flow in, drawn by the net negative electric charge and the general tendency for entropy to rise as particles (in this case, protons) spread out in space ever more evenly. Cells have rigged up all kinds of molecular machines that, like water mills on rivers, take advantage of proton currents coming into the cell.
“It boggles the normal human understanding of how things work,” Manson said. “How can you have thousands and thousands of protons coming into the cell every second and still have only a few dozen inside the cell? Because they bind to something, they get pumped out again. The equilibria are so incredibly fast.”
So what makes the cell go, what breathes life into the atomic arrangements, is the efficient removal of protons so that more protons will flow. “If you were to open up a channel to protons, they would come pouring into the cell, and the proton motive force would be gone instantly,” Manson said. He’s seen this happen, when cells starve and can’t pump enough protons out. The voltage drops to nothing, and the cell’s machinery shuts down. If you’re a bacterium, your flagellar motor stops. You’re stuck.
Rarely have I loved biology more than when marveling at the flagellar motor and the influx of protons that turns its gears. “The entropic energy of the proton motive force gets converted into the kinetic energy of the rotation,” Manson said. “That’s all it is. All of it is just that. If you understand that, you basically understand the underpinnings of all that happens in biology.”