Mitochondria May Hold Keys to Anxiety and Mental Health

Feelings of stress and anxiety may depend not just on the neurons in our brains but the mitochondria inside our cells.
Maggie Chiang for Quanta Magazine
Introduction
Carmen Sandi recalls the skepticism she faced at first. A behavioral neuroscientist at the Swiss Federal Institute of Technology in Lausanne, she had followed a hunch that something going on inside critical neural circuits could explain anxious behavior, something beyond brain cells and the synaptic connections between them. The experiments she began in 2013 showed that neurons involved in anxiety-related behaviors showed abnormalities: Their mitochondria, the organelles often described as cellular power plants, didn’t work well — they produced curiously low levels of energy.
Those results suggested that mitochondria might be involved in stress-related symptoms in the animals. But that idea ran contrary to the “synapto-centric” vision of the brain held by many neuroscientists at the time. Her colleagues found it hard to believe Sandi’s evidence that in anxious individuals — at least in rats — mitochondria inside key neurons might be important.
“Whenever I presented the data, they told me, ‘It’s very interesting, but you got it wrong,’” Sandi said.
Yet a growing number of scientists have joined her during the past decade or so in wondering whether mitochondria might be fundamental not just to our general physical well-being but specifically to our mental health. In particular, they have explored whether mitochondria affect how we respond to stress and conditions like anxiety and depression.
Carmen Sandi, a behavioral neuroscientist at the Swiss Federal Institute of Technology in Lausanne, suspected that a deficit in cellular energy might explain the lack of motivation that anxiety-prone people experience. Courtesy of Swiss Federal Institute of Technology Lausanne

Although much of the evidence so far is preliminary, it points to a substantial connection. Mitochondria seem to be central to the very existence of a stress response, serving both as mediators of it and targets for the damage it can do. To some of the researchers involved in this work, the stress response even looks like a kind of coordinated action by mitochondria throughout the body that interacts with the neurological processing.
“I think mitochondria are underrated,” said Martin Picard of Columbia University’s Irving Medical Center in New York, whose laboratory has helped to pioneer this research. “They’re the chief executive organelle of the cell.” Now scientists can explore what the implications of the organelles’ importance might be for future therapies.
Mitochondria and Mental Health
Mitochondria are the tiny structures inside complex (eukaryotic) cells that manufacture adenosine triphosphate, or ATP, the chemical fuel for most metabolic processes. “ATP is the energy that sort of allows for living cells to do what they do when they’re alive,” said Lisa Kalynchuk, vice president for research at the University of Victoria in Canada. The organelles are ancient invaders — the remnants of symbiotic bacteria that integrated themselves into host cells about 2 billion years ago and specialized for energy production. Mitochondria still carry a small amount of DNA of their own, although with just 37 genes, they have less genetic material than any living bacteria.
A relationship between mitochondria and disease started to become apparent in 1975, when Douglas Wallace and his colleagues, then at Yale University, described an association between mitochondrial DNA and a genetic disorder. During the 1990s, researchers linked the effects of mutations in mitochondrial DNA to various other conditions. One in 5,000 people has an inherited mitochondrial disease of some kind, with consequences that can include diabetes, vision and hearing problems, learning difficulties and other disorders. Only in the last decade or so, however, have scientists seriously explored the influence of mitochondria on mental health and well-being, especially when it comes to stress, anxiety and depression.
Sandi’s work sprang from an intuition that mitochondria might alter the operation of select brain pathways. Our brains eat up 20% of the oxygen our bodies take in, even though the brain accounts for only 2% of our weight. A deficit of cellular energy production in critical neural circuits, she hypothesized, might explain an overall lack of motivation and self-esteem seen in anxiety-prone people.
When Sandi put rats in competition to establish a social hierarchy, she saw that the animals with less anxiety were more likely to acquire dominant rank. Further study showed that these less anxious animals had greater mitochondrial function in the nucleus accumbens, a part of the brain vital to motivated behavior and the production of effort.

Martin Picard, a researcher at Columbia University’s Irving Medical Center, leads a laboratory that studies the involvement of mitochondria in psychobiological phenomena.
Courtesy of Martin Picard
Other research in many laboratories unearthed further ties between stress and mitochondria. In 2018, Picard and the stress research pioneer Bruce McEwen, who died earlier this year, published a meta-analysis of 23 studies on mitochondria and anxiety: 19 demonstrated “significant adverse effects of psychological stress on mitochondria” and even the other four noted changes in mitochondrial size or function in response to stress.
A 2018 review article by Anke Hoffmann of the Museum of Natural History in Berlin and Dietmar Spengler of the Max Planck Institute of Psychiatry in Munich summarized evidence that mitochondria could mediate the brain’s structural and functional responses to early life stress and serve as “a subcellular substrate in the programming process.” The experimental evidence for connections between mitochondrial function and mental health is still tentative and has important limitations, but it is strong enough to convince scientists to look deeper.
The Cross-Talk of Mitochondria
One mystery still under investigation surrounds the details of what happens to mitochondria under stress. Picard’s best guess is that it starts when a trigger for stress makes cells in the adrenal glands release the hormone cortisol. Within those cells, mitochondria synthesize the hormone (with the assistance of another organelle, the endoplasmic reticulum) by converting cholesterol into cortisol. Cortisol then travels throughout the body in the blood. Specialized receptors carry the cortisol into the nuclei of cells, where it activates about 1,000 genes to help cells prepare for the “fight or flight” response. But the receptors also ferry some of the incoming cortisol into the mitochondria, where it interacts with the mitochondrial DNA and makes energy production more efficient.
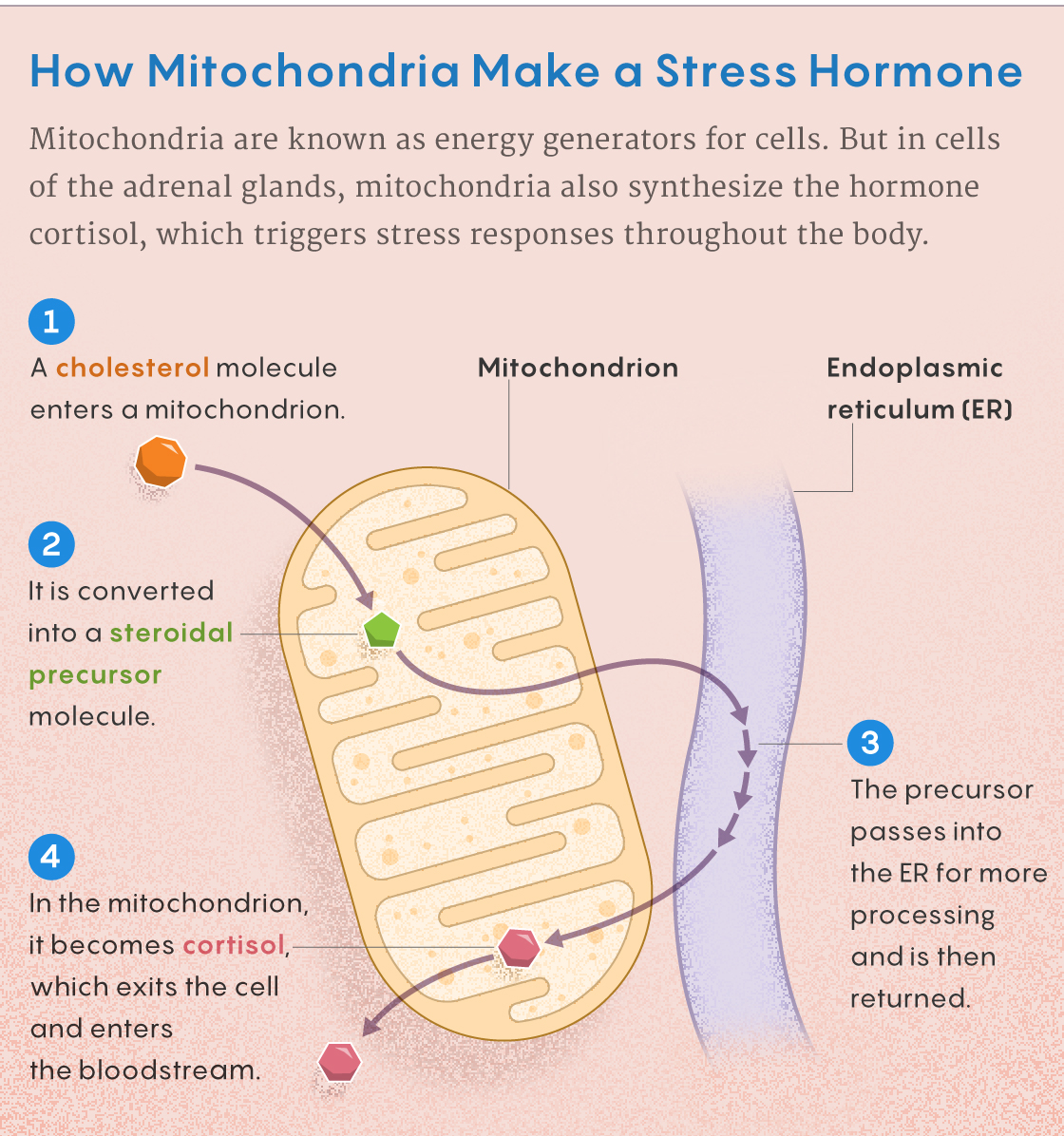
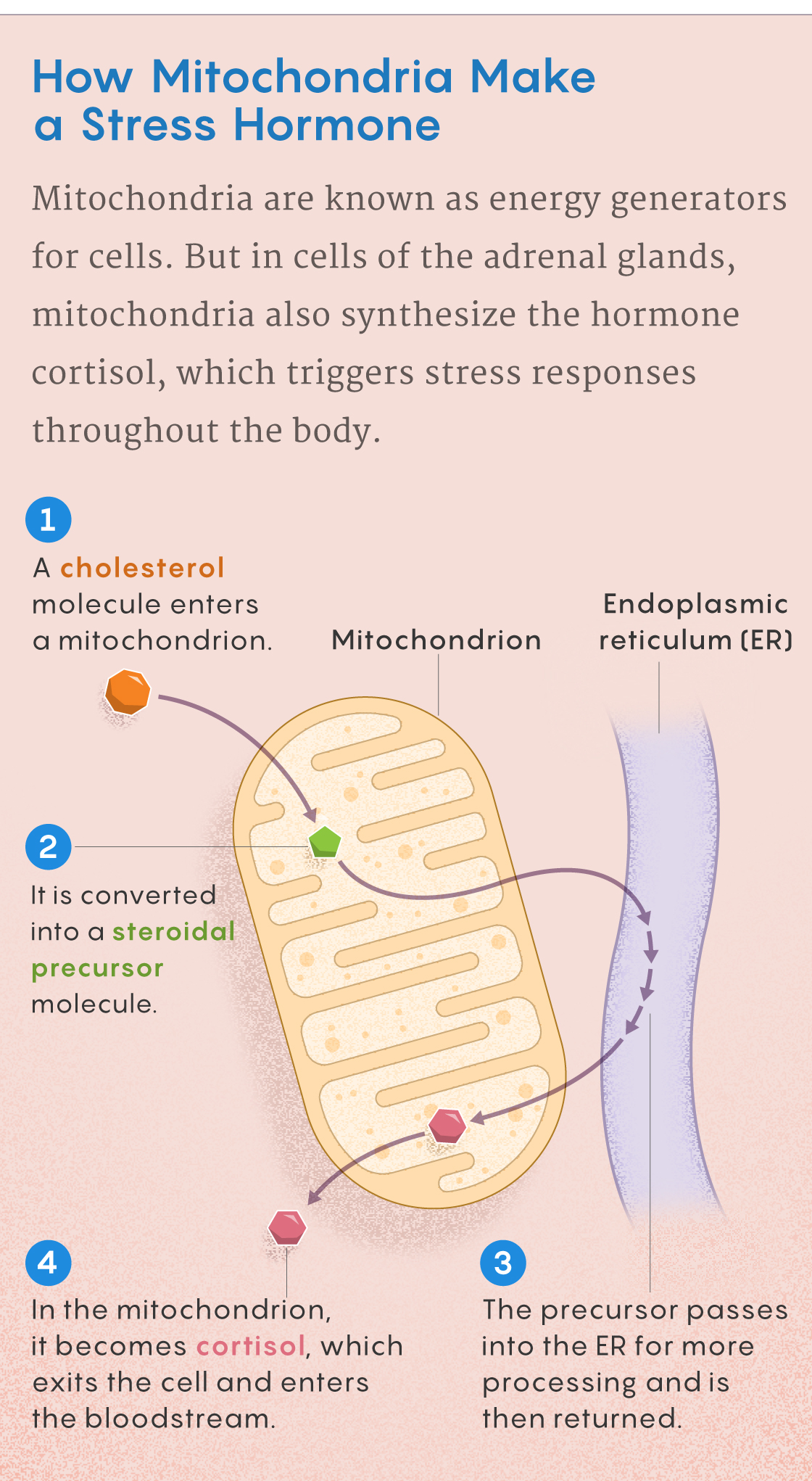
Samuel Velasco/Quanta Magazine
In effect, mitochondria in adrenal glands produce the stress hormone, and it travels to other mitochondria throughout the body, bringing about an integrated stress response. “It creates a beautiful mitochondrial cross-talk between organs that hasn’t been discussed or developed very much,” Picard said.
As the mitochondria adjust in response to those signals, they may change shape, from a bean form to an elongated, spaghettilike structure, split apart or fuse with one another. Disrupting these fusion and fission processes can lead to cell damage and even death. Picard likens this disruption to social isolation — when mitochondria can’t talk to each other, they do worse.
Determining how stress affects the mitochondria in brain cells often involves sacrificing animals — a practice that is obviously out of the question with humans. Nevertheless, the few studies that have found ways to explore the problem in people have found indications of a link.
One was a 2019 study in Psychoneuroendocrinology, led by Caroline Trumpff, a postdoctoral researcher in Picard’s lab. She and her collaborators Anna Marsland and Brett Kaufman at the University of Pittsburgh looked at a small sample of healthy middle-aged adults and found an association between acute psychological stress and a rapid surge in short segments of mitochondrial DNA floating outside cells. Such pieces of mitochondrial DNA are normally released during damaging events related to injury or disease. The effect was more pronounced in men than in women.
Identifying how that kind of stress-related mitochondrial damage occurs is a work in progress. One likely explanation is that overactivity by mitochondria in response to stress makes them generate more of the molecules called reactive oxygen species, which can be toxic for cells, Sandi said.
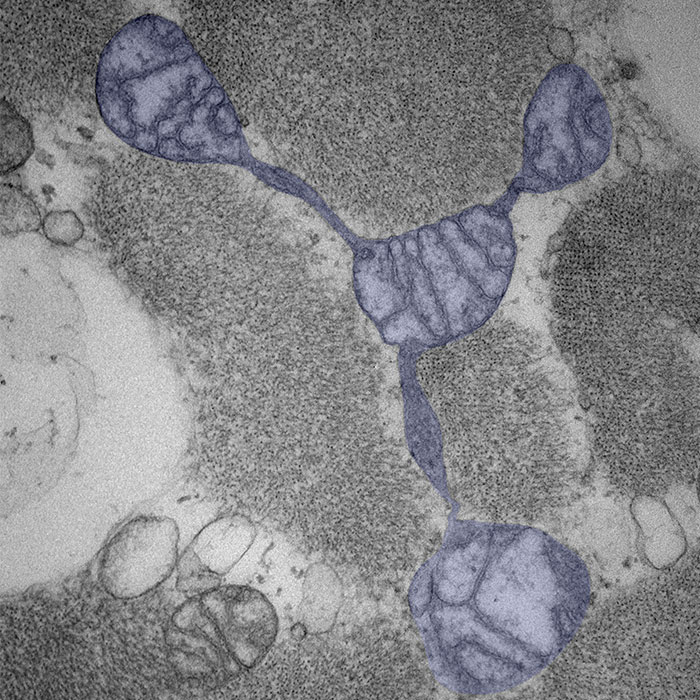
Mitochondria can form interconnections, fuse with one another and split apart in response to a cell’s needs and environmental signals.
Scientists also seem to agree that ATP is involved. “The ability the body has to be sensitive to the outside world, and then to mount a response internally, is all energy-driven,” Picard said. Studies suggest that overly stressed animals may show dampened ATP production, which can impair many processes but especially cell division. This is especially relevant to the hippocampus, a brain structure vital for memory-encoding, learning, emotion — and processing stress. It is one of the few parts of the brain where, even in adult mammals, there is some evidence that new neurons may be created all the time.
And although mitochondria are ubiquitous in the body, their structural and functional diversity within tissues and even within individual cells offers ways in which mitochondrial damage could be felt differently in specific brain pathways, as Carmen Menacho and Alessandro Prigione of Heinrich Heine University in Germany discussed in June in a review in the International Journal of Biochemistry and Cell Biology. Researchers have found that mitochondria are often anchored at key positions within neurons, such as near the synapses, apparently to help their function. Mitochondrial effects might also be exerted through the brain’s non-neuronal glial cells, such as the oligodendrocytes that sheath neurons in myelin and the astrocytes that support neurons’ health. Neurons sometimes eliminate damaged mitochondria by passing them to astrocytes, and astrocytes send them healthy mitochondria. If stress disrupts any of these processes in brain areas such as the nucleus accumbens, it could have an impact on anxiety disorders.
Toward Interventions
The field of psychiatry needs new therapeutic drugs, Kalynchuk notes, and interventions aimed at the mitochondria may have promise for helping people vulnerable to stress, depression and anxiety. Her lab is looking at the potential antidepressant effects of a large extracellular protein called reelin, which provides a scaffold for migrating cells and facilitates intercellular communication. Stress reduces the levels of reelin in laboratory animals, and this drop seems to stem from the mitochondrial response. Early results published in January 2020, in which reelin was tested on male rats, looked promising; Kalynchuk’s group expects to report soon on whether females respond differently.
Sandi’s lab showed in 2017 that the anti-anxiety drug diazepam increases mitochondrial function in rats. She and her colleagues also demonstrated last January that a mitochondria-boosting supplement (acetyl-L-carnitine) protected mice vulnerable to behaviors associated with depression in humans. She is currently collaborating with drug companies on further preclinical investigations into therapies that would boost mitochondrial output, and she is gathering information about metabolites related to mitochondrial function.
Picard is more skeptical about the potential of developing drug therapies aimed at mitochondria. As an alternative, he suggests that behavioral interventions based on mitochondrial function, such as exercise, could be the way to go. Exercise, he says, may be “the best thing you can do for your mitochondria.”
Stress is too complicated a phenomenon to be reduced to a single cause or a simple pathway. Matthew Hirschey of Duke University’s Molecular Physiology Institute suspects that cortisol is a major driver of the relationship between mitochondria and stress and that there are links to metabolism, but also emphasizes that these mechanisms are not the whole story. “Mitochondria are clearly important for proper neurological function — neurons are tremendously active cells that require a lot of energy production to make action potentials, and therefore need mitochondria to do this,” Hirschey wrote in an email. “But if, and how, external psychological stressors influence neuronal mitochondrial function isn’t really known.”
To him, it is more important to see the role of mitochondria as a piece in the much larger puzzle of anxiety and stress physiology. But it is a piece that has energized a growing number of researchers, who will puzzle over it intently for years to come.





