Disorder Drives One of Nature’s Most Complex Machines
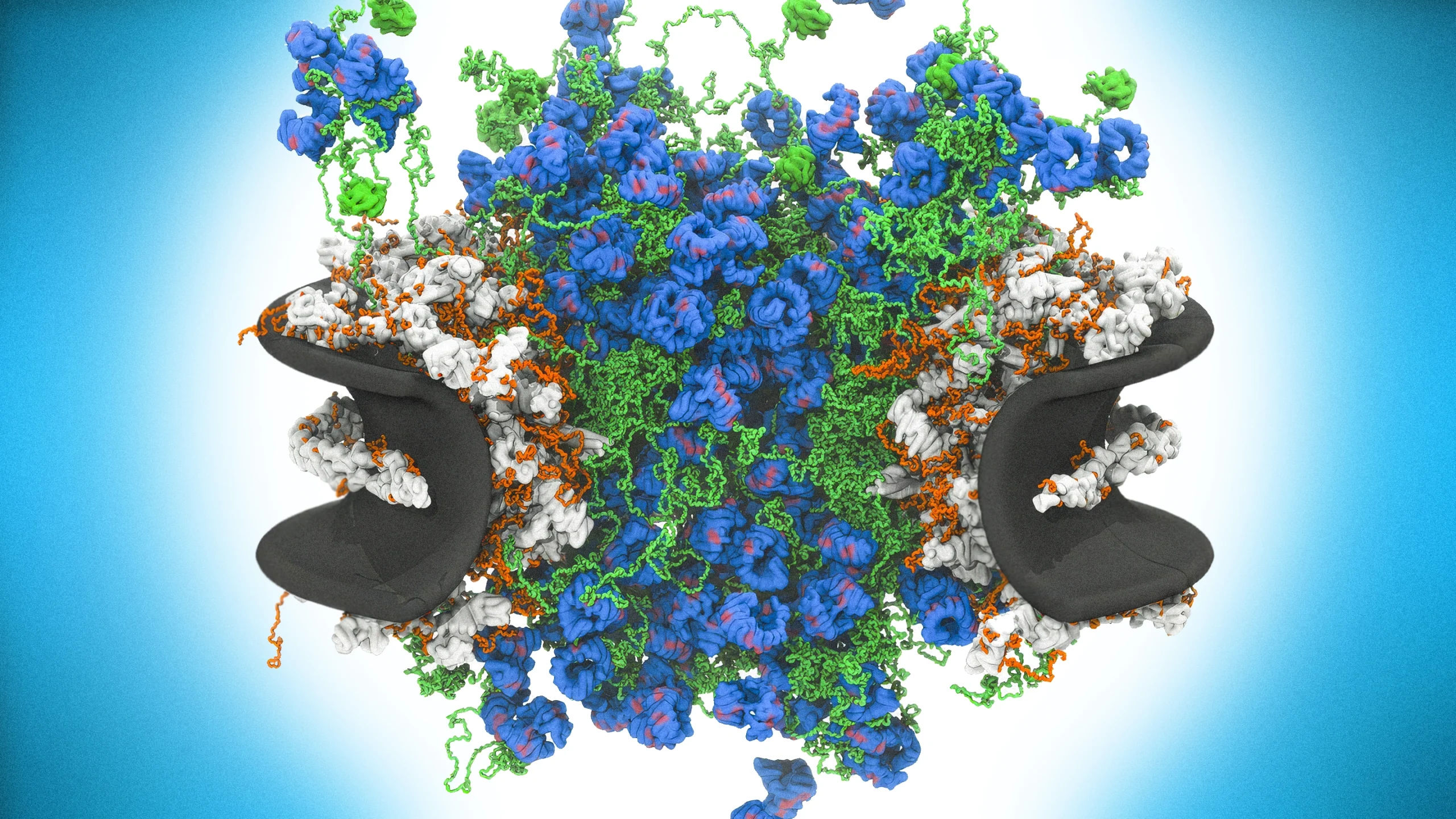
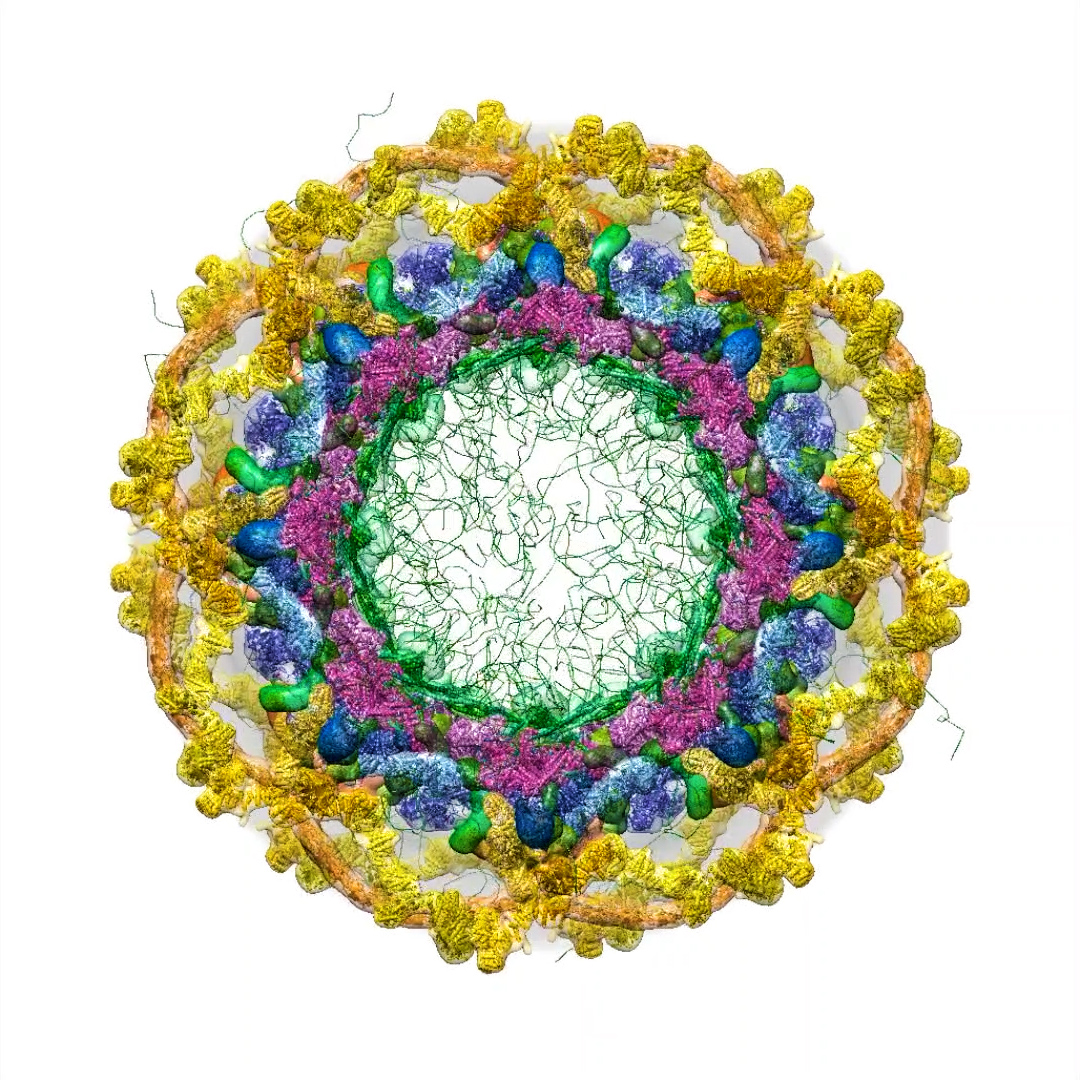
The inner channel of the nuclear pore complex in all its messy glory. Far from a simple pore, the complex is filled with dynamic proteins that move molecules into and out of the nucleus. In this computer model, green strands represent disordered nucleoporin proteins and blue knots are karyopherin transport proteins.
Introduction
At the dawn of complex life, evolution created a container for DNA, its most treasured item. A few billion years later, 20th-century microscopists looked at this container — the nucleus — up close and saw that it was covered in tiny openings. At the time, they didn’t know what to make of these structures, but as microscopy improved, something grand came into focus: what we now call “nuclear pore complexes,” some of the largest and most marvelous molecular machines ever formed.
Every nuclear pore complex is constructed from hundreds of proteins, of around 30 different types. From the front, it looks like an eight-petaled flower; from the side, like a flying saucer. Its center opening spills over with spaghetti-like proteins tethered to the inner walls of the complex.
“It’s a thing of enormous beauty,” said Brian Chait, a chemical biologist at Rockefeller University. “It’s marvelous. It’s a wonder. … It’s phenomenal.”
This machine has a vital job: directing molecular traffic into and out of the nucleus. More than an open door, the protein complex recognizes different molecules as they approach — and lets only some through. “The nuclear pore complex is ultimately the gatekeeper for the nucleus,” said Roderick Lim, a biophysicist at the University of Basel in Switzerland. “Everything that has to get in and out of the nucleus has to go through these pores.”
Nearly every eukaryotic cell has a nucleus punctured with nuclear pore complexes, and the main components of the complex are incredibly conserved across species, from single-celled yeasts to multicellular humans. Evolution “came up with that thing one time and got stuck with it,” said André Hoelz, a structural cell biologist at the California Institute of Technology.
A single mammalian nucleus can contain thousands of them. Every second, each nuclear pore lets hundreds to thousands of molecules of all shapes and sizes pass through so that they can travel to their destinations to make proteins, regulate genes, and generally help the cell function. Some large molecules are carried through the channel by proteins, while small ones diffuse across on their own. And while some effortlessly glide through, to others it is an impenetrable barrier.
How this gate works with such selectivity is a mystery. Over decades, biologists have worked out what most of the static parts of the machine look like. But its center is restless, endlessly moving and morphing, which makes it difficult for even the best methods to visualize.
That’s gradually changing. In a study published in Nature Cell Biology at the end of 2025, high-resolution microscopy showed the central barrier in motion at millisecond resolution, revealing a flexible structure that constantly rearranges itself. The experimental imaging, backed by computational modeling, suggests that the function of the nuclear pore complex is guided by flexibility and movement.
This is more than an intellectual curiosity. A surge of recent research has linked neurodevelopmental disorders, viral disease, and cancers to problems with the complex. If biologists can discern how its cellular trafficking system works, they could develop methods to stop unwanted pathogens from getting through — and also try to guide therapeutic molecules into the cell nucleus, where the genome itself awaits.
“We are starting to approach a near-atomic picture of nuclear pores from various organisms,” said Mike Rout, a cell biologist at Rockefeller University who led the new work that offers insight into a molecular machine at the heart of eukaryotic life.
A Thing of Beauty
For many scientists who study the nuclear pore, it was an instant fascination: They were awed that this (relatively) massive biological machine could allow so many kinds of molecules, and so many of them, to move in and out of the nucleus efficiently, with little room for error.
“It is beautiful,” Hoelz said. “How do you generate an aircraft hangar that has a door where a 747 can go through,” but a small marble can’t fly out?
Somehow the gate itself must play bouncer for life’s important molecules. RNA molecules must get out of the nucleus to the cytoplasm to make proteins. Ribosomes, the structures that do the protein-making, are themselves made of molecules assembled in the nucleus and moved out through the pores. Meanwhile, molecules involved in regulating the genome must be ushered into the nucleus preferentially. As these processes unfold, the nuclear pore is also blocking things that don’t belong, such as harmful enzymes or misfolded RNAs.

The cell biologist Mike Rout has spent decades studying the nuclear pore complex. This vital structure, built from hundreds of individual proteins, is the gatekeeper and traffic controller of a cell’s nucleus.
Lori Chertoff/Rockefeller University
In the decades since the discovery of the nuclear pore complex, biologists have slowly gained a greater understanding of its structure using electron microscopy, tools of physics, and computer modeling. With the advent of cryo-electron microscopy, a powerful technique that images cells after flash-freezing them, they have been able to assess the static parts of the machine. These efforts have generated a near-atomic view of each individual protein that builds the eight petal-like radial units. But the technique hasn’t worked for the very center, called the transport channel, where most of the pore’s action happens.
“Whenever anyone uses some of these microscopes to peer inside nuclear pores, they just see this amorphous cloud inside,” Lim said. “It’s like a haze.”
The reason, which became apparent in the 1990s, is that the center of this giant machine is filled with proteins that have no obvious or specific structure. These proteins, called FG-nucleoporins, have tails that wiggle around like seaweed and that can’t be captured in static images. These tails are “the dark matter of the nuclear pore complex,” Hoelz said.
Most proteins fold into specific structures or shapes essential to the functions they perform in a cell. But intrinsically disordered proteins, a category that includes these nucleoporins, don’t have a single structure. They flail about, changing shape and binding to different molecules.
This means that in the center of the nuclear pore complex, “everything is mediated by disorder,” said Patrick Onck, a computational physicist at the University of Groningen in the Netherlands. “It’s not order that generates this function. It’s disorder.”
In the early 2000s, the field was awash with bickering about the organization and behavior of FG-nucleoporins in the transport channel and how they might traffic molecules into and out of the nucleus. Researchers agreed that most macromolecules can’t get through the channel without the help of cargo-carrying proteins called transport factors. Transport factors, which mingle with the nucleoporins in the channel, bind only to those molecules that include a specific short stretch of amino acids — a molecular tag that “says, in effect, ‘send me into the nucleus,’” Rout said. But researchers differed on how the channel itself might be organized, which would impact how molecules move through it. Some argued that nucleoporins snap onto one another, forming a gel-like mesh network. Others argued that the disordered proteins don’t interact with each other much, but rather constantly undulate like bristles on a brush.
The debate became contentious. “It used to be very hostile,” Hoelz recalled. “People are very passionate about what they’re doing. It’s not a personal animosity.”
“For a very long time, it was like, ‘Are you a gel person, or are you a brush person?’ You’re one or the other,” said Siegfried Musser, a cell biologist at Texas A&M University.

The biophysicist Roderick Lim and his team used high-speed atomic force microscopy to image the inner workings of the nuclear pore complex.
Biozentrum, University of Basel
In recent years, new collaborations and discussions, including at an annual conference where the principals gather to chat, have quieted the conflict. But the truth about the structure and functioning of the transport channel hadn’t been resolved. The researchers would need to get more creative to understand how the complex functions in its native environment.
“We wanted to simply go back to the nuclear pore complex and really ask the nuclear pore to tell us what it’s doing,” Rout said, “not for us to impose on it what it’s doing.”
Fast Tapper
How do you interrogate a molecular complex? Rout and Lim, who have both advocated for a more dynamic model of the nuclear pore, started with a simple idea: Poke it.
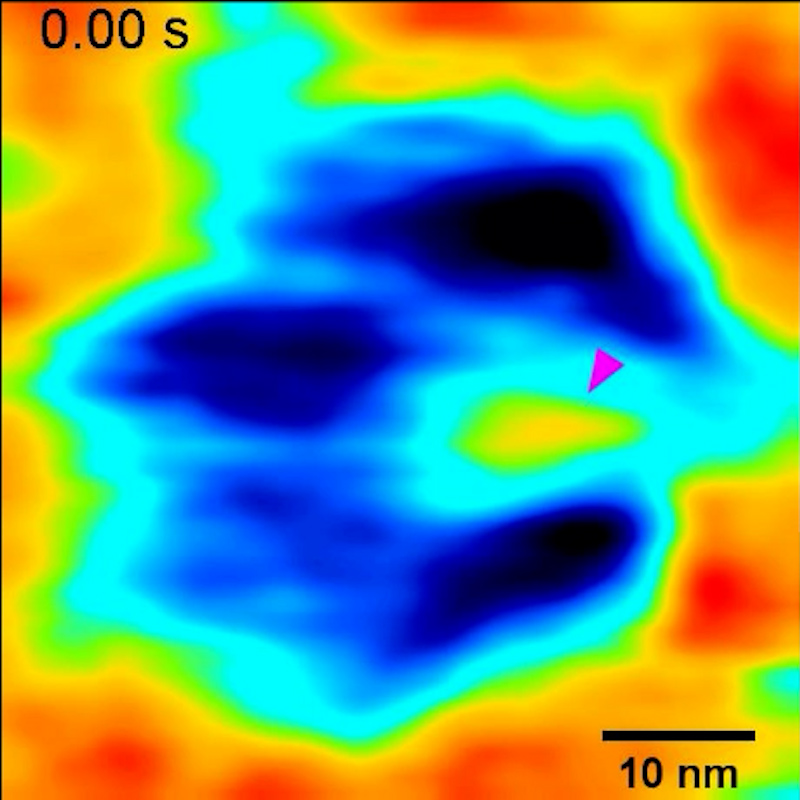
Rout has spent decades characterizing yeast pore complexes. Lim is an expert on high-speed atomic force microscopy, a technique that runs a very sharp probe over a surface, lightly and rapidly tapping it, to get a sense of how it moves.
A few years ago, in a dark basement laboratory, Lim’s graduate student Toshiya Kozai began unpacking plastic vials containing millions of yeast nuclear pore complexes. He toiled for days in the “dungeon of our department,” Lim recalled, working nonstop to get the samples into the high-speed atomic force microscope while they were fresh. “It’s a real labor of love.”
His efforts enabled the team to watch the transport channel change, millisecond by millisecond. “We could see very clear, super rapid motion,” Rout recalled. The resulting videos may look blurry to us macroscopic creatures, but they are some of the highest-definition looks at the behavior of the nanoscopic transport channel.
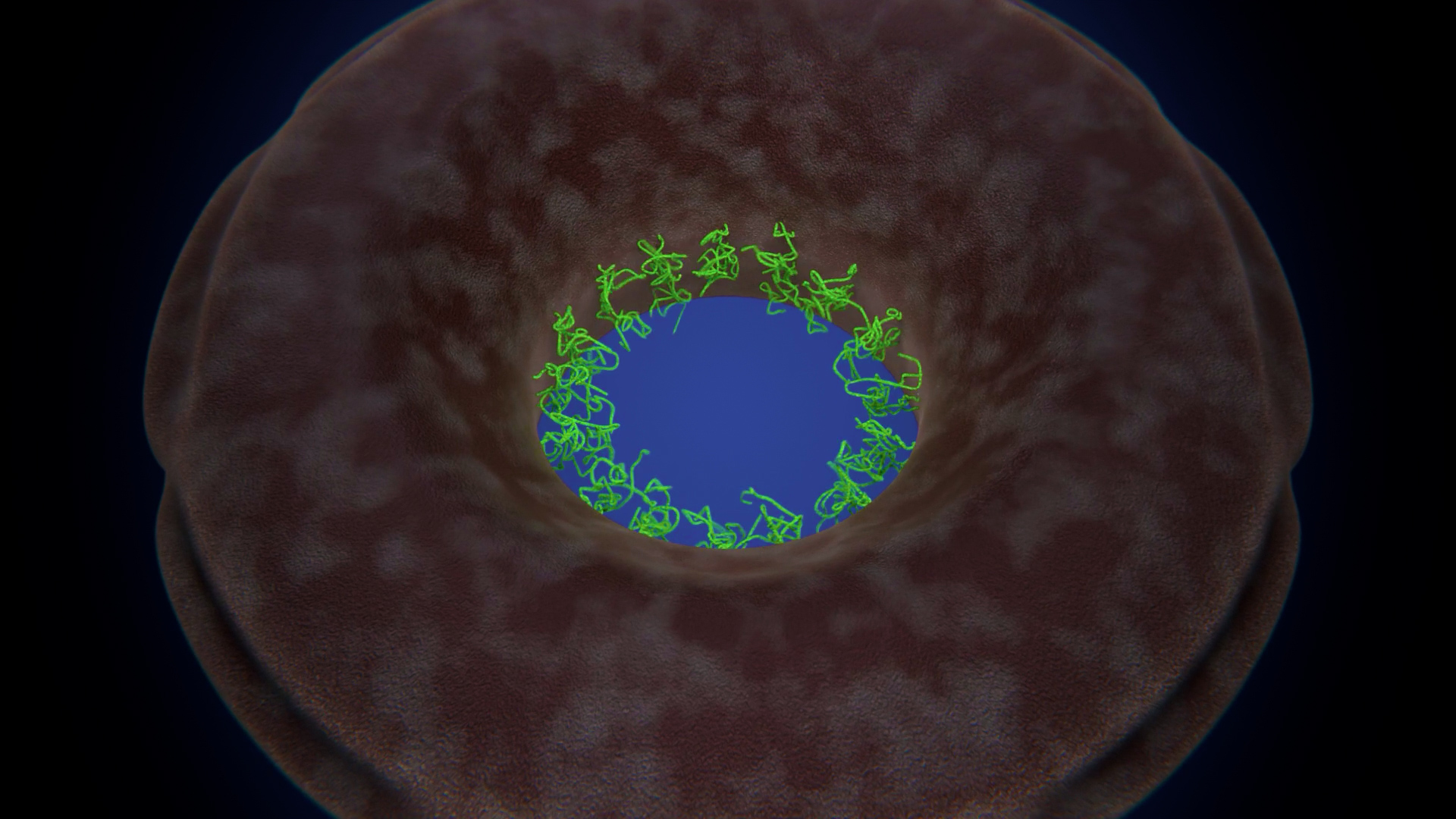
At the edges of the channel, near the walls, Rout and Lim’s team saw rapid fluctuations of molecules — those were the wiggly nucleoporins. At the center they saw a fuzzy blob, known as the central plug, that continuously moved around and repositioned itself within the transport channel. For a long time, biologists had suspected that a central plug might exist, but in previous microscopy images it wasn’t always visible.
“Seeing is believing,” said Hoelz, who was not involved in the study. To be able to show this “is a beautiful thing.”
Led by Chait and using mass spectrometry, the team discovered that the plug was made of transport proteins called karyopherins, or “kaps” for short, plus their molecular cargo. As kaps move through the nuclear pore, they latch onto the nucleoporins, tugging them toward the channel’s center. This creates a temporary, dynamic obstacle that slows other molecules or stops them from moving through. In that way, kaps not only help carry molecules across, but also push away anything that’s not supposed to be there, Rout said. When they added in more transport factors, they saw the plug grow bigger.
The heart of the nuclear pore complex is being constantly rearranged by kaps, creating an ever-changing environment. The results suggest that the pore’s organization and behavior are much more dynamic than a gel network, Rout said, and much closer to a brushlike environment. The findings support a model he previously proposed called the “virtual gate” in which the dynamics of the environment is critical for directing proteins away or through. He likened it to a crowded dance floor.
“If you know how to dance, you can simply enter the dance and swing from partner to partner, holding hands, just quickly exchanging going across,” he said. “If you don’t know how to dance, all you see is this turbulent melee, and you try to get in. No one’s taking hold of you to help you enter the dance, so you just get pushed away.”
To further test their observations, Rout and Lim created synthetic pores the same size as natural nuclear pore complexes. When they tethered nucleoporins inside and added transport factors, the synthetic pores behaved like the wild yeast nuclear pore complexes. They saw the central plug appear.
“What I find actually very striking is that they reproduce the result with a very simple model,” Musser said. “It’s a pretty stunning result.”
Door Not Closed
Hoelz is not totally convinced that this is a nail in the coffin for the gel model. The “answer is [probably] somewhere in the middle, which is very often the case in science,” he said. Researchers likely catch the central channel in different configurations because it’s changing all the time, he suggested, or perhaps the inner channels of some nuclear pores are more like a gel and others are more like a brush.
One recent modeling study led by Onck and published in Nature Communications suggested that the central transport channel could have some parts that are brushlike and others that are similar to condensates — membraneless, liquidlike compartments that have characteristics of gels and brushes. It could even be the case that the channel’s denser center has qualities more like a gel or condensate, Musser said, while the periphery is more brush-like.
Hoelz said that only new technologies that can fully see inside the pore will resolve the debate — and they could come any day now.
“People are constantly trying to develop new tools or new strategies to try to figure out what’s going on,” Musser agreed. In 2025, he and his team published results in Nature that used a powerful 3D imaging tool called Minflux to trace molecules in high resolution as they move through nuclear pore complexes in intact nuclei of human cells. Learning of this method was “a complete game changer for me,” said Hoelz, who was not involved.
Musser’s team observed molecules moving only near the edge of the transport channel. This also complements Rout and Lim’s study, given that the central plug might be blocking the center. “But it doesn’t make sense for the middle not to be used,” Musser said. “I think we just haven’t found the right substrate or developed the right tools to see stuff go through the middle.”
No matter what the inside looks like, it’s clear that the nuclear pore complex is incredibly malleable and robust — which also makes it the cell’s “Achilles’ heel,” Rout said. It is critical to a cell’s health, and central to its most important processes: protein production and gene regulation. But because it is resilient and can endure damage, it can be altered by disease without shutting down.
Some proteins that make up the nuclear pore “show up again and again and again as weak spots for disease,” Rout said, including neurodevelopmental disorders, viral diseases, and cancers. Both cancer cells and viruses likely interfere with proteins in the complex to swing the protein-making machinery in their favor or shut down an immune response.
In that sense, the nuclear pore complex is far more than a molecular gate. “It’s a nexus for integration of information,” Rout said. “And I think if the cell had thoughts, that would be how it thinks of its nuclear pores.”