Viruses Can Scatter Their Genes Among Cells and Reassemble

A classical tenet of virology is that viruses fully infect individual cells and replicate within them. But recent work shows that some viruses instead scatter their genes among many cells. The cells then make new complete viruses by sharing the products of the genes.
Rune Fisker for Quanta Magazine
Introduction
For a virus, a compact genome neatly packaged in a coat of proteins, survival is all about invading a cell, taking over the protein-making machinery to replicate itself and then spreading to other cells. To do this successfully, it might seem self-evident that the entirety of a virus’s small genome would have to be inside an infected cell. A new study recently published in eLife, however, overturns that assumption.
Not only are some viruses split into multiple segments that infect host cells separately, but as researchers in France have now discovered, those fractured viruses can flourish with their genomes scattered like puzzle pieces across a multitude of host cells. Something — presumably, the diffusion of molecules among the infected cells — allows complete viral particles to replicate, self-assemble and infect anew.
“You can get all of the necessary gene products together to produce new viruses in a cell that doesn’t actually have all the gene segments in it,” explained Christopher Brooke, a virologist at the University of Illinois at Urbana-Champaign.
“A classical view in virology assumes that the viral replication cycle occurs within individual cells,” said Anne Sicard, the lead author of the new study and a plant pathologist at the French National Institute for Agricultural Research (Institut National de la Recherche Agronomique, or INRA) in Montpellier. But in the case of this “multipartite” virus that she and her colleagues examined, “it seems that this is not true. The segments infect cells independently and accumulate independently in the plant host cells.” She added, “It really shows that the virus doesn’t work at a single-cell level, but at a multicellular level.”
Multipartite viruses have been known for over half a century, when researchers realized that a virus could be composed of two or more independent pieces, all of which were vital for infection. One piece might be necessary for making essential viral enzymes, for instance, while the other would be needed to make the capsule in which the viral particles (or virions) are packaged and transported to other cells.
But being multipartite carries considerable risks. Parts of the genome can easily be lost or left behind, dooming the rest by breaking the cycle of infection. Because the segments are frequently found in different proportions — some may be common, while others are rare — the rare ones can be lost especially easily.
Scientists have therefore wondered about multipartite viruses since they discovered them. “Why on earth would a virus do this? Why would you separate your genome? What are the advantages to having these segments that are packaged separately?” asked Mark P. Zwart, an evolutionary virologist at the Netherlands Institute of Ecology.
To explore these questions, theoreticians developed models to predict the circumstances under which this multipartite lifestyle would evolve from a more typical viral ancestor, all built on the assumption that the full set of viral segments had to coinfect one cell. But the results were perplexing. A study from 2012 concluded that, whatever the benefits of multipartition might be, the disadvantages were so great that a virus with more than four segments should be impossible. Yet some multipartite viruses, like the faba bean necrotic stunt virus (FBNSV), were known to have as many as eight segments, each carried in a different particle. Theoretically, it couldn’t have evolved. What could explain its existence?
“We thought that the way we conceptualize these viruses must be wrong,” said Stéphane Blanc, a plant virologist at INRA and the senior author of the new study. They decided to verify the key assumption that all the segments must be together within a cell for the infection to work. “It was not done before because it was so evident that they have to be together that no one actually tested it,” he said.
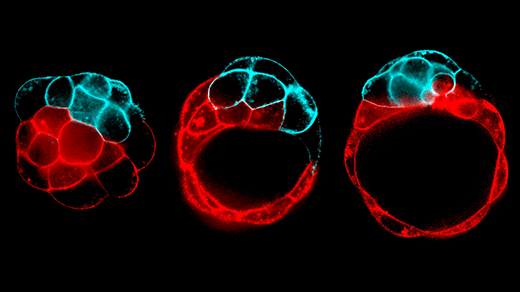
What they found when they scrutinized FBNSV infections blew them away. By tagging two viral segments at a time with different colored fluorescent probes, the team could see that the full complement of viral segments was absent from the vast majority of individual host plant cells they examined. Furthermore, the researchers showed that a protein required for viral replication was present in cells that did not have the genome segment coding for it.
From this, they inferred that the virus particles must be sharing gene products — either messenger RNA molecules or proteins — among cells, so that each particle could replicate and package itself into a capsule to spread. How exactly those necessary components are shared across plant cells isn’t fully understood, but Blanc and his team are looking into it. The answer may involve the plasmodesmata, networks of microscopic canals that extend through plant cell walls and allow adjacent cells to share other proteins.


The plant pathologist Anne Sicard (top) of the French National Institute for Agricultural Research and her colleagues made the recent discovery that the segments of the genomes of multipartite viruses infect and accumulate independently in the host cells. “It really shows that the virus doesn’t work at a single-cell level, but at a multicellular level,” she said.
The plant pathologist Anne Sicard (left) of the French National Institute for Agricultural Research and her colleagues (right) made the recent discovery that the segments of the genomes of multipartite viruses infect and accumulate independently in the host cells. “It really shows that the virus doesn’t work at a single-cell level, but at a multicellular level,” she said.
Courtesy of Anne Sicard
This new understanding explains how a multipartite virus can sustain infections within a plant, but it opens up new mysteries about how one spreads. FBNSV, for instance, depends on aphids that eat faba bean plants to transmit it. But the little insects must collectively capture all eight segments of FBNSV and introduce them to the same plant to successfully pass on the infection. Presumably, a large proportion of infection events never succeed because the aphids pick up only a subset of the eight particles.
The discovery “alleviates the problem at the within-host level because the particles don’t have to reach all the cells together, but we still have a problem for between-host transmission,” Blanc said.
Why a virus would benefit from a multipartite lifestyle is also up for debate. One idea, Blanc says, is that partitioning the genome allows each segment to vary in frequency as a quick-and-dirty way to regulate gene expression, because a given gene’s level of activity can depend on the number of copies of it in a cell. Each time the virus infects a new host, the frequency of the segments changes — which could enable the virus to test how much gene expression works best in a new cellular environment.
Eric Freundt, a virologist at the University of Tampa, speculates that if a host plant’s innate defenses destroy only those cells that express certain viral proteins, then distributing the genes for the proteins into different particles might guarantee that the virus goes undetected in some cells. Another possibility, Freundt suggests, is that distribution tiptoes around the “unfolded protein response,” which can kill cells when a virus overwhelms them by attempting to produce all of its proteins at once. By distributing its genome across many plant cells, the virus may avoid overwhelming the machinery of any single cell.
Still, Blanc and Freundt are quick to acknowledge that these are just hypotheses. “The reason for their evolution is still quite a mystery,” Blanc said.
Zwart points out that most ideas about the advantages of multipartition are really about genome segmentation, not partition of the virus into different infective units. Separating the genome into segments allows different viruses to recombine various advantageous forms of their genes easily.
Arvind Varsani, a virologist at Arizona State University, agrees. “From a modularity perspective, you could see the advantages of multicomponent viruses where each module can be independent,” he said. In a plant with multiple coinfecting viruses, “you can gain elements much more quickly, and adapt to the environment by mixing and matching.”
Proof of the strength of this strategy can be found in the influenza virus, a master of reassortment. The flu genome also has eight segments, although those segments are packaged together in one viral capsule. That allows it to reap the benefits of segmentation without paying all the costs of multipartition.
But the flu may be more similar to the FBNSV than first meets the eye. Brooke discovered that, depending on the strain, only a tiny fraction (1-10 percent) of flu virus particles contain functional copies of all eight genome segments. “The vast majority of flu particles are these incomplete, or what we call semi-infectious, particles that are incapable on their own of initiating productive replication,” he explained. “This was a surprise because it suggests that this virus, which is super successful and highly transmissible, is largely existing as these particles that cannot replicate on their own.”
He predicted, “Asking how viruses operate as populations rather than individual virion particles is going to end up being important for lots of different viral systems.”
Part of that importance is conceptual: It may be too limiting to think of the DNA inside any individual viral capsule as defining its genome. Instead, it might be better to imagine a viral genome as the suite of genes represented across an entire viral population. Theoreticians are already delving into the implications.
Zwart expects that new theoretical models will come out soon to explore these insights, such as ways of framing the evolution of these viruses in terms of multiple levels of natural selection. Within an individual host plant, local forces of natural selection will allow the virus to balance the production rates of its segments successfully. But when the virus moves on to a new plant, it must be able to adapt to that new host environment as well, so it needs to retain some versatility. A higher level of selection may therefore sometimes temper the local level and rebalance the ratio of segments more evenly.
“There is such a richness in all those dynamics,” Zwart said. “It’s really fascinating.”