‘Trained Immunity’ Offers Hope in Fight Against Coronavirus
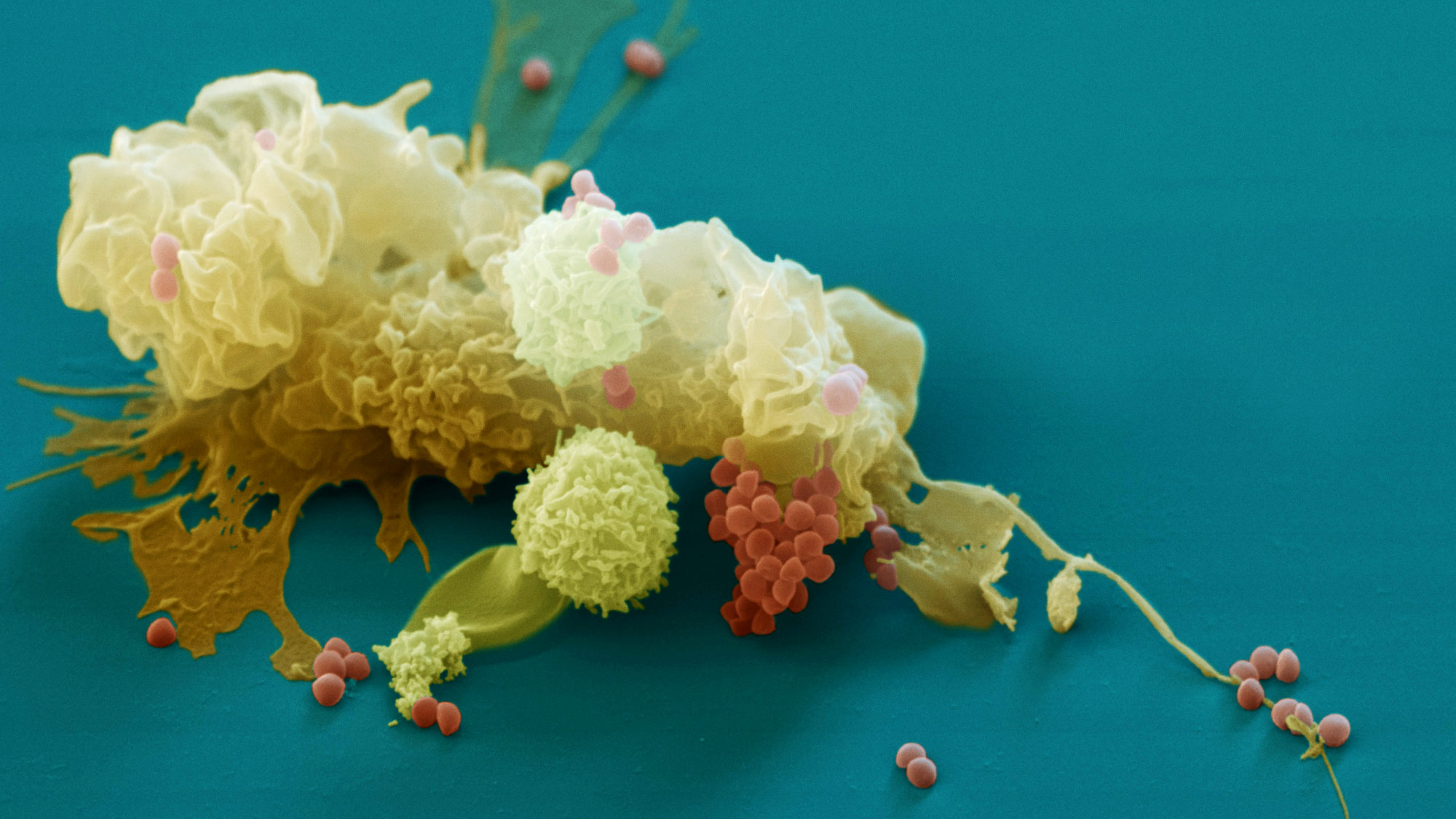
A macrophage (tan) engulfs Staphylococcus bacteria (orange). Macrophages and other cells of the innate immune system scavenge bacteria and other invaders as part of the body’s first line of defense. Research finds that they also contribute unexpectedly to the immune system’s long-term memories of past infections.
Eye of Science / Science Source
Introduction
Laboratories around the world are in a high-profile race to create vaccines that might help end the COVID-19 pandemic. More quietly, however, other scientists are investigating whether a vaccine that has already been in use for decades could also confer some level of protection.
Recent analyses of global epidemiological data by several teams in the United States and in Israel found that in places with higher rates of bacillus Calmette-Guérin (BCG) tuberculosis vaccination, the spread of COVID-19 is slower and pandemic death rates are lower. And in a small study reported in a preprint on August 11, hospital workers who received a booster BCG vaccine in March had no cases of COVID-19 infection, while the infection rate was 8.6% in a comparable unvaccinated group.
Vaccines aren’t supposed to work like that, though, at least according to classical immunology. The tuberculosis bacterium and the SARS-CoV-2 pandemic virus are completely different pathogens, and vaccines are, by design, highly specific. Their specificity is related to their long-lasting effects, because vaccines engage the adaptive branch of the immune system — the B and T lymphocytes and antibodies that recognize a given pathogen. Some of these lymphocytes become “memory cells” that persist for months or years, equipping the body to mount faster, stronger responses if the pathogen ever returns.
“It was thought for a long time that this is the only way in which an immune response remembers an infection, by these memory lymphocytes,” said Mihai Netea, a clinician and infectious disease specialist at Radboud University in the Netherlands.
Netea is one of the scientists challenging that dogma. He has called attention to decades of evidence from epidemiological studies as well as laboratory research in mice, plants and invertebrates, all of which suggests that immunological memory can work in a way that he described in 2011 as “trained immunity.”
Trained immunity is a form of memory exhibited by the innate immune system — a less studied, much older branch of our defenses that evolved more than a half-billion years ago, before vertebrate animals and the adaptive immune system existed. In the past few years, researchers have begun to learn how the innate immune cells, which are fairly nonspecific and short-lived, remember old invaders. Recent work has also found evidence that pathological manifestations of trained immunity may be involved in some chronic inflammatory diseases and neurodegenerative disorders. And in an August 12 Cell Host & Microbe study, an international team that included Netea revealed how the BCG vaccine brings broader health benefits by triggering trained immunity.
“This Is Crazy”
Netea’s introduction to trained immunity came in 2010, when a student intern in his lab was studying how vaccines shape the immune response. Working with blood from volunteers collected before and after BCG shots, the student spiked the samples with the tuberculosis microbe, Mycobacterium tuberculosis. The samples from vaccinated people reacted positively, as expected. As a negative control, she also mixed some samples with the yeast Candida albicans, an irrelevant pathogen that the samples should have ignored.
Except they didn’t. Samples from the first five volunteers reacted to both tuberculosis and Candida. When Netea saw the indiscriminate responses of the first five samples, he told his student, “Maybe it’s a mistake. Just do the next five and take care not to put TB twice.”
But the same thing happened: The samples reacted to both pathogens. “This is crazy,” Netea recalls saying. “Something is wrong.”
Flummoxed, he scoured the scientific literature. To his surprise, he found quite a few reports describing this sort of immune cross-protection. Throughout the history of immunization, going back to the introduction of the smallpox vaccine in the 1800s, some scientists noted that immunizations seemed to guard against more than the disease they were designed for.
For example, in the 1920s it was relatively common for children in northern Sweden to die within their first few years. But among children who received the BCG vaccine at birth, the mortality rate was two-thirds lower — a curious outcome given that tuberculosis generally strikes later in life. The leader of the study, the physician Carl Näslund, speculated about this in a 1932 paper: “One is tempted to explain this very low mortality among vaccinated children by the idea that BCG vaccine provokes a nonspecific immunity,” he wrote.
That hunch found confirmation decades later. Starting in the 1970s and continuing into the early 2000s, epidemiological studies by the Danish researchers Peter Aaby and Christine Stabell Benn found that children vaccinated for measles in Guinea-Bissau and other developing countries had about 70% lower mortality than unvaccinated kids — even though measles itself didn’t cause more than 10-15% of deaths. Data gathered in West Africa and elsewhere during the 1990s also built a case that BCG vaccination, in addition to preventing tuberculosis, protected people from a broad set of infections.
By the late 1980s, researchers in Italy led by Antonio Cassone of the University of Perugia had started working out which cells were responsible for this cross-protection. Infecting mice with a weakened strain of yeast not only protected them against more pathogenic yeast but also helped them fight unrelated Staphylococcus aureus bacteria. Using drugs to selectively disable sets of immune cells in the animals, the researchers pinned the nonspecific protection to the white blood cells called macrophages. And that conclusion posed a real conundrum for immunologists.
Unlike B and T lymphocytes, which take weeks to deploy their high-precision weapons of adaptive immunity, macrophages are like shock troops that rush onto a battleground, waving clubs at all foes. Lymphocytes have receptors that respond to exquisite molecular details on specific pathogens, but macrophages, natural killer (NK) cells, neutrophils and other cells of the innate immune system rely on a blunter, more generic approach. They are equipped with sets of “pattern recognition receptors” that recognize molecular features common to many pathogens or damaged cells.
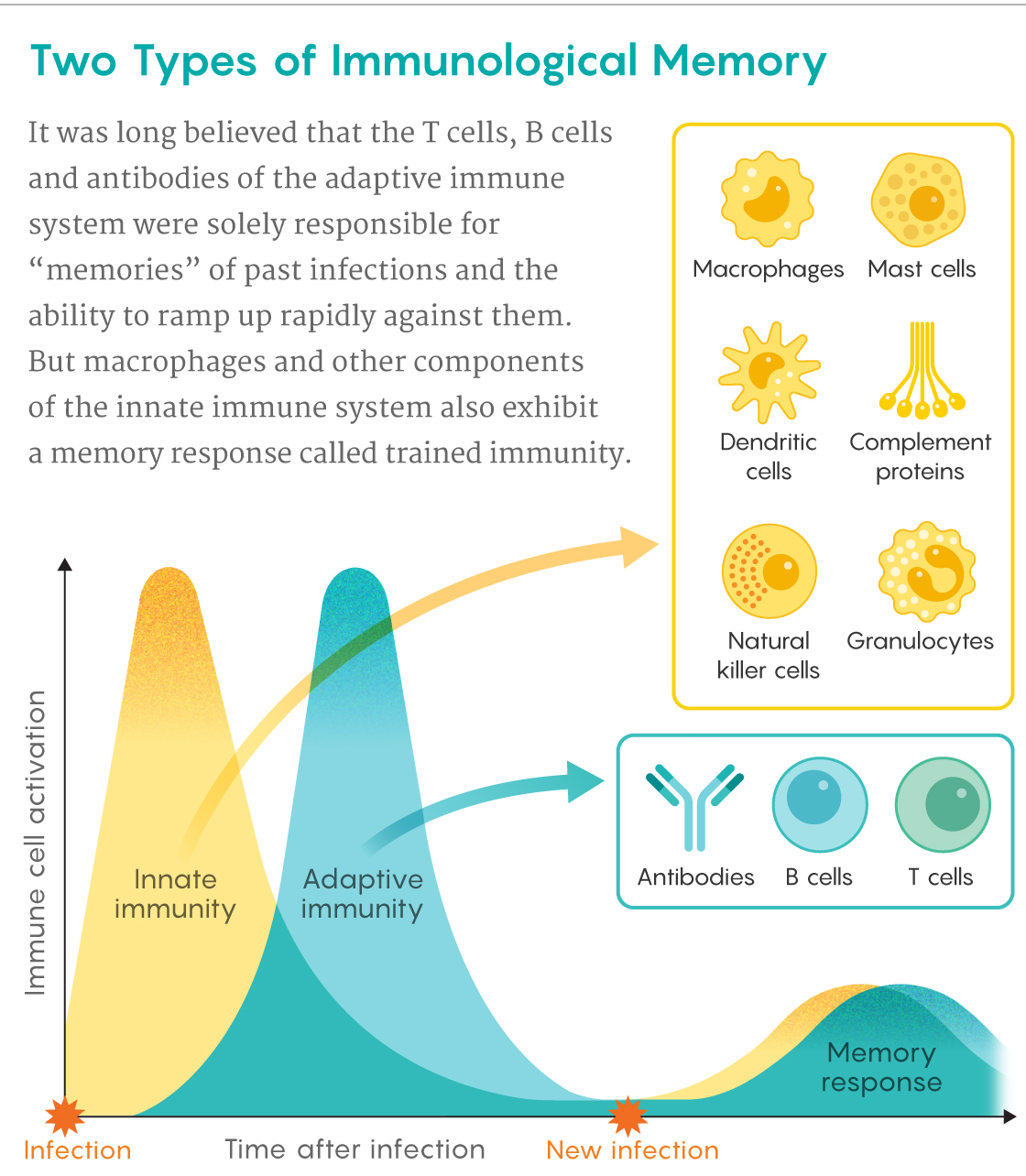
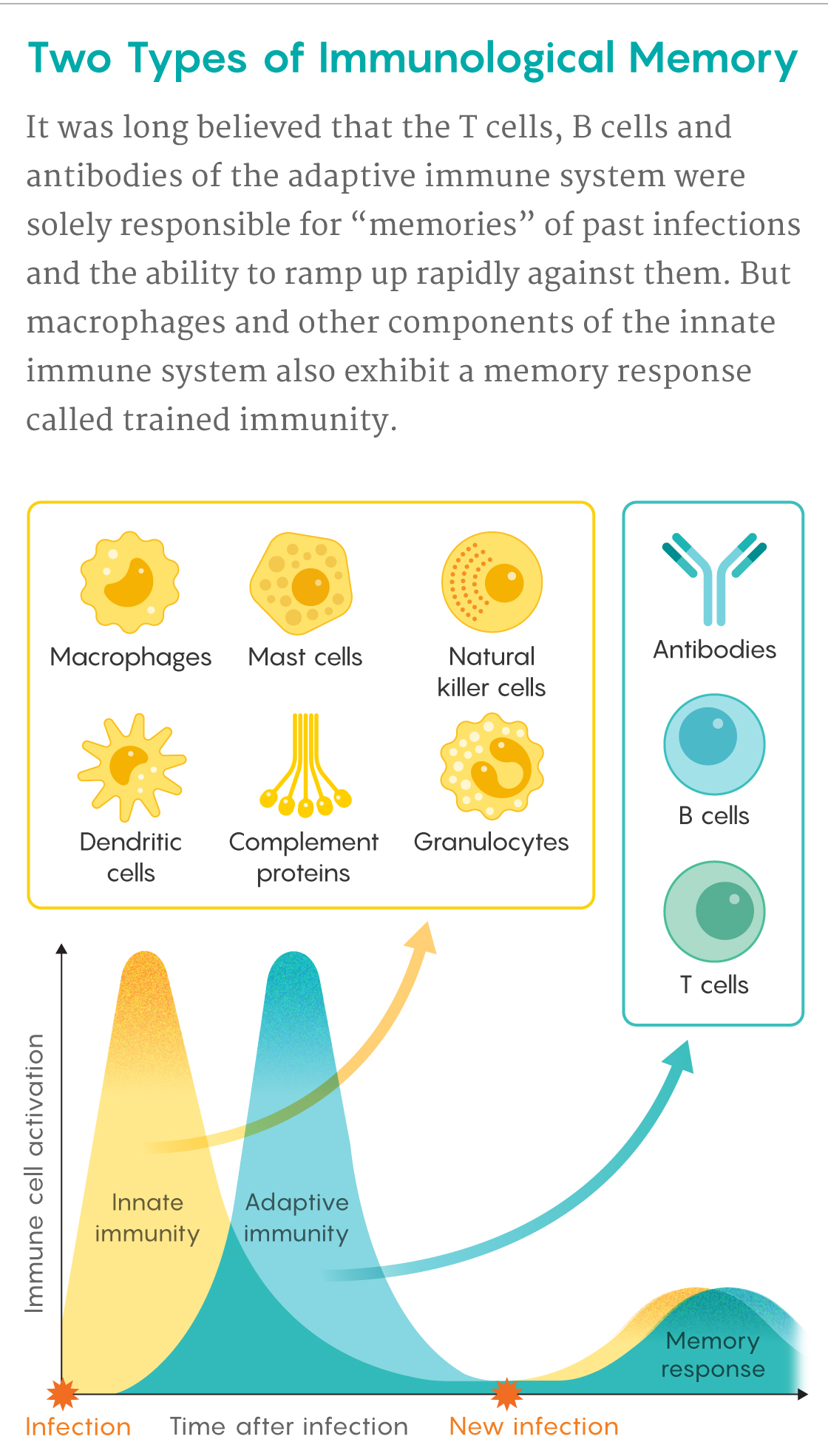
Samuel Velasco/Quanta Magazine
Because of these differences, innate immune cells can speedily pounce on unwelcome intruders and diseased tissue. This can buy time for B and T cells from the adaptive immune system to multiply into an army that can deliver a more precise and devastating assault if one is needed. Later, some of these lymphocytes stick around in the blood and lymph as memory cells, ready to renew the charge if the pathogen resurfaces months or years later. “This very strong memory is what we base vaccines on,” Netea said.
Because T and B cells exist only in vertebrates, scientists believed that immunological memory was unique to them too. It seemed that invertebrate species could get by with innate immune responses alone, since the animals generally didn’t live a long time and could breed rapidly enough to offset deaths from disease.
And there remained this mystery: If macrophages were undiscriminating cells that did little more than gobble up foreign material, how could they be responsible for the enduring and broadly protective effect that the Italian researchers were seeing in their experiments? It didn’t seem to make sense, especially since macrophages live for only a few days or weeks.
This riddle of immune memory that defied general perceptions sat unanswered in the scientific literature, Netea realized. “When we don’t understand something, we tend to forget it,” he said. “That’s why some of the studies were forgotten. But they were important.”
Evidence From Invertebrates
Netea also saw evidence that unorthodox immunological memory might pop up in even less likely places. The literature held reports of memory-like behavior in plants and invertebrates — organisms with no adaptive immune cells.
One of those reports was a landmark 2003 Nature paper by the evolutionary biologist Joachim Kurtz, then at the Max Planck Institute of Limnology in Germany, and his master’s student Karoline Franz. Kurtz and Franz found that tiny crustaceans called copepods got better at warding off parasitic tapeworm larvae with repeated exposure — but the results were inconsistent. The researchers realized that one variable was the source of the parasites. Could it be, Kurtz wondered, that copepods become more resistant to tapeworms from the same family?
The prevailing view at the time was that invertebrate immune systems were incapable of such discernment. Yet in a new round of experiments, copepods clearly resisted sibling tapeworms better than less related ones. “It was against the dogma,” said Kurtz, who now heads a research group at the University of Münster.
That 2003 paper, entitled “Evidence for Memory in Invertebrate Immunity,” irritated some immunologists. “They said ‘immune memory’ is only when you have an adaptive immune system, meaning that you have lymphocytes and antibodies,” said Kurtz. “We said, well, ‘memory’ is more like a broader term.”
Lewis Lanier, an immunologist at the University of California, San Francisco, can sympathize. His lab made headlines in 2009 by showing that in mice, NK cells can learn from past experience. Like Kurtz’s work, the UCSF paper turned heads by ascribing memory-like properties to simple immune cells that lack the diverse antigen receptors of B and T cells. Some researchers “would argue with me about the word ‘memory,’ but they were all convinced that the NK cell remembered its past and worked better when it encountered the virus a second time or third time,” Lanier said. “That they didn’t dispute.”
How Trained Immunity Works
The seeming heresy of these reports of memory in invertebrate immune systems and mouse NK cells paved the way for Netea’s 2011 proposal in Cell Host & Microbe that the innate immune system exhibits trained immunity as a kind of memory of past infections. His paper in the Proceedings of the National Academy of Sciences the next year went further by showing that epigenetic changes are responsible for this training. When macrophages and other innate immune cells respond to pathogens, their DNA gets epigenetic modifications that make it easier to activate the genes that direct the cell to make pattern recognition receptors and disease-fighting proteins. The DNA alterations act like bookmarks that help cells to quickly retrieve those genomic instructions and carry them out — not only “for the infection you saw the first time but any infection,” Netea said.
So if the pathogen returns, the cell is already primed to respond faster. Moreover, when the innate immune cells divide, they pass on these epigenetic DNA bookmarks to their progeny. That is how trained memory can persist while relying on cells that seem so short-lived: The record of the pathogen-fighting experience is passed on from one generation of cells to the next.
Various kinds of immune memory, including some with mechanisms similar to trained immunity, likely also helped invertebrates to survive. And without the earlier studies in invertebrates, “people would probably not have looked for such effects of memory in the innate immune system,” said Kurtz. But researchers studying invertebrates “didn’t have the mechanisms. Vertebrate immunologists, once they realized there is such a phenomenon, have all the tools to study the mechanisms in far more detail than we could ever do it.”
Although trained immunity was originally proposed to describe how innate immune cells remember previous encounters with pathogens, the phenomenon is turning up in cells that aren’t traditionally seen as part of the immune system. In a 2017 mouse study, for example, wounds healed faster in animals that were previously exposed to an inflammatory stimulant. The protection was conferred by epithelial stem cells.
It’s also beginning to look as though trained immunity isn’t limited to offering purely generic protection to the body. This past June in Science, Martin Oberbarnscheidt and Fadi Lakkis of the University of Pittsburgh, Xian Li of the Houston Methodist Research Institute and their colleagues reported that macrophages and some other white blood cells can develop memories for infections keyed to specific major histocompatibility complex proteins, which the adaptive immune system uses to recognize the body’s own cells. The researchers proposed that trained immunity could be an overlooked factor in the rejection of transplanted tissues.
Their results and others point to a possible downside of trained immunity: Some scientists think this enhanced sensitivity in the innate immune system could raise an organism’s susceptibility to autoimmune and hyperproliferative disorders, such as cancer. (Netea, on the other hand, believes that BCG vaccine may offer some protection against cancer, so the jury is still out.) Other research suggests that trained immunity could also contribute to chronic inflammation associated with age-related neurodegeneration, and with chronic liver disease, Type 2 diabetes and other diseases linked to the Western diet.
The trained-immunity connection to possible COVID-19 protection through the BCG vaccine, however, is currently the real attention grabber. Last month, Netea and a team of researchers in Germany, Denmark, Australia and the Netherlands published the results of their research into how the BCG vaccine induces trained immunity. They found that the vaccination sets up epigenetic changes not only in white blood cells circulating through the body but in the progenitors of those cells in the bone marrow that churn out replacements.
What is still uncertain is whether this trained immunity from BCG (or other vaccines) can be harnessed to slow the COVID-19 pandemic. As Netea and Alberto Mantovani of Humanitas University noted in a commentary for The New England Journal of Medicine that appeared last week, it is still not recommended to use BCG vaccine to prevent or treat COVID-19 outside of clinical trials. Such trials are now in progress: Thousands of health care workers in the United States, the Netherlands, Australia and elsewhere are rolling up their sleeves to see if they become any less prone to catching the virus after getting the BCG vaccine. Those studies are scheduled for completion over about the next year and a half. By then, some vaccines specifically targeting the coronavirus may be available. But every bit of protection may still be valuable — and good to know about for future pandemics.
Correction: September 15, 2020
The studies in Italy during the 1980s by Antonio Cassone were originally misattributed to a different researcher.
This article was reprinted in Italian at le Scienze.



