A New Type of Neuroplasticity Rewires the Brain After a Single Experience
Introduction
Every experience we have changes our brain, the way a ceramicist reshapes a slab of clay. Every corner we turn, every conversation we have, every shudder we feel causes cascading effects: Chemicals are released, electricity surges, the connections between brain cells tighten, and our mental models update.
The brain is “incredibly plastic, and it stays that way throughout the lifespan of a human,” said Christine Grienberger, a neuroscientist at Brandeis University. This plasticity, the quality of being easily reshaped, makes the brain really good at learning — a quintessential process that allows us to remember the plotline of a novel, navigate a new city, pick up a new language, and avoid touching a hot stove. But neuroscientists are still uncovering fundamental rules that describe how neuroplasticity reshapes brain connections.
Recently, neuroscientists described a new form of neuroplasticity that might be helping the brain learn across a timescale of several seconds — long enough to capture the behavioral process of learning from a single experience. In two recent reviews, published in The Journal of Neuroscience and Nature Neuroscience, they describe “behavioral timescale synaptic plasticity,” or BTSP. This type of learning in the hippocampus, the brain’s memory hub, is caused by an electrical change that affects multiple neurons at once and unfolds across several seconds. Researchers suspect that it may help the brain learn in a single attempt.
“It’s pretty clear that [BTSP is] a strong, powerful mechanism that can lead to immediate memory formation,” said Daniel Dombeck, a neuroscientist at Northwestern University who was not involved with the theory’s development. “It’s something that has been missing in the field for a long time.”
By uncovering BTSP, neuroscientists have unraveled more of the story of how the brain changes with experience, bringing us closer to understanding how learning happens. “Neuroplasticity is … one of the last frontiers of the brain,” said Attila Losonczy, a neuroscientist at the University of Texas Southwestern Medical Center who studies BTSP. “If we understand this, I think we take a major step towards understanding how the brain works.”
A Plastic Brain
Today, neuroplasticity is taken as fact, but for much of the 150-year history of neuroscience, the adult brain was thought to be static. “The idea that the adult brain can change wasn’t actually widely accepted until very late [in] the history of modern neuroscience,” said Moheb Costandi, a trained neuroscientist and author of Neuroplasticity, a primer from MIT Press. “It was taken for granted that the adult human brain can’t change.” In 1928, Santiago Ramón y Cajal, the oft-cited founder of modern neuroscience, wrote that “in adult centers the nerve paths are something fixed, ended, immutable.” This idea would prevail well into the middle of the 20th century.
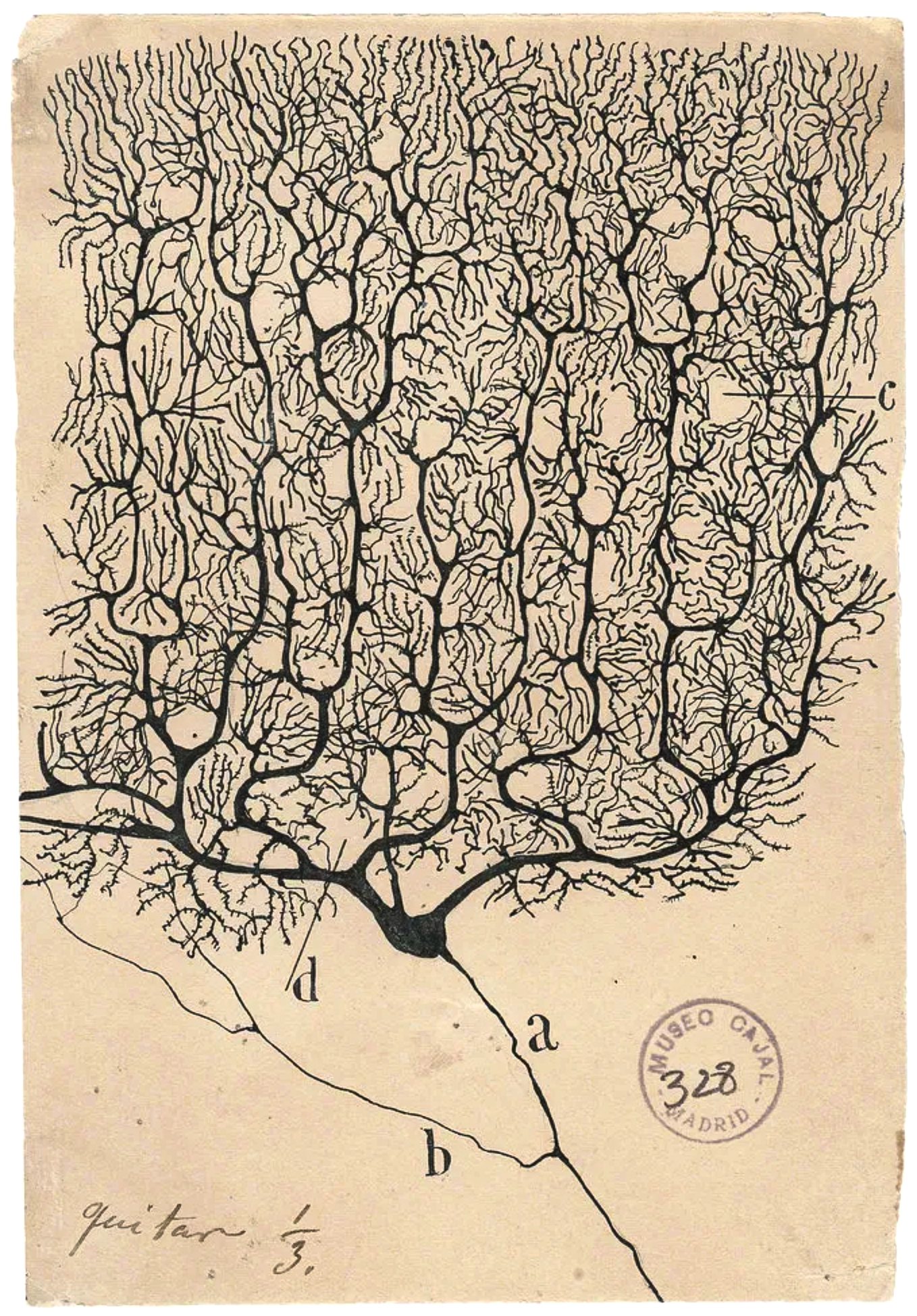
Santiago Ramón y Cajal’s drawings of neurons, such as this Purkinje cell with branching dendrites, led to his theory that the nervous system contains many interacting cells.
Santiago Ramón y Cajal/Public Domain
We now know that the brain is constantly remolding itself, both functionally and structurally, across many scales — from the molecules that flow between neurons to the connections that stretch across the brain and beyond.
The power of neuroplasticity is perhaps best demonstrated by case studies. One patient born without an olfactory bulb could smell because other parts of her brain remolded to serve as substitutes. Another patient had the entire left side of her brain removed as a baby; after her right side reorganized to take on the left’s former roles, today she has a functional life. When a stroke or an accident damages the brain, other neurons fill in to recover patients’ everyday functions such as speaking and walking.
Neuroplasticity also drives everyday learning. This process is mainly thought to result from synaptic plasticity, or changes to the trillions of connections between neurons. And although the brain learns in various ways, one particular idea has dominated for more than 70 years.
In 1949, Donald Hebb, a Canadian psychologist, articulated a theory of learning now known as Hebbian plasticity. According to this model, when neurons are activated within milliseconds of each other, the connection between them is physically strengthened, so that in the future they are more likely to fire together. Over time, they form a network that represents a concept or an experience. In other words, the more the networks in the brain are used, the stronger they get, an idea often summarized as “neurons that fire together, wire together.”

In 1949, Donald Hebb, a Canadian psychologist, articulated neuroscience’s dominant theory of learning, now known as Hebbian plasticity.
UBC Archives Photograph Collection; University Archives, University of British Columbia Library. UBC 41.1/2039-1
But neuroscientists “always had a sneaking suspicion that Hebbian plasticity wasn’t quite right,” said Jeffrey Magee, a neuroscientist at Baylor College of Medicine. Or at least, it wasn’t the full story. It required an experience to be repeated multiple times to imprint the lesson on the brain — a framework that may explain how we learn a new city or language, but not how we learn from a single, highly charged experience, such as touching a hot stove.
Even so, finding more explanatory mechanisms hasn’t been top of mind for neuroscientists. “It wasn’t a quest, like in particle physics for missing particles,” Losonczy said. Maybe there were a couple of gaps that needed to be filled, but most researchers assumed that the Hebbian framework would require only tweaks. Few were thinking that a fuller understanding of neuroplasticity might include a new mechanism.
Mighty Trees
In 2014, when Magee attached electrodes to rodents to record their neural activity, he wasn’t looking to challenge Hebbian plasticity. Magee, then at the Howard Hughes Medical Institute’s Janelia Research Campus, and his students Grienberger and Katie Bittner were looking to observe the behavior of neurons’ arms, called dendrites, in a living animal.
These branches receive molecular signals at one end of a neuron and induce the cell to rapidly fire an electrical charge that ripples down the cell body, known as an action potential. This process ends with the neuron releasing its own batch of molecular signals, which latch onto the dendrites of the next neuron in the network, continuing the process.
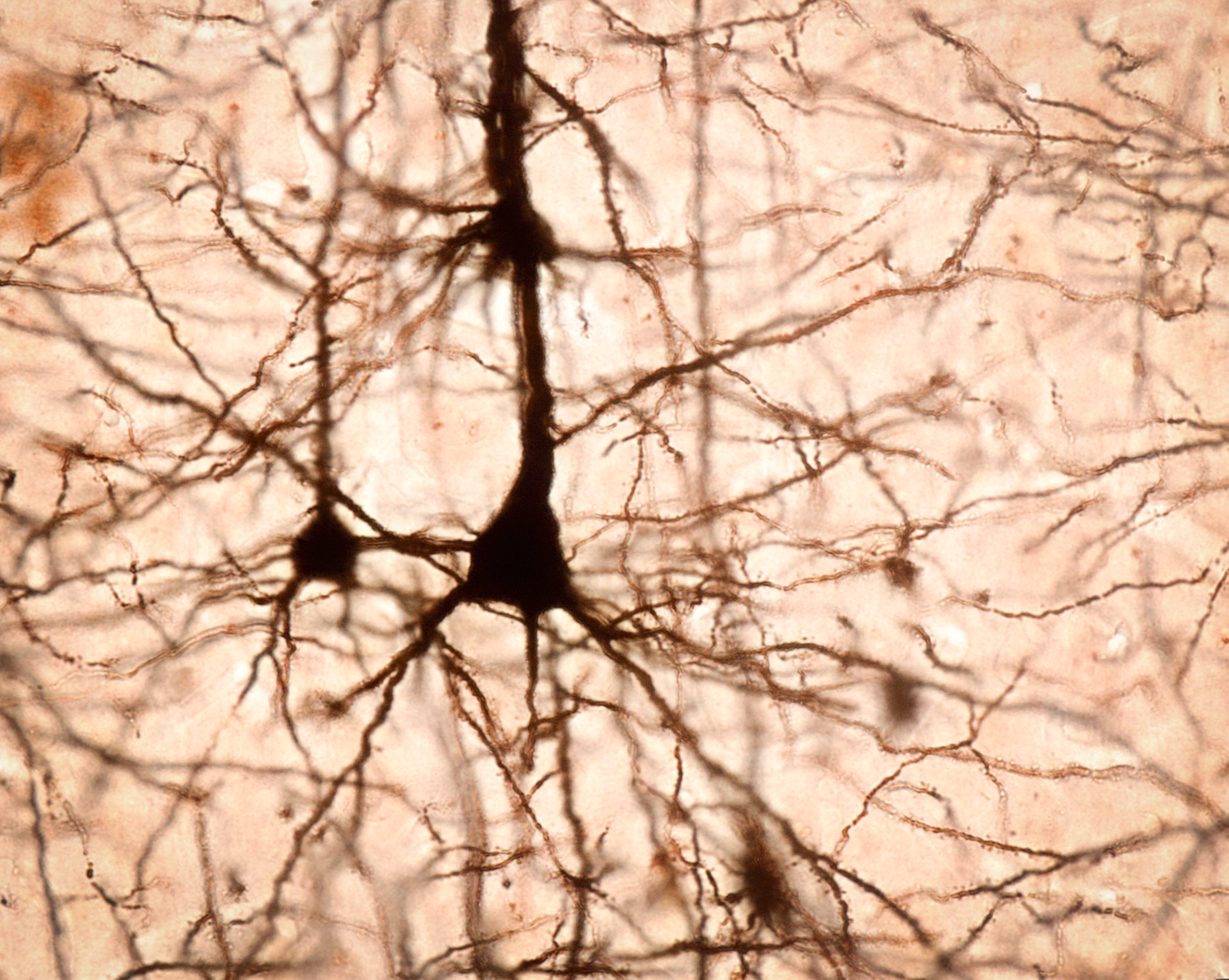
Dendrites, the extended branches that receive signals from other neurons, are the star players in a recently described type of neuroplasticity. In this image of stained pyramidal neurons from the cerebral cortex, rootlike dendrites extend from the cell bodies.
Jose Calvo/Alamy
In recent decades, neuroscientists have come to a “slow realization that dendritic activity is super important for plasticity and for neuronal computations in general,” said Antoine Madar, a postdoc at the University of Chicago, who led the 2025 review of a Society for Neuroscience symposium on BTSP in The Journal of Neuroscience.
There is a “zoo” of different events that take place at dendrites, he said. They can fire their own local or global electrical spikes. They can cover a larger or smaller area, and they can surge for longer or shorter periods of time. Neuroscientists have found that these events at dendrites can allow even single neurons to perform complex computations — meaning that dendrites are the reason why a single neuron can have the same amount of computational power as a deep artificial neural network.
Still, there was much unknown about dendrites’ behavior. Neuroscientists have mainly characterized them in brain slices, where neurons are alive and can be activated but aren’t attached to a living animal. “We were trying to take that into the actual behaving animal, or the actual behaving brain,” Magee said.
In 2014, they began to home in on the hippocampus, an especially plastic area of the brain where we form experiential memories. It’s also home to place cells, which fire when an animal moves through its environment. Each of these neurons learns to fire at specific locations; later, if the rodent reenters that place, the cell will fire, recalling relevant information stored in the network.

Jeffrey Magee, a neuroscientist at Baylor College of Medicine, led the team that first described behavioral timescale synaptic plasticity in 2017.
Courtesy of Jeffrey Magee
As the rodents ran on a circular track, Magee and his team recorded what was happening in their hippocampal dendrites. That’s when they observed something interesting.
Neuroscientists had long known that dendrites can sometimes stay active, with a slightly higher charge than when they’re resting, for long periods of time without firing — creating what’s known as a plateau potential. Because a plateau potential increases the odds that the neuron will fire, the activity was considered important to neuroplasticity. But while examining the rodent data, Bittner saw that place cells whose dendrites had produced just a single plateau potential began to fire.
In other words, a single burst of activity at the dendrite had tuned that cell to fire in that location. It was previously thought that encoding a place cell would take multiple action potentials, via Hebbian learning, which would require the animal to explore the same spot multiple times.
“So we were like, ‘Wow, what’s going on here?’” Magee said. When they experimentally triggered these plateaus, the cells fired in that location 99.5% of the time after a single dendritic plateau.
The researchers were elated. “We were kind of running back and forth between offices, like, you know, waving papers around — like, ‘Look at this result,’” said Aaron Milstein, a neuroscientist at Rutgers University, who worked in Magee’s lab at the time. It seemed that dendrites weren’t just passively nudging a neuron to fire — they were causing the change themselves, strengthening the synapse in a single, swift step.
Magee and his team published their findings in 2015. At that point, they thought they had observed some weird subtype of Hebbian plasticity. But when they looked more closely at brain recordings of live animals plus brain slices, they recognized the biggest difference between the dendrites’ activity and Hebbian plasticity: time.
In most studies of Hebbian plasticity, neurons can strengthen or weaken their connection if they are activated within milliseconds of each other. Dendrites’ plateau potentials, on the other hand, persist for tens to hundreds of milliseconds (sometimes approaching one second), and through BTSP they can strengthen synapses active six to eight seconds before or after the plateau event.
“It became pretty obvious that this wasn’t at all the standard kind of Hebbian plasticity,” Magee said. “That made it even more interesting, of course, and a little bit intimidating, because then we were going to be facing up to nearly 100 years’ worth of dogma.”
It also addressed another big question that Hebbian plasticity had left open: how our cells can capture our relatively slow human behaviors.
“If you imagine even the simplest of the behavioral learning — for example, learning to stop at a red light signal, or to even explore and figure out what are the main parts in a particular room — it will take you at least a few seconds,” said Anant Jain, a neurophysiologist at the Center for High Impact Neuroscience and Translational Applications in India. BTSP explains how the brain can encode behaviors in a single burst of brain activity that unfolds across several seconds.
Because this new mechanism seemed more behaviorally relevant than Hebbian learning, Magee named it “behavioral time scale synaptic plasticity” in a 2017 Science paper. “I’m not very good at naming things,” he admitted. Then he waited for the response from fellow neuroscientists.
One-Shot Learning
Initially, BTSP received pushback within the field. There was good reason for that, Magee said, as it challenged the dogma of neuroplasticity that had dominated for decades. But over the past few years, other researchers have started to investigate it themselves.
This is “a very compelling model for single-shot learning,” said Losonczy, who worked in Magee’s lab prior to the discovery and now studies BTSP at his lab. Unlike the mechanisms that allow an animal to learn a new skill slowly, BTSP might help it to learn — after just a single exploration of its cage — that food exists in the northwest corner or that a shock exists to its south. “Sometimes you need to remember events you only have one chance to remember, [such as] where the predator is,” Losonczy said. “Otherwise, you will be taken out of the genetic pool.”
While it’s a neat explanation, the exact mechanism remains elusive. “There are still so many unanswered questions, at least at the level of molecules,” Jain said. However, neuroscientists are starting to get some hints.
Early findings suggest that certain experiences cause synapses, the gaps between neurons where dendrites extend, to be tagged with elusive biochemical signatures called eligibility traces. These tags stick around for several seconds and indicate that those neurons were recently active and therefore relevant to a particular experience. Then, in the next neuron, a dendritic plateau potential causes a widespread voltage change that spreads across the entire dendrite. This plateau triggers all the synapses with the eligibility trace to strengthen.
Some studies are starting to zoom in on the molecular process. In 2024, Jain and his team reported that dendritic plateaus might cause a cascade of biochemical signals to build up over several seconds and then activate one of the most important proteins for learning, known as CaMKII. This protein directly influences synaptic strength by physically increasing the surface area and the number of receptors on dendrites, allowing more neurotransmitters to bind there the next time the cell fires.
BTSP may also address an ongoing conundrum in neuroscience. Because it strengthens only relevant active neurons, as opposed to any active neuron, BTSP may help address the “credit assignment problem” — how the brain can tell which neurons should encode a given experience. Now, Magee and others are looking into the role that BTSP might play not only in learning but also in consolidating memories.
However, Dombeck is cautious about overreaching on BTSP’s significance. It has been observed in limited circumstances: only in the hippocampus as an animal learns locations (although researchers have found some evidence for BTSP in the neocortex, where the brain’s higher-order processes happen). In his lab, Dombeck has found that BTSP occurs in some hippocampal cells, but not in all of them.
Jain is not even convinced that BTSP should be categorized as a non-Hebbian type of learning. Hebbian learning is often vaguely defined, and Hebb himself was vague about the timescales upon which it works. “Donald never really specified that it has to happen within milliseconds,” only that the neurons need to repeatedly fire together, he said. Only later did neuroscientists mechanistically refine it to include millisecond timescales, Jain said.
Most neuroscientists agree that BTSP doesn’t replace Hebbian learning, but rather works alongside it. “Hebbian plasticity probably plays a huge role in development, in the initial wiring” of the brain, Grienberger suggested, while BTSP may be more important for forming episodic memories in adults.
There’s still much unknown about BTSP, especially the mechanism, which Madar said is “quite speculative.” However, he also acknowledged that before becoming the archetypal model for learning, “Hebbian plasticity was also a hypothesis.” Our understanding of how the brain learns through endlessly changing is itself endlessly changing.



